BioAge’s Longevity Drug Cleared Phase 1 with an 86% Drop in Inflammation
Inflammaging, CRP, and the longevity drugs coming to reduce them
BGE-102, the lead asset at BioAge Labs, is an oral inhibitor of the NLRP3 inflammasome, an inflammation pathway underlying many age-related conditions. The Phase 1 trial enrolled people with obesity and elevated inflammation. Those taking 60 milligrams once daily for three weeks saw their high-sensitivity C-reactive protein, the standard blood marker of inflammation, drop by a median of 86%. The 120 milligram dose performed almost identically. Side effects were mild, with no serious adverse events and no patient stopping treatment early.
BioAge is one of a small number of biotechs that picks drug targets by analyzing what changes in human aging itself, then works backward to indications. BGE-102 is the first of their programs to reach this stage of human data, which makes the Phase 1 result a clinical test of whether the inflammation pathway running through aging biology can be moved with a drug.
How the NLRP3 inflammasome works
Inside immune cells, there is a sensor protein called NLRP3 that acts as the cell’s smoke detector, responding to a wide range of cellular stress signals rather than to any single specific threat. The triggers it picks up on include cholesterol crystals in artery walls, amyloid plaques in the brain, uric acid crystals in joints, oxidized fats, and damaged mitochondria releasing their contents.
When NLRP3 detects one of these triggers, it assembles into a larger structure called the inflammasome. The inflammasome activates an enzyme that cleaves an inactive precursor of interleukin-1 beta, a powerful inflammation signal, into its active form. The same enzyme also punches holes in the cell membrane, releasing the inflammation signals and causing the cell to break open in a process that recruits more immune cells to the site.
This system evolved as part of the rapid response arm of the immune system. When functioning correctly, it amplifies a real danger into a recruitment signal that brings other immune cells where they are needed. When functioning incorrectly, persistently activated at low levels in tissues with no acute threat, it drives the chronic inflammation that contributes to most age-related disease.
How NLRP3 connects to normal aging
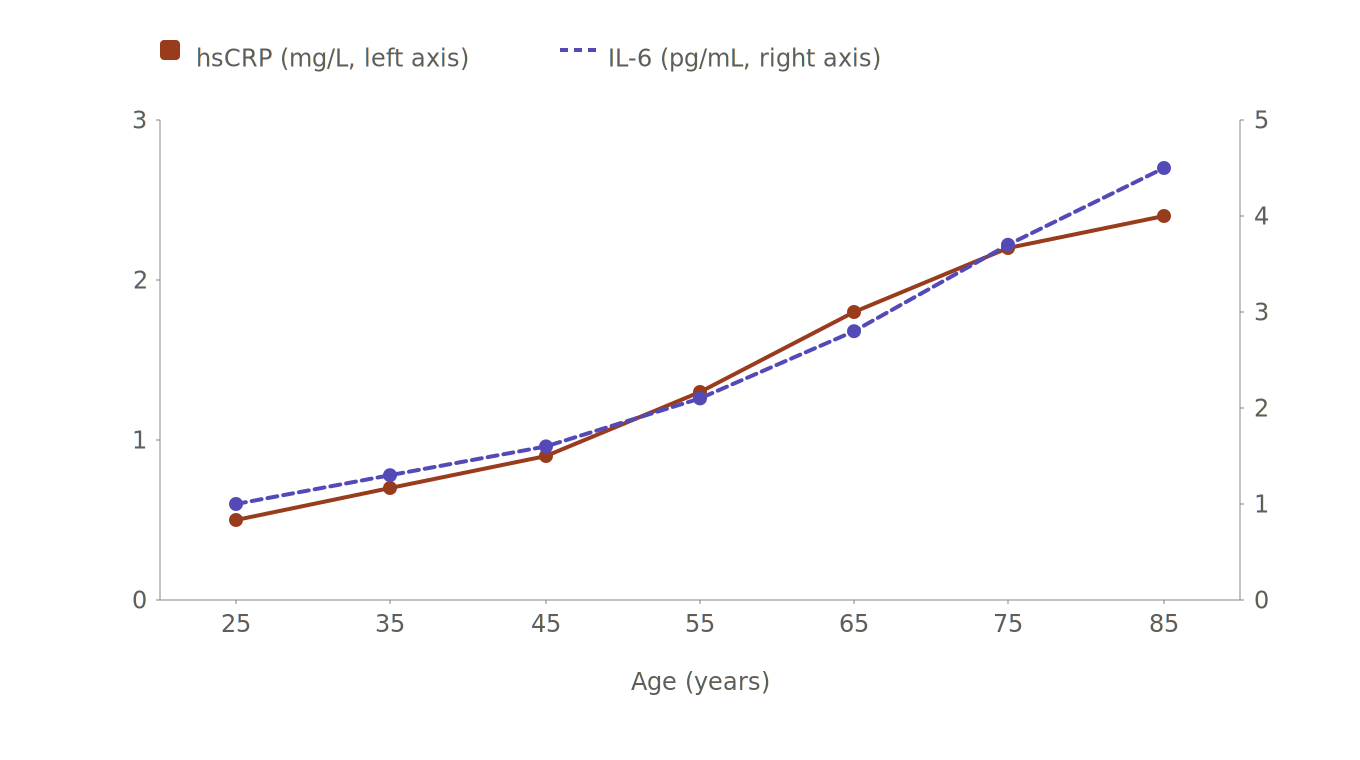
The reason BioAge is targeting this pathway runs through what is called inflammaging, a term coined by Dr. Claudio Franceschi for the chronic low-grade inflammation that accompanies aging in nearly every animal studied. Most people over 60 have elevated CRP, IL-6, and IL-1 even when they appear healthy. The inflammation is not driven by infection but by the accumulation of cellular debris and stress signals that the immune system reads as danger.
Approximate median hsCRP and IL-6 levels in healthy adults by age decade. Both markers rise roughly four to five fold across a normal lifespan, the observation that grounds the inflammaging concept. Values shown are illustrative ranges drawn from published cohort studies including Wyczałkowska-Tomasik et al. 2016 and the CoLaus study, with substantial individual variation around these medians.
NLRP3 sits in the middle of this. It senses cholesterol crystals from oxidized lipids, fragments of mitochondrial DNA released from damaged mitochondria, urate crystals, amyloid aggregates, and the secretory products of senescent cells. All of these accumulate with age, and as they accumulate, NLRP3 activity rises, IL-1 and IL-6 rise, and CRP rises along with them. The pathway evolved to respond to acute danger but in old tissue it fires chronically against a steady stream of internally generated stress signals.
A 2017 paper in Nature Medicine by Dr. David Furman and colleagues at Stanford sharpened the picture in humans. Studying people over 85, they found that expression of inflammasome-related gene modules split the cohort into two groups. People with elevated inflammasome expression had higher blood pressure, more arterial stiffness, and worse all-cause mortality. People with low inflammasome expression were the ones who looked metabolically and cardiovascularly youthful at very old age.
This kind of human longitudinal data is the basis for BioAge’s drug discovery approach. Rather than starting with a disease and looking for a target, they start with biobank samples from large cohorts followed over decades and ask which molecular features predict who ages well. The flagship partnership is with the HUNT biobank in Norway, which contains samples and clinical records from more than 100,000 people followed since 1995, with mortality and morbidity outcomes attached to each participant. Multi-omics analysis of these samples (proteomics, metabolomics, transcriptomics) reveals which pathways differ between people who reach late age in good health and those who develop chronic disease. The inflammasome pathway is among those that fall out of this analysis, and BGE-102 became one of BioAge’s lead programs as a result.
Whether NLRP3 is the master regulator of inflammaging or one of several central nodes is still debated. The case for centrality is that almost every danger signal that accumulates with age, including mitochondrial DNA fragments, oxidized lipids, urate, amyloid, advanced glycation end products, and cell-free DNA from apoptotic cells, converges on this single sensor. Inhibiting it should suppress the inflammatory consequences of all of them at once. The clinical bet BioAge is making is that this leverage point is enough to move outcomes that age-related disease drugs working further downstream have not.
The Alzheimer’s connection
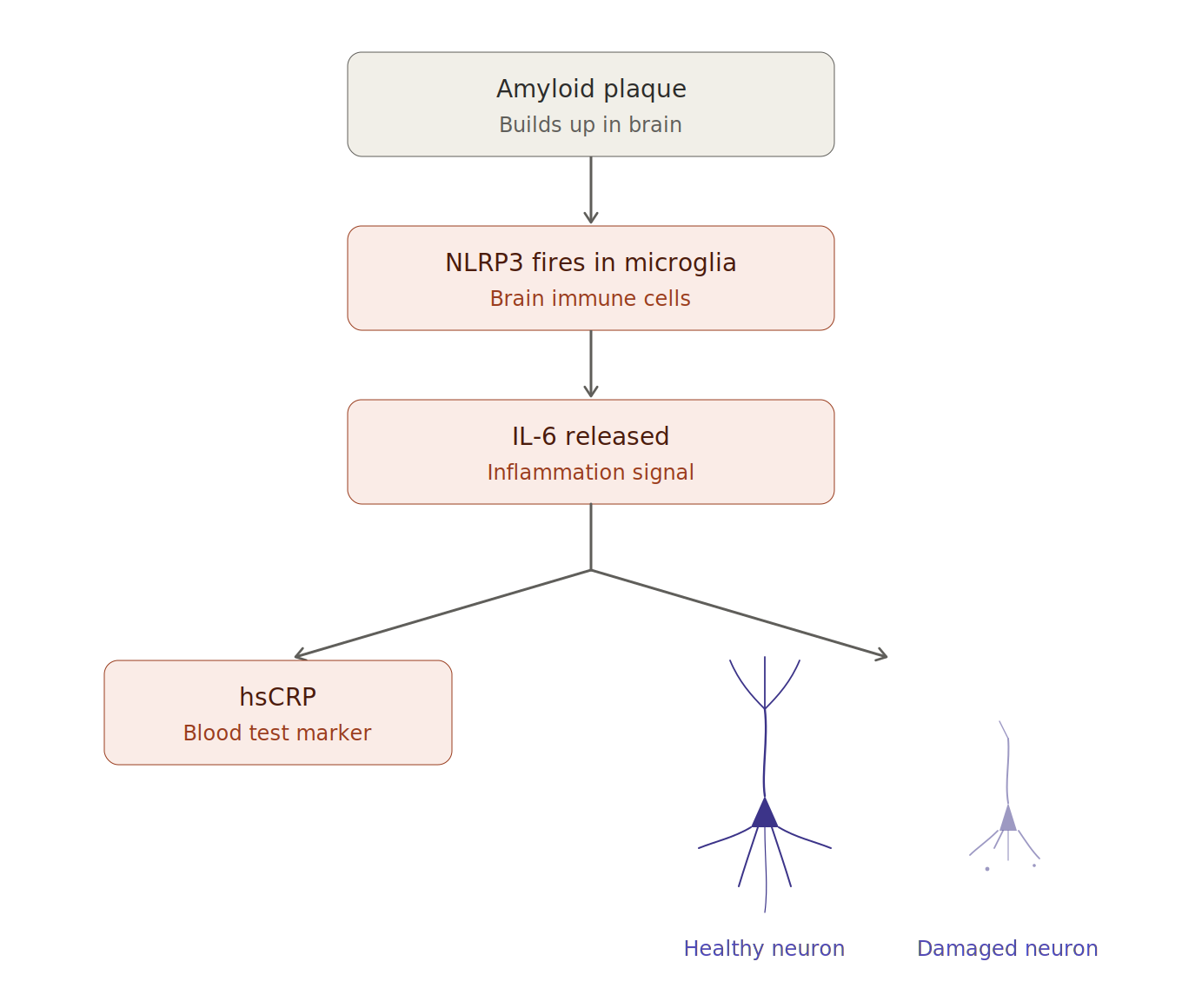
In the brain, the same NLRP3 machinery operates inside microglia, the brain’s resident immune cells. The IL-6 produced reaches the liver and drives hsCRP production while also damaging nearby neurons. Because both readouts share the same upstream cause, hsCRP can correlate with cognitive risk even though it is measured in blood, not in brain tissue.
Amyloid plaques activate NLRP3 inflammasomes inside microglia, which release IL-6. IL-6 then has two downstream effects. It tells the liver to produce hsCRP, the inflammation marker most people see on their blood test, and it damages nearby neurons by disrupting synapses and impairing the receptors they need to communicate.
The link between NLRP3 and Alzheimer’s was demonstrated in a 2013 paper in Nature by Dr. Michael Heneka and colleagues. They took mice carrying the genetic mutations that cause familial Alzheimer’s, then crossed them with mice lacking the NLRP3 gene. The double-mutant mice were largely protected from the memory loss, brain inflammation, and amyloid buildup that affects unmodified counterparts.
A 2017 follow-up paper in Nature by Dr. Carmen Venegas and the Heneka lab showed that the ASC specks released by microglia accelerate the formation of amyloid plaques. The inflammasome both responds to amyloid and actively contributes to building it. Tau pathology, the other hallmark of Alzheimer’s, also activates NLRP3, creating a feed-forward loop where pathological proteins drive inflammasome activation, and inflammasome activation drives more pathological protein accumulation. Sustained exposure to IL-1β and IL-6 in the brain disrupts synaptic plasticity, the cellular process underlying memory formation, and impairs the glutamate receptors neurons need to communicate. This brain-side activity correlates with the systemic blood markers because they share the same upstream machinery, which is why BioAge designed BGE-102 to penetrate the brain rather than work only in the periphery.
Why the APOE4 connection matters
The strongest known genetic risk factor for late-onset Alzheimer’s is the APOE4 variant. Carrying one copy raises lifetime risk roughly threefold. Carrying two copies raises it twelvefold. Roughly a quarter of people of European ancestry carry at least one copy. (As I’ve written about in my own NeuroAge journey, this is a number I have a personal stake in.)
In 2017, Dr. Perry Ridge and Dr. John Kauwe published a study in Genome Medicine using the Utah Population Database, which combines genealogical records with public health information for tens of millions of people. They identified families where APOE4 carriers should statistically have developed Alzheimer’s but had reached old age cognitively healthy. Whole-genome sequencing of these resilient individuals revealed a rare variant in a gene called RAB10 that segregated with protection from disease.
When researchers studied stem cells from people carrying the protective variant, the cells showed reduced inflammation, with the protective effect operating through reduced NLRP3 inflammasome activity. This finding became the founding hypothesis for Halia Therapeutics, a company developing its own NLRP3 pathway drug now in late-stage clinical trials for an unrelated bone marrow disorder.
A substantial portion of APOE4-driven Alzheimer’s risk appears to flow through chronic inflammation, with NLRP3 as a central node. If drugs that suppress this pathway prove safe and effective at scale, they would represent a path to risk reduction that does not depend on the amyloid-clearing antibodies now reaching the clinic, which have shown modest benefit at high cost in recent trials and carry elevated risk for APOE4 homozygotes specifically.
Reading your hsCRP number
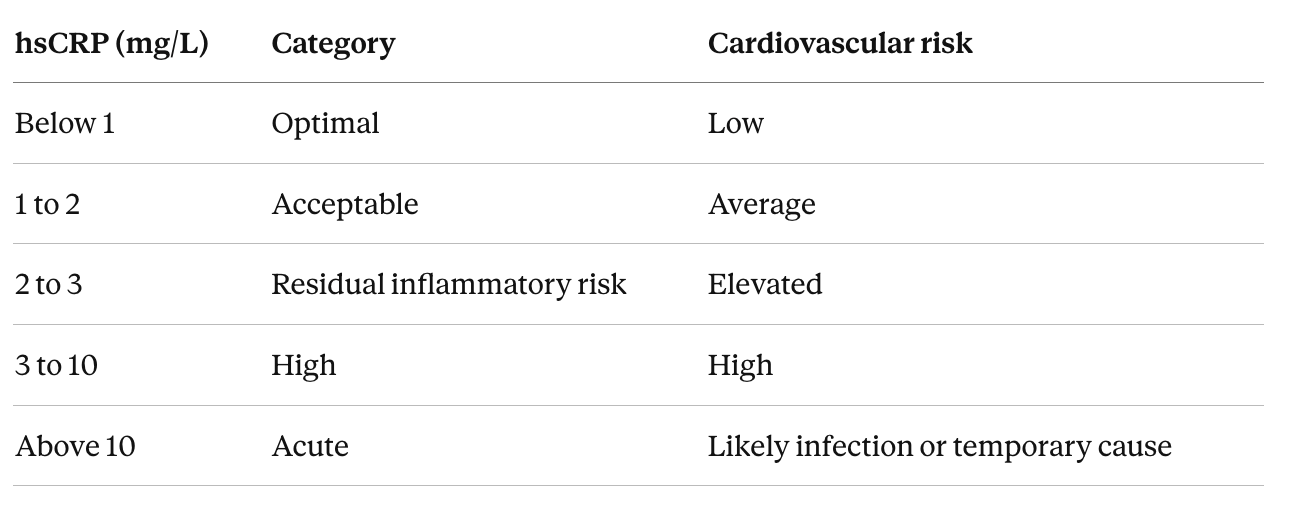
Many people now have hsCRP results from Function Health, Quest, or their primary care doctor without much guidance on interpretation. The American Heart Association categories are reasonably simple.
The 2 mg/L threshold matters because it was the entry criterion for the major inflammation-reduction trials, including the CANTOS trial of canakinumab that first showed reducing inflammation could reduce heart attacks, and the new BioAge and ziltivekimab trials use the same threshold. Values above 10 should be retested in two to three weeks once any acute trigger has passed.
The number can move day to day. Recent infections, vaccinations, intense exercise within the past 48 hours, dental work, and even a sinus infection can push it up. For an accurate baseline, two readings two to four weeks apart, with neither during a period of acute illness, is the standard recommendation.
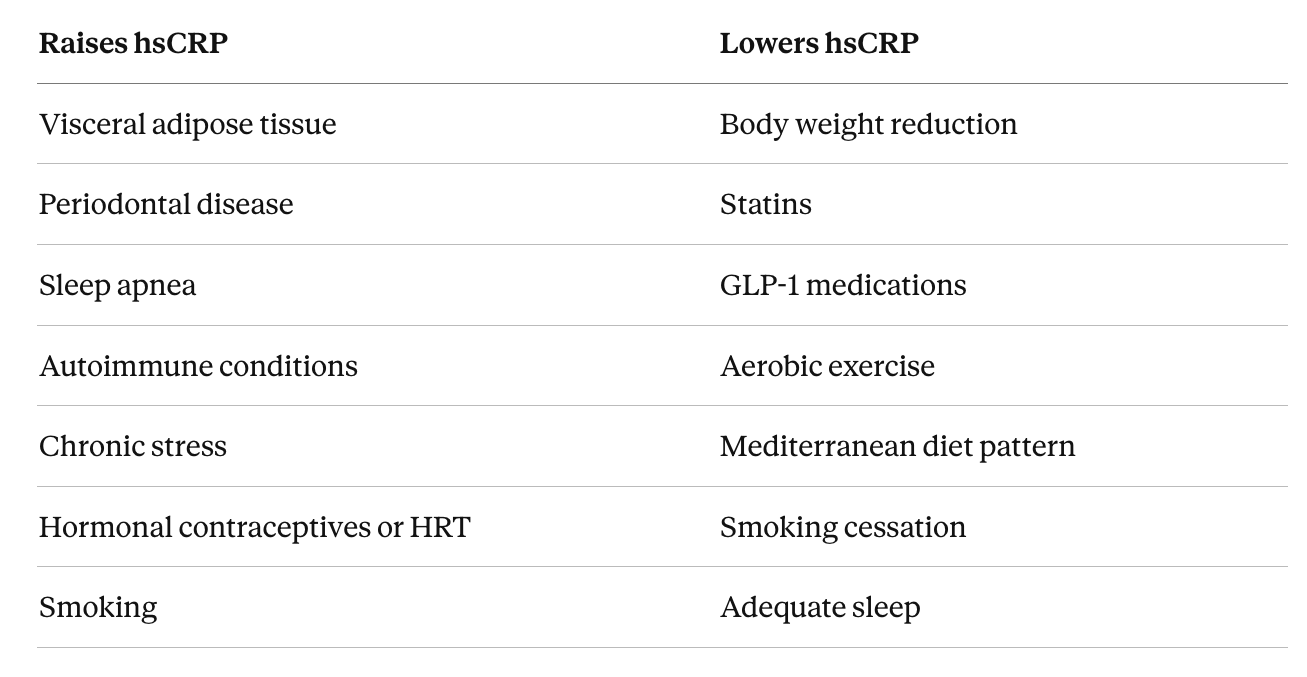
Several factors push chronically elevated hsCRP up or down in otherwise healthy people.
Visceral adipose tissue produces inflammation signals continuously, which is why it is the single largest modifiable contributor for most people.
hsCRP is one of the more actionable markers in a standard panel. A persistently elevated value above 2, in the absence of obvious acute causes, is worth investigating for sources of chronic inflammation. The drugs in development by BioAge and others are aimed at people in roughly this category.
The IL-6 blood test, available through Quest and Labcorp for around 130 dollars cash, measures the cytokine one step upstream of CRP. It is becoming more common in longevity-focused panels because Mendelian randomization studies suggest IL-6 is causally involved in cardiovascular disease while CRP itself is a marker rather than a driver. The two tests give somewhat different information, with discordance between them sometimes pointing toward inflammation sources other than the inflammasome axis. I covered the IL-6 axis in more depth in my piece on IL-6 genetic variants, levels, and meaning, and the connection between vagal tone and inflammatory cytokines in my piece on heart rate variability and longevity, where the MIDUS II cohort showed higher HRV was associated with lower CRP, fibrinogen, and IL-6.
What comes next
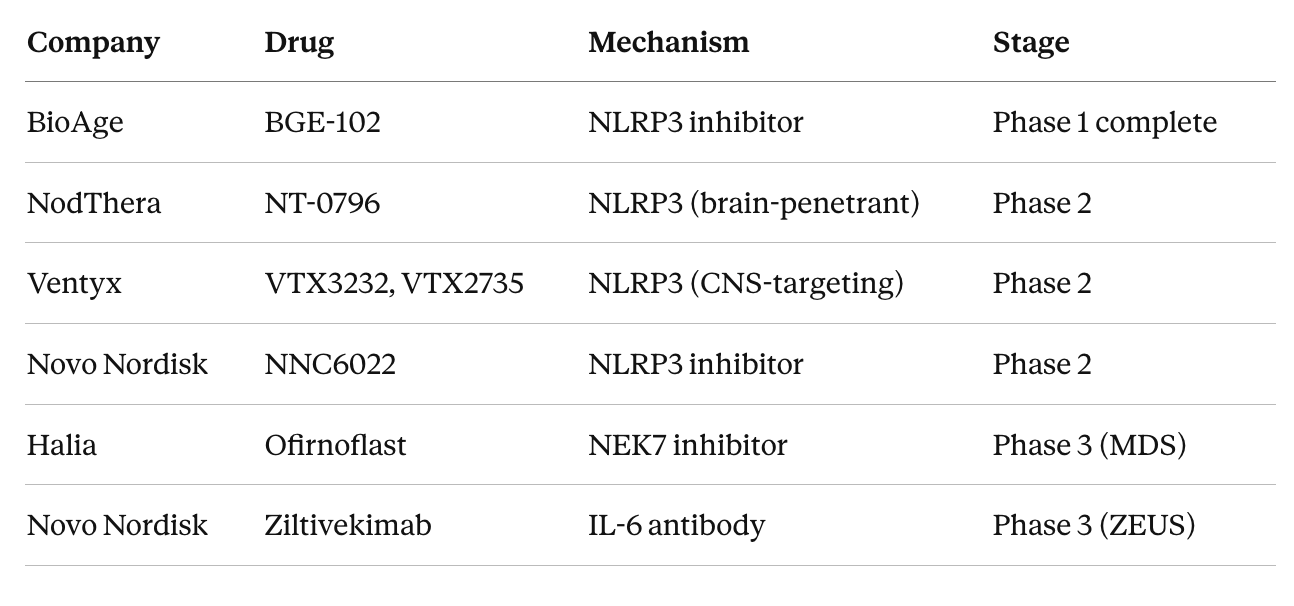
BioAge plans to take BGE-102 into a Phase 2 cardiovascular risk trial, with a separate Phase 1b/2a trial in diabetic macular edema starting mid-2026. More than twenty programs across the industry are pursuing related targets, with several in late-stage trials.
Halia’s ofirnoflast has shown a 72% response rate in a Phase 2 trial for myelodysplastic syndromes and is heading into Phase 3 in early 2026, which puts it furthest along in the clinical pipeline.
The largest readout to watch is the ZEUS trial, which is testing whether the IL-6 antibody ziltivekimab reduces cardiovascular events in over 6,000 patients with chronic kidney disease and elevated inflammation. Topline results are pending, and ZEUS will provide the strongest evidence yet for whether reducing inflammation, independent of cholesterol or blood pressure, prevents heart attacks and strokes.
For people thinking about their own risk, an hsCRP reading above 2 mg/L flags residual inflammatory risk that a standard lipid panel will not catch. APOE4 carriers in particular stand to benefit from inflammation-reducing strategies, whether the lifestyle approaches available now or the pharmaceutical options coming through the pipeline. What was theoretical five years ago is becoming testable in clinical trials, with answers arriving over the next two to three years.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso