The future of medicine is invisible, frictionless, AI-matched, continuous, and at home
This is not only going to be nice, it's also going to save us a lot of money
It is a Tuesday in 2036. The toilet has already finished a microbiome read and a urine analysis while I brushed my teeth, and the bathroom mirror noticed a small new mole on my left temple that the dermatology AI thinks is benign but wants to recheck in three months. The bed gave me a sleep report at 6 a.m., showing that deep sleep was lighter than usual, probably from the glass of red wine I had with dinner.
I head out for a morning run. By the time I am back, the patch on my arm has logged the BDNF spike from the workout, a 38 percent rise from my pre-run level. BDNF is a growth factor that drives the formation of new connections between brain cells, and exercise is the most reliable way to raise it. The system updates my weekly plasticity score, with today’s run clearing the threshold for my brain health goals.
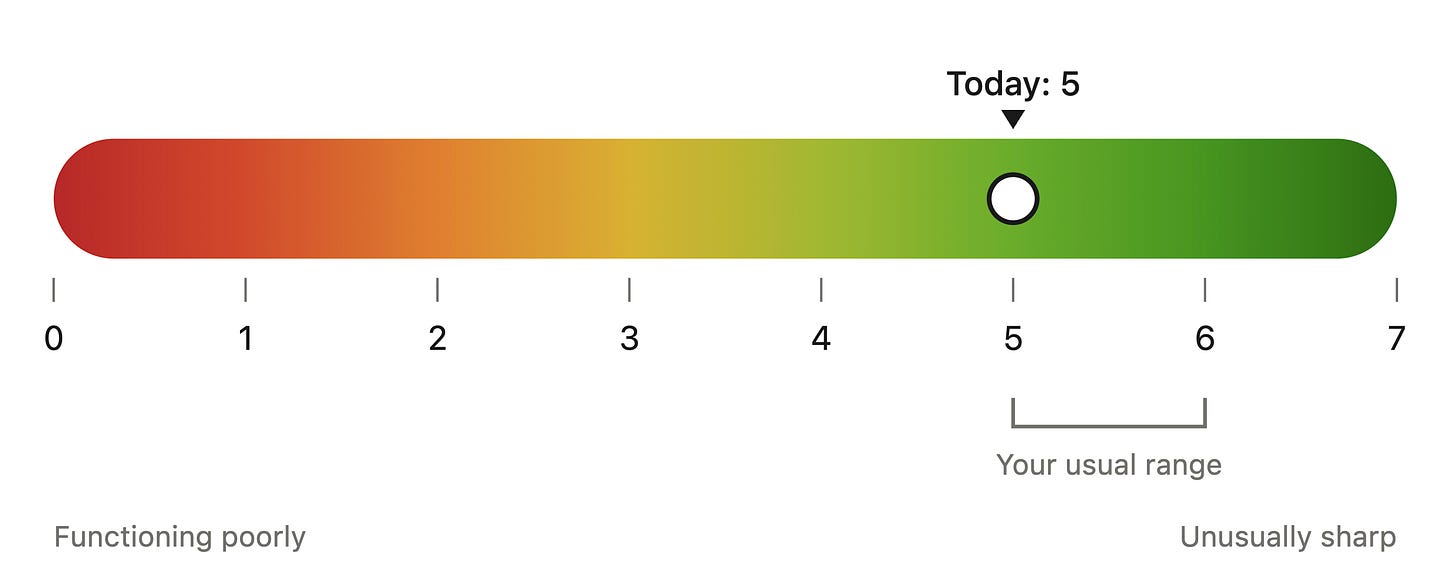
The more important alert came overnight. My GFAP trend, the brain inflammation marker that the patch on my arm has been tracking, crossed the threshold the system flags as worth investigating. GFAP is the protein that astrocytes (the brain’s support cells) release when they are stressed or inflamed, and it has been climbing slowly for nineteen weeks. My sharpness score, the composite cognitive performance index calibrated to me over the past two years, has slipped from my usual 6 to a 5 over the same window. The pattern is the kind of slow drift that, in the old model, would have been invisible until it produced symptoms a decade from now.
While I am making coffee, my phone surfaces the matching engine’s recommendations for what to do about the GFAP drift, ranked by predicted effect. At the top is a Phase 2 trial for a brain-penetrant anti-inflammatory drug that targets the reactive astrocytes producing my signal. Drug delivery, blood draws, and continuous monitoring all happen from my home. My APOE4 status (one copy of the strongest common genetic risk factor for late-onset Alzheimer’s), my GFAP trajectory, my cognitive baseline, and the pattern of slow drift over the past several months match the inclusion criteria, and patients with profiles similar to mine in the database have shown the largest reductions in inflammation markers on this drug class. Enrollment closes in eight weeks.
Below the trial, the engine surfaces lower-risk lifestyle options with smaller predicted effects on my GFAP signature. The trial drug has the highest predicted effect but with unknown side effects from a novel class, while the lifestyle alternatives are gentler and slower-acting and unlikely to fully reverse the drift on their own. The trial protocol lets me continue exercise and lifestyle interventions alongside the drug, and the continuous monitoring will tell me within two weeks whether the drug is engaging my biology. I book the imaging vehicle for my work commute to confirm my brain’s structural eligibility, the imaging-based piece of the inclusion criteria the matching engine cannot verify from continuous data alone.
On the way to work, the self-driving imaging vehicle picks me up at my building. During the thirty-minute commute, the cabin’s portable MRI maps hippocampal volume (the seahorse-shaped memory center that shrinks early in Alzheimer’s), cortical thickness, and white matter integrity. All come back within range, and the imaging report arrives in my inbox before I reach the office.
Every component above either exists today in research or early commercial form, or has a working proof of concept being engineered toward consumer scale. The remaining work is both technical and institutional, and the next decade is the integration. The next decade is when biotech companies gain the chops and advantages of tech companies. Six shifts have to come together for that Tuesday morning to be ordinary:
Diagnostics run continuously through devices you already use, not at scheduled appointments
Biology is measured as real-time streams of dozens of biomarkers, not annual snapshots
Monitoring connects directly to specific recommendations, not just alerts on your phone
AI matches you to treatments that worked for patients with your specific data profile, not population averages
Drugs and doses are chosen to fit your full molecular profile, not the average patient
Clinical trials find you when your data fits, instead of you searching for trials
Diagnostics that disappear into daily life
The first shift is that most measurements stop being scheduled events. Annual physicals, lab orders, and imaging appointments are artifacts of a model where data was scarce and required deliberate collection. In a world of ambient sensing, data accumulates whenever you are using the bathroom, brushing your teeth, sleeping, commuting, walking, typing, or talking. Removing the friction of current medicine, where you have to schedule, travel, wait, and remember, is itself preventive, before any specific test result comes back.
The physiological pieces are arriving. Smart toilets sequence stool and analyze urine on every visit. Bathroom mirrors scan skin for new moles, the color of the whites of the eyes, and facial asymmetry. Bed sensors capture nightly heart rate, breathing rate, and sleep stages without a wearable. Phone cameras with clip-on lenses photograph the back of the eye, where early Alzheimer’s, diabetic, and cardiovascular changes appear before symptoms. Self-driving cars analyze breath through cabin air for chemical signatures of metabolic state and early disease, and cabin cameras track pupil response and signs of fatigue that wearables cannot see. Sleep masks that read brain activity capture sleep architecture and deep-sleep brain waves. Continuous cuffless blood pressure tracks the nighttime drop in blood pressure that single office readings miss. Failure of blood pressure to fall during sleep, called non-dipping, is a strong predictor of stroke and dementia risk independent of daytime readings, and it is invisible to standard 9 a.m. cuff measurements.
The cognitive and behavioral pieces arrive through devices people already carry. Voice patterns from everyday calls track cognitive decline, depression, and Parkinson’s. Keystroke dynamics, including how long each key is pressed, the time between keys, error rate, and backspace frequency, form a signature that varies with cognitive load and fatigue. The Neuroqwerty work from MIT showed that Parkinson’s signatures appear in typing patterns years before clinical diagnosis. Self-driving cars capture reaction time on takeover prompts, when the car asks the human to resume control, and attention through eye tracking on the cabin display, turning what was once dead commute time into passive cognitive monitoring.
Even imaging is shifting toward life rather than life shifting toward imaging. A subway car with a low-field MRI in the corner during a commute. Hyberberic oxygen showers and red light therapy cars. A dental chair that captures a low-dose head and neck CT during a routine cleaning, picking up early plaque buildup in the neck arteries or sinus disease in a visit you were going to attend anyway. A manicure that doubles as imaging of the small blood vessels at the base of your nails plus a skin scan for accumulated aging markers, picking up early small-vessel damage and signs of cellular aging from years of high blood sugar, during the thirty minutes you are sitting still with your hands extended. Medicine should fit into your life rather than the reverse.
The continuous data layer
The second shift is from snapshots to streams. Continuous glucose monitors were the first proof that biology is much more dynamic than annual labs suggest, and that real-time feedback changes behavior in ways retrospective data cannot. The next generation extends this to everything that matters.
Continuous lactate, ketones, cortisol, electrolytes, and hydration tell you about metabolic state in real time. Continuous inflammation markers including CRP, IL-6, and TNF-alpha catch infections before symptoms and chronic low-grade inflammation that drives aging. Continuous hormone tracking captures the actual daily and monthly patterns rather than the morning serum draw that captures a single point. Continuous tracking of toxin and pollutant exposure builds a personal record of what your body has encountered. Continuous biological age tracking makes the aging clock a daily readout rather than an annual one.
Continuous neurotransmitter chemistry shows up in peripheral fluids. Smart toilets capture the breakdown products of serotonin, dopamine, and the stress hormones adrenaline and noradrenaline in urine. Microneedle patches capture tryptophan, the building block for serotonin, along with the inflammatory byproducts that compete for it when the body is inflamed. SSRI response, exercise effects on mood, and the cognitive cost of chronic inflammation become measurable on a daily timescale.
Real time blood protein tracking is technically more difficult but coming. The patch on my arm reads three at very low concentrations every few hours: GFAP, BDNF, and Klotho. Klotho is a longevity protein that declines with age and supports brain and overall healthspan.
Continuous sensing of proteins like these exists today only in research labs, since they circulate far below what conventional sensors detect, but the recognition chemistry is mostly solved. Aptamers, short DNA sequences that fold around a target protein and produce an electrical signal at an electrode, can be raised against almost any purified protein in a few weeks of selection. A GFAP-specific aptamer was published in 2025, and a high-affinity BDNF aptamer was reported in 2024. The Plaxco group at UC Santa Barbara has shown week-long continuous in vivo measurements using these sensors. Aptamer-based graphene transistors have already demonstrated GFAP detection in patient plasma at concentrations far below physiological levels. The pieces exist in different labs, and the work is the integration.
A continuous brain biomarker patch is plausible on a five to ten year horizon. The realistic interim architecture is a patch with tiny needles sampling the fluid just under your skin into a small chip with sensors for ten to twenty key proteins, replaced every seven days. Without this layer, my GFAP drift would never have been visible.
The sharpness meter
The unifying output of the digital biomarker layer is a sharpness meter, a continuous cognitive performance index displayed as a red-to-green band on the watch face. The scale runs from 0, the red end where you are functioning poorly, to 7, the deep green end where you are unusually sharp. Most people hover around 5 to 6 in their normal range, with the specific numbers calibrated to your personal baseline. It builds the way a good wearable HRV reading does. You wear the sensors for a few weeks while the system learns your personal range, your daily rhythm, and your normal day-to-day fluctuation. After that, every channel produces a score relative to your baseline, the channels are weighted by their validated correlation with formal cognitive testing, and the composite is smoothed over a moving window of an hour or so.
The inputs are layered. Keystroke timing is the most stable channel for short-window detection, since most people type hundreds of words a day in patterns the system can fingerprint within a week. Voice patterns from incidental phone use add a second channel and are sensitive to fatigue, alcohol, and early signs of brain disease. Eye movement speed from the front camera captures attention, reading speed inferred from scroll patterns captures cognitive load, and reaction time on self-driving car takeover prompts captures real-world cognitive function. The molecular layer feeds in too, with BDNF and inflammation markers from the patch, glucose stability from the CGM, sleep quality from the previous night, and HRV from the watch all contributing weighted inputs.
The output is useful in three modes. The instantaneous reading tells you whether you are sharp enough for the deep work you have planned this morning. The trajectory over weeks tells you whether your cognitive baseline is rising, holding, or drifting. The pattern recognition over months identifies the upstream drivers of your personal fog episodes, including poor sleep, alcohol, blood-sugar-spiking meals, specific stressors, infections, or specific medications.
This is the layer that flagged my drift. The slow shift from a 6 to a 5 over nineteen weeks never crossed an acute threshold and no single bad day stood out, but the pattern became visible because the system was watching the trajectory rather than the snapshot.
The same architecture works for mood and anxiety. Reduced total sleep time, fragmented sleep, reduced HRV, reduced step count, smaller geographic radius, more time stationary, increased night-time screen time, slower speech, reduced pitch variability, more first-person singular pronouns in messages, a flattened daily cortisol rhythm, and elevated IL-6 are all validated digital and molecular biomarkers of depression. Aggregated and calibrated to personal baseline, they catch a downward trajectory at week two rather than at week eight.
From data to action
The third shift is closing the loop between physiological state and behavior. Continuous monitoring without a matched intervention layer is just better surveillance.
The joint trajectory of GFAP, BDNF, Klotho, sharpness, and mood says something different about a season than any annual draw could. GFAP rises with brain inflammation, head impacts, sleep deprivation, alcohol, infection, and air pollution exposure. BDNF responds acutely to exercise and tracks plasticity capacity over weeks. Klotho responds slowly to sustained training, vitamin D status, and heat exposure. Layered with the sharpness meter and mood index, the protein signatures distinguish acute fluctuation from underlying drift.
My signature is specific, with GFAP up, sharpness down, BDNF holding steady, and Klotho mildly down. The pattern fits chronic brain inflammation rather than reduced plasticity capacity, which points toward different interventions than if BDNF had been the primary driver. The data suggests checking my recent environmental exposures for inflammatory drivers, screening for low-grade infection, looking at recent air quality data, and considering anti-inflammatory interventions targeting the astrocyte response.
The current model of brain health asks you to do exercise, sleep, diet, and stress management because population studies say they help, and find out decades later whether your particular biology cooperated. The continuous model collapses that timescale. The behaviors are the same, but each one acquires a personal effect size you can see within days, and the protocol becomes individually optimized rather than guideline-based.
This matters most for APOE4 carriers, who have a different baseline biology than non-carriers and respond differently to interventions. Building a personally validated prevention plan is the difference between hopeful prevention and engineered prevention. This is the framework that NeuroAge Therapeutics has been building toward, where multi-modal brain biomarkers integrated across imaging, RNA, genetics, and cognition produce a personal aging trajectory rather than a population estimate. The same data substrate feeds therapeutic discovery targeting the biological signatures the cohort reveals.
What worked for people like you
The fourth shift is from individual trial-and-error to causal inference at scale. Data from millions of continuously monitored patients lets the system see which interventions actually moved the markers in people like you, rather than which ones merely correlated with success. The current model treats every patient as starting from scratch, with guidelines derived from population averages and minimal use of the accumulated experience of every other patient who looked like you, tried what you are about to try, and succeeded or failed. Pattern matching inverts this.
Once enough patients are continuously monitored across genetic, biochemical, behavioral, and clinical dimensions, you become a point in a high-dimensional space populated by millions of others. Some of them are close to you on the dimensions that matter, including your genetics, your baseline biomarkers, your phenotype, your environmental exposures, your sex, your age, and the specific pattern of dysregulation you are trying to correct. The system identifies that cluster, sees what interventions moved their markers toward optimal, and ranks those interventions by predicted effect size for someone with your specific signature.
The architecture is the same as the recommendation engines on Netflix and Spotify. Each user is represented as a vector in a high-dimensional space, similar users are identified, and the system surfaces what those similar users responded to. The mathematics has been well-developed for two decades. The constraint in healthcare has not been the algorithms but the data.
For my GFAP drift, the matching engine shows that among similar APOE4 carriers in the database, the largest reductions in GFAP came from a new class of brain-penetrant anti-inflammatory drugs targeting astrocyte reactivity, the same class being tested in the Phase 2 trial my phone had surfaced that morning. The next-best response came from lion’s mane plus structured aerobic exercise plus a Mediterranean diet. TMS plus exercise showed reliable mid-sized effects. Sauna five times per week showed smaller GFAP reductions and larger Klotho gains.
The implications for behavior change are substantial. Generic advice to exercise more, sleep better, and eat well is hard to follow because the dose-response curves are abstract. Personalized advice that says “patients with your profile who improved their Klotho the most over a year did 150 minutes per week of moderate-intensity cardio plus two resistance sessions, took 5,000 IU of vitamin D, kept HbA1c below 5.4, and spent four weekly sessions in sauna over 80 degrees Celsius” is a more actionable target. The pattern is built from real people who succeeded.
The ongoing learning compounds. Every patient who tries an intervention and has their markers tracked continuously contributes to the database. Drugs that work only in subgroups become identifiable as such, rather than labeled as moderately effective on average. Combinations no individual physician would have thought to try emerge from the data because the matching engine is not constrained by the boundaries of any one specialty.
This also reframes what diversity in the monitoring layer means. A pattern-matching engine trained on patients who skew younger, wealthier, and tech-literate produces recommendations that work best for those patients. The diversity of the input data determines the quality of the matching for each subpopulation. Decentralized monitoring infrastructure that reaches rural patients, older patients, and underrepresented groups becomes important not just for fairness but for the technical quality of the system itself.
Drugs matched to your biology
The fifth shift is in how prescriptions get made. The personalized model uses the matching engine described above and adds the multi-omic dosing layer that the recommendation alone cannot capture.
The genetic layer is the most mature. The Clinical Pharmacogenetics Implementation Consortium already has dosing guidelines for over a hundred drug-gene pairs, and most patients never get tested. In the future model, your genome is sequenced once and every prescription is filtered through it automatically for both efficacy and adverse event risk. The other omic layers add resolution beyond what genetics alone can provide. Gene activity from RNA panels captures how the genome is actually being expressed in your tissues at a given moment, which can shift with age, stress, and disease. Protein levels capture the actual signaling state. Gut microbe composition captures how oral drugs are metabolized before they reach the bloodstream and how systemic inflammation is being modulated. Metabolite profiles capture the downstream consequences of all of the above. Drugs that work in 30 percent of patients on average might work in 90 percent of patients with the right multi-omic signature.
The prescription experience changes too. Algorithmic recommendations come from the data continuously. Coaches help patients understand what the data is showing and support sustained behavior change, but the prescribing, dosing, and monitoring decisions are surfaced from the matching engine in real time. Compounding pharmacies deliver personalized doses calibrated to your weight, metabolism, and full molecular profile. The same continuous monitoring that justified the prescription tracks whether the drug is working. If GFAP stays elevated despite three weeks of intervention, or the sharpness meter fails to recover, or the mood index keeps drifting after a sleep protocol, the algorithm flags non-response and suggests the next-best match. The model of “try this for three months and come back” becomes “we know within two weeks whether this is engaging your biology.”
Clinical trials as a default option
The sixth shift is in clinical trials. About 80 percent of trials fail to enroll on time, and the recruitment costs run into the billions per year. Patients who would benefit rarely hear about trials that match them, and most physicians do not know what is open for their patients. The screening process is laborious, geography excludes the majority, and cost and time exclude most working people.
The pieces of a fix exist. Continuous biological profiling generates the patient signature. Continuous digital biomarkers including the sharpness meter and mood index provide cognitive and mood baselines that meet most trial inclusion criteria without a separate screening visit. Natural language processing parses trial criteria from ClinicalTrials.gov into machine-readable form. Decentralized trial infrastructure, including home blood draws, home blood collection devices like the Tasso, telemedicine visits, wearable data ingestion, portable imaging, and home medication delivery, lets most trial visits happen wherever the patient lives.
The result is that trial opportunities flow to patients automatically. Your profile is checked against active trial criteria daily. When a match appears, you get the notification I described in the opening scene. The trial visits happen at home or at a nearby site. Continuous data flows back to the sponsor in real time, accelerating Phase 2 readouts from three years to eighteen months.
This is also the dimension on which personalized medicine could become a force for equity rather than its opposite. Today’s trials underrepresent women, older adults, rural patients, and racial and ethnic minorities. The drugs we approve are tested mostly on populations that do not match the patients who eventually take them. Decentralized trials with continuous data streams could enroll the populations that current trials systematically exclude.
So I tap, and the trial protocol opens. Inclusion criteria are met without a screening visit, the visits will happen at home except for the initial dose, and the first follow-up draw is in two weeks, with my patch streaming data to the sponsor continuously. I register my interest, and the site contacts me within the hour.
The infrastructure underneath
Uber did not invent ride-hailing. The cars existed and so did the roads, the drivers, and the demand. What Uber built was the platform layer that connected drivers, riders, GPS, mobile payments, and dispatch into a coherent system.
Personalized medicine is structured the same way. The sensors, molecular technology, drugs, trials, and patients all exist. What is missing is the integration layer that connects them, and the integration is happening one vertical at a time.
The model is vertical specialists running matching engines on consented data. Tempus has built this for oncology by aggregating multi-modal cancer data from hundreds of thousands of patients and using it to power clinical decision support, trial matching, and therapeutic discovery. Flatiron and Foundation Medicine have done similar work in community oncology and cancer genomics. Each company runs a defensible engine inside its vertical because the data accumulates with scale and the network effect is real. Brain aging needs the same thing, and this is the layer NeuroAge Therapeutics is building toward, where multi-modal brain biomarkers from a large cohort become the substrate for personalized aging trajectories, trial matching, and the therapeutic discovery flowing out of the same dataset. Cardiometabolic disease, autoimmune disease, mental health, and rare disease all need their own equivalents.
The cross-vertical layer that I would have expected to require interoperability standards turns out to mostly run on patient uploads. When the value proposition is clear and their data is privacy protected, patients freely upload their data. Function Health, InsideTracker, Levels, and similar platforms already operate this way. Patients upload their cardiovascular reports, lab panels, wearable exports, genetic data, and PDFs of clinic visit notes because doing so gives them better insight into their own health. The HIPAA right of access and the 21st Century Cures Act information blocking provisions give them the legal hook to obtain the records, and motivation closes the loop. A brain aging matching engine that wants cardiovascular context does not need an industry-wide data standard, just a clear upload path and a reason for the upload to be worth the patient’s time.
The Uber of personalized medicine is distributed across vertical specialists rather than concentrated in one company. Each specialist builds a matching engine on its own clinical assays plus patient-uploaded data. The companies that own the data layer for a high-value condition become the platform for that condition, much as Tempus owns that position in oncology. The version of personalized medicine that gets built is the one where every major condition has its own well-funded vertical, and where patients can carry a PDF or a CSV from one vertical to another whenever they want a richer picture.
Why this is taking time
The system is being built piece by piece, and each piece is at a different stage of maturation, with the curves accelerating across all of them.
The vertical specialist model has proven itself in oncology with Tempus and Foundation Medicine, and the equivalent companies for brain aging, cardiometabolic disease, autoimmune disease, and mental health are now being built. Each one is accumulating the patient data and clinical context that makes its matching engine useful, and the curves are accelerating as AI methods reduce the data volume needed to reach clinical utility.
The continuous biomarker layer is moving from research labs to consumer engineering. Aptamer-based protein sensing works in research demonstrations for GFAP, BDNF, Klotho, and dozens of other targets, and the transition to consumer-grade patches with multi-week reliability is the active engineering frontier. The same trajectory played out with continuous glucose monitoring over the past fifteen years, and the playbook is now well understood.
Patient awareness is growing fast through direct-to-consumer genetic testing, longevity clinics, and tools like Function Health and InsideTracker. APOE4 carriers increasingly know their genotype, and the population that wants continuous biomarker tracking is expanding well beyond the early adopter base. Demand pull is now meaningful enough to attract capital and engineering talent.
The capital allocation pattern is shifting too. The vertical specialist position requires longitudinal patient data infrastructure plus clinical assays plus AI, a hybrid profile that did not fit cleanly into standard healthcare VC categories. Tempus has now proven the model can reach public-market scale, and the resulting investor pattern recognition is unlocking funding for the next wave of vertical specialists.
A common objection to this hybrid is “pick a lane” between diagnostics and therapeutics. The implicit assumption is that one company should measure the biology and another should drug it. The framing is backwards. The diagnostics infrastructure of continuous biomarker readouts, longitudinal patient cohorts, and AI matching is precisely what makes drug discovery faster and cheaper, because the data layer identifies which biological signatures matter and which patients respond to which interventions. Drug development flows out of this infrastructure, not parallel to it. Tempus has proven the model works in oncology, where their diagnostic platform feeds their therapeutic discovery work, and the next decade of medicine depends on the same integration being funded for brain aging, cardiometabolic disease, autoimmune disease, and mental health.
Reimbursement follows clinical evidence, and the evidence is being generated. CGM coverage expanded from Type 1 diabetes only in 2017 to all insulin-treated diabetics by 2023, a six-year shift driven by accumulated outcomes data. Continuous brain biomarker patches and multi-omic monitoring will follow the same trajectory once they hit the market and the cohorts demonstrating cost-benefit are built. The interim model of patients paying out of pocket through services like Function Health and Levels has validated demand and is generating the early evidence base. The Tuesday morning above becomes ordinary as reimbursement opens for monitoring before disease, employers add this to wellness benefits, and hardware costs come down with scale.
What this would save
The total addressable savings from a fully realized version of this vision is somewhere in the $1 to $2 trillion per year range, or roughly 20 to 40 percent of current US healthcare spending. The savings split roughly evenly between direct services that get eliminated, administrative overhead that disappears, and downstream costs that get prevented.
The breakdown:
$440 to $660 billion per year in direct services. US physician and clinical services spending was $1.1 trillion in 2024. Routine primary care and specialist follow-up visits, the visits most replaceable by continuous monitoring and AI-mediated interpretation, represent roughly 40 to 60 percent of that spending.
$200 to $300 billion per year in administrative overhead. Billing and insurance-related costs in US healthcare have been estimated at around $500 billion, with provider overhead alone accounting for $282 billion. Removing the appointment-and-billing scaffolding eliminates most of the provider overhead and a meaningful share of the payer overhead.
$40 billion or more per year in patient time. Around 860 million physician office visits happen in the US annually, each consuming 1 to 4 hours of patient time including travel, waiting, and follow-up. At median wage rates that is more than $40 billion per year in unpaid patient time, plus indirect costs from missed work, childcare, and care avoidance.
$500 billion or more per year in downstream prevention. Earlier detection of major chronic conditions reduces the much higher costs of late-stage treatment. The cancer case is the clearest example, where shifting late-stage cancer rates from roughly 20 percent to 2 percent through risk-stratified screening, multi-cancer early detection blood tests, and enhanced surveillance saves an estimated $80 to $150 billion per year alone, given that first-year Stage IV cancer treatment costs run 1.6 to 7.7 times higher than Stage I across 17 cancer types in Medicare data. Applied across cardiovascular disease, diabetes, kidney disease, and dementia, total avoidable downstream spending sits in the 10 to 20 percent of total healthcare spending range, or $530 billion to $1 trillion annually.
$30 billion or more per year in faster, cheaper drug development. Bringing a single drug to market currently costs around $2.6 billion, with 80 percent of trials failing to enroll on time and patient recruitment alone accounting for 40 percent of trial budgets. When matching engines auto-identify candidates from continuous data and trials run from the patient’s home, recruitment costs collapse, trial duration compresses by years, and the same R&D budget brings more drugs to market.
The downstream effects on drug pricing and pipeline diversity are even larger than the direct savings. With cheaper and faster trials, the same R&D budget can advance more candidates through development, including for rare diseases that historically failed to recruit enough patients to run a trial at all. Earlier market entry also means longer effective patent life, which gives sponsors room to price drugs lower while still recovering their development costs.
The work ahead
The engineering work is to bring continuous protein sensing, the at-home diagnostics, and the integration layer from research labs and early prototypes to consumer scale. The institutional work is to build payer reimbursement frameworks for continuous monitoring and biomarker panels in asymptomatic high-risk patients, regulatory pathways for novel methods like continuous-monitoring as a primary endpoint, and the patient and capital flows that let new vertical specialists reach the scale where their matching engines become useful.
The constraints loosen each other. A vertical specialist that proves the model in one domain pulls capital and patients toward neighboring domains. Continuous protein sensing that works at consumer scale for one analyte makes the engineering tractable for many. Patient awareness built by direct-to-consumer health platforms creates demand pull, and payer reimbursement follows the cost-benefit data that scaled cohorts generate.
Four months later in this story, my GFAP trajectory has reversed. The patch shows the protein returning to my baseline range, my sharpness has recovered, and the matching engine has updated my predicted aging curve to a slower one. Catching the drift at week nineteen instead of waiting for symptoms a decade later is what this whole system is built for.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso