The first Alzheimer's blood test is approved by the FDA
Contextualizing the new p-tau 217/ beta-amyloid 1-42 Alzheimer's test and what it and other commercially available tests mean for consumers
This past week the first blood test for Alzheimer’s disease (AD) diagnosis was approved by the US FDA. This landmark breakthrough builds upon 30 years of Alzheimer’s diagnostic testing and opens up a new horizon for early detection and Alzheimer’s prevention. AD diagnostic blood testing is cheaper (about $600 depending on the test) and safer than the alternatives (brain scans with radioactivity or spinal taps), which means it will be accessible to many more people.
Here I discuss the history of AD diagnostics and the newer prognostic tests that are not yet approved but are available for purchase. I also discuss the practical implications of this testing for staying sharp and preventing or reversing Alzheimer’s Disease and other dementias.
Postmortem diagnosis of Alzheimer’s using BRAAK staging (1991)
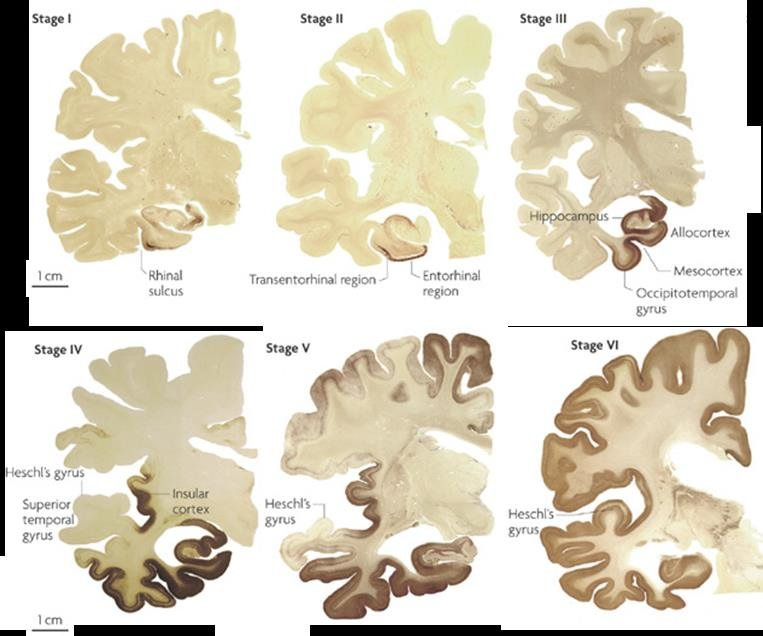
Thirty years ago the only way to test for Alzheimer’s was post-mortem dissection of the brain investigating pathology, known as BRAAK staging. Pathological Tau proteins that accumulate in the brain are depicted in the dark staining of the human brain slices below. The more the Tau “spreads”, the later the BRAAK stage and in general, the more advanced the Alzheimer’s Disease.
PET scanning for Alzheimer’s (2004)
In 2004, a method for using PET (Positron Emission Tomography) scans to identify Alzheimer’s pathology in living people was invented. These scans use radioactive substances injected into the blood stream that bind to pathological proteins that build up in the brain.
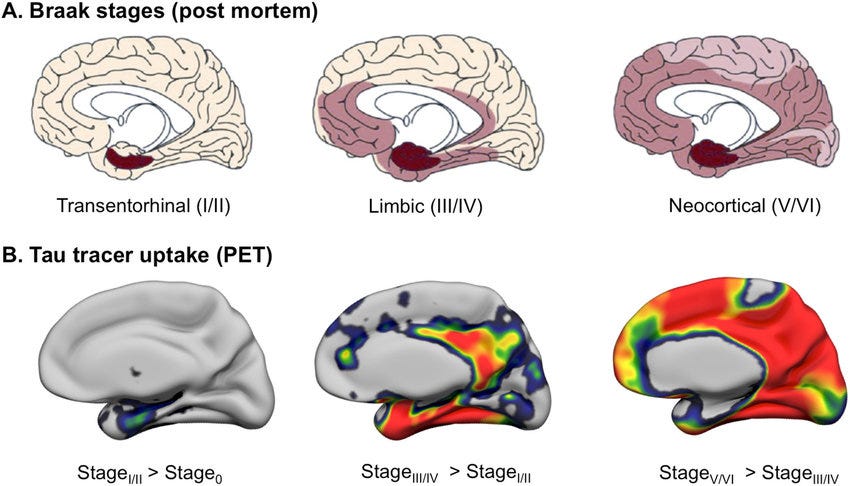
The below figure shows how PET scans for Tau mirror BRAAK staging in postmortem brain. This leap moved us forward from only being able to diagnose Alzheimer’s in people after death, to being able to diagnose it in living people.
We still use PET scans today as the gold standard for Alzheimer’s testing. They have the disadvantages, however, of using radioactivity and therefore posing some risk of cancer if used frequently, being expensive, and only being available in specialized clinics, limiting accessibility.
Blood Testing for Alzheimer’s Diagnosis (2025)
Just this past week, the first blood test for Alzheimer’s, developed by the Japanese biotech, Fujirebio, was approved by the US FDA:
”The U.S. Food and Drug Administration today cleared for marketing the first in vitro diagnostic device that tests blood to aid in diagnosing Alzheimer’s disease. The Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio is for the early detection of amyloid plaques associated with Alzheimer’s disease in adult patients, aged 55 years and older, exhibiting signs and symptoms of the disease.
“Alzheimer’s disease impacts too many people, more than breast cancer and prostate cancer combined,” said FDA Commissioner Martin A. Makary, M.D., M.P.H. “Knowing that 10% of people aged 65 and older have Alzheimer's, and that by 2050 that number is expected to double, I am hopeful that new medical products such as this one will help patients.”…
The Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio measures two proteins, pTau217 and β-amyloid 1-42, found in human plasma, a component of blood, and calculates the numerical ratio of the levels of the two proteins. This ratio is correlated to the presence or absence of amyloid plaques in the patient’s brain, reducing the need for a PET scan…
During review of the Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio, the FDA evaluated data from a multi-center clinical study of 499 individual plasma samples from adults who were cognitively impaired. The samples were tested by the Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio and compared with amyloid PET scan or CSF test results…
In this clinical study, 91.7% of individuals with Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio positive results had the presence of amyloid plaques by PET scan or CSF test result, and 97.3 % of individuals with negative results had a negative amyloid PET scan or CSF test result. Less than 20% of the 499 patients tested received an indeterminate Lumipulse G pTau217/β-Amyloid 1-42 Plasma Ratio result.
These findings indicate that the new blood test can reliably predict the presence or absence of amyloid pathology associated with Alzheimer’s disease at the time of the test in patients who are cognitively impaired.”
Importantly, this test is for Alzheimer's diagnosis, not risk, and people must have cognitive decline as well to be diagnosed. If we screen the general population with this test, there would be many people who would test positive and not have cognitive decline. Those people do not have Alzheimer's and may never have it in the future.
A diagnosis of Alzheimer’s requires that people also score in the Alzheimer’s range on specific cognitive tests, which test for cognitive problems more severe than run of the mill forgetting of keys or not remembering names.
Blood testing for Alzheimer’s prevention in people without cognitive impairment (not yet FDA approved but commercially available)
Tau begins to accumulate 25 years before the onset of the Alzheimer’s Disease symptoms. Amyloid begins to accumulate even earlier.
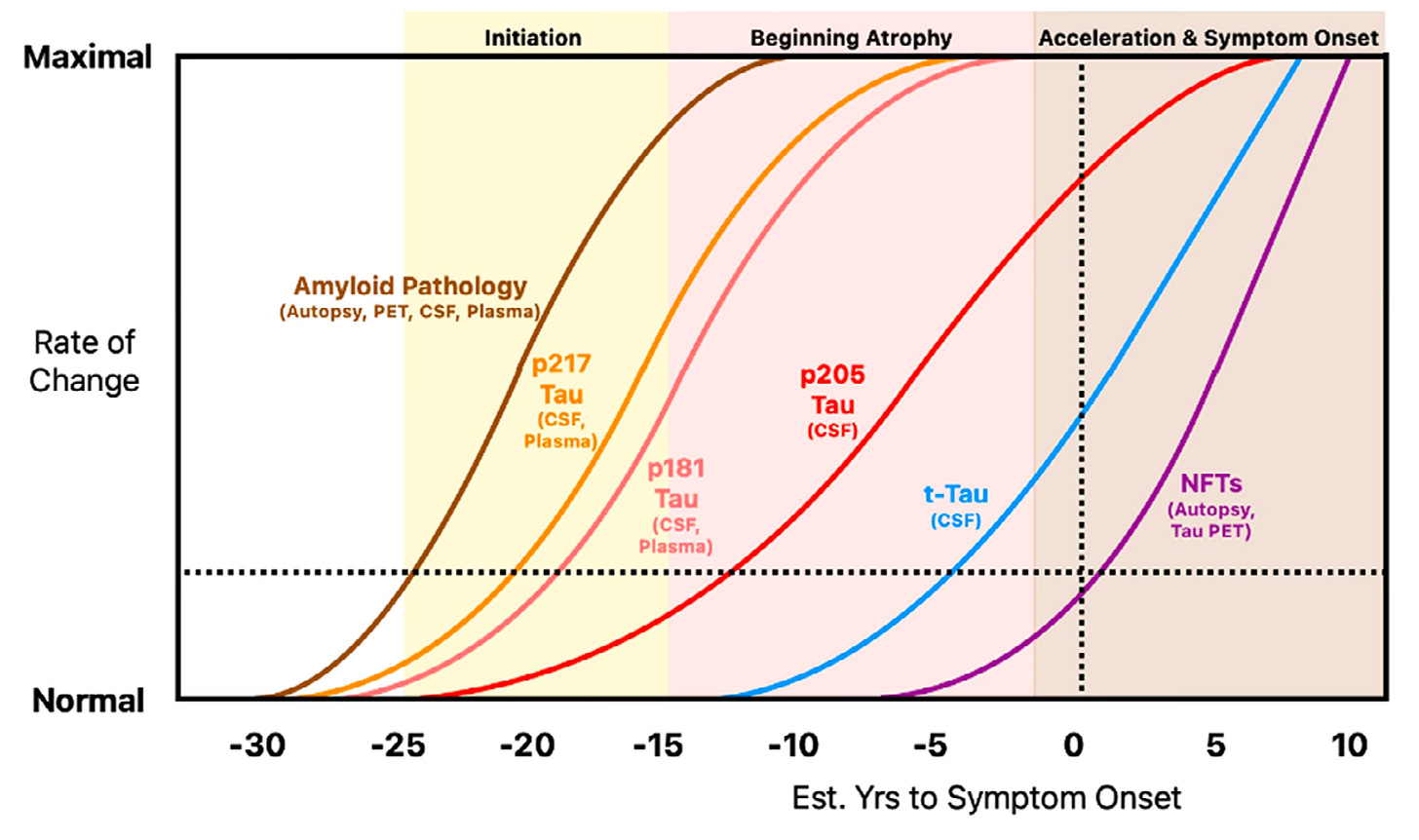
Not everyone who has amyloid or Tau accumulation goes on to develop Alzheimer’s Disease, in the same way that not everyone with high cholesterol goes on to have a heart attack. Tau accumulation along with amyloid accumulation (another pathological protein) are two risk factors, amongst many, for Alzheimer’s.
Some of these risk factors are known, like genetic factors, age, sex, and lifestyle factors. Some of them are still being uncovered, which a goal of NeuroAge. No single risk factor is sufficient to cause disease on its own. Rather, it’s an individual’s particular set of risk factors that determines if and when they will develop Alzheimer’s and also which treatments will work best for them.
Enter the era of personalized Alzheimer’s diagnostics and therapeutics.
In the last 5 years, blood tests have come to market that predict brain pathology pretty well. They are cheaper ($500-$1000) than PET scans and available many places through testing chains like LabCorp, Quest, and Lilly Direct with a doctor’s prescription. I think that many people will be taking these tests in the next 5 years in the same way that most people have yearly cholesterol testing.
In general the Alzheimer’s blood tests are good, but not perfect, at predicting whether there is pathological protein build up in the brain, which is what is important for Alzheimer’s risk. Depending on the test, they have 88-95% percent accuracy of predicting pathology on PET scan. It is good to keep this in mind when choosing a test provider and interpreting your results. I would suggest following up any positive blood test with a PET scan to confirm. If you receive negative results, assume that that this is correct, which is 97% likely for the newly approved Lumipulse G pTau217/ß-Amyloid 1-42 Plasma Ratio, and test again in a year. Two negative tests are better than one to confirm that you are negative for these proteins.
What if you receive a positive test result?
There is emerging evidence, discussed in a Substack post by Dr. Eric Topol, that lifestyle interventions can successfully decrease the build up of p-tau and amyloid in the brain. This also means that you can improve your cognition and decrease your risk of future Alzheimer’s through lifestyle. To learn more about how to do that, read “How to reverse your brain aging”. You can track your progress with blood testing.
Are there any prescription drugs for amyloid?
There are anti-amyloid therapeutics (donanemab, lecanamab) that are FDA approved currently for people with an Alzheimer’s diagnosis. These drugs are administered IV and have severe side effects- brain bleeding and swelling in ~20% of people. They are being tested for AD prevention but are not expected to be FDA approved for this purpose soon, if ever, because of their safety profile. There are pill forms of anti-amyloid and anti-Tau drugs that are in phase 1-3 clinical trials that are expected to be safer. These may end up becoming approved for prevention in the future.
The NeuroAge blood biomarkers test
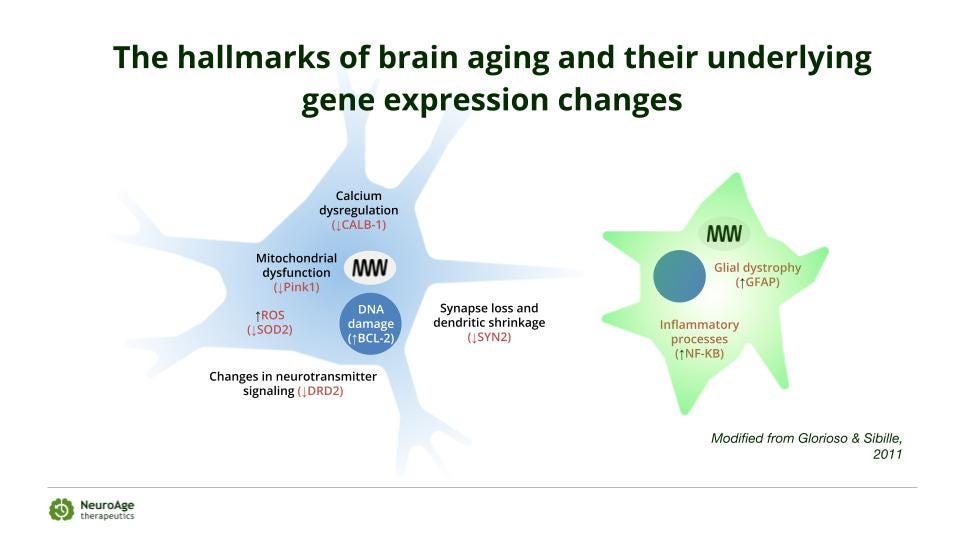
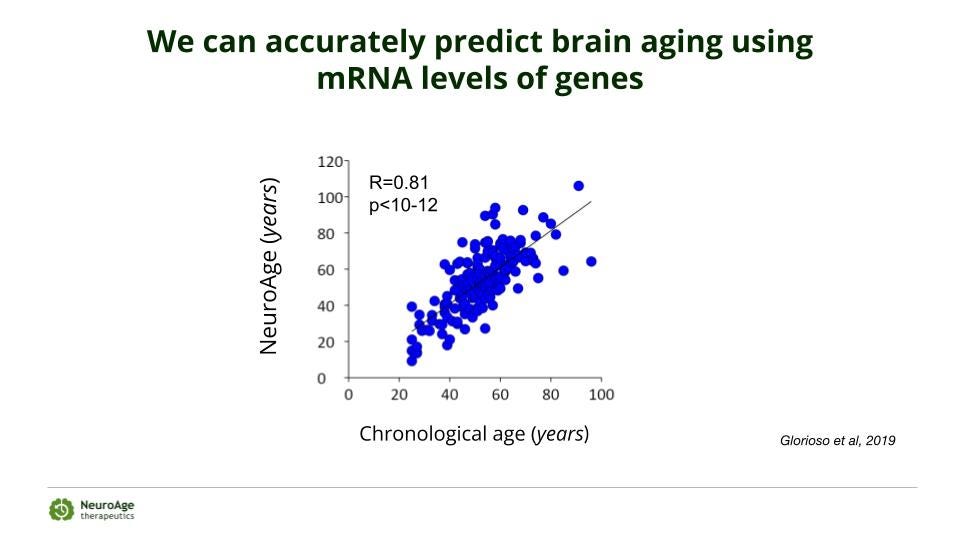
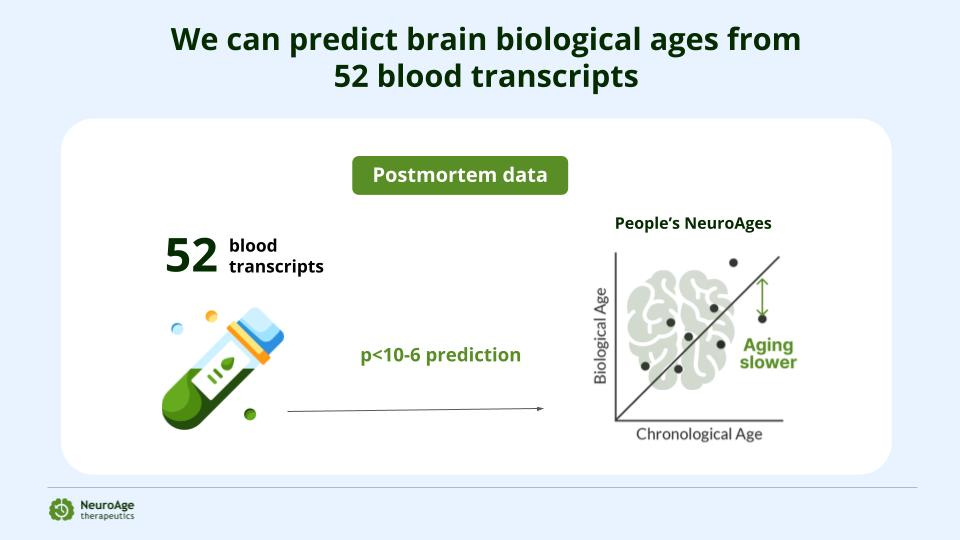
The NeuroAge blood biomarker test is an entirely different and new category of testing available exclusively through NeuroAge. Based on the last 20 years of my research out of MIT, it tests for 52 RNA levels of genes that change with age and predict RNA levels of genes that cause the hallmarks of brain aging. These hallmarks are separate from the build up of amyloid and Tau in the brain and are also risk factors for Alzheimer’s.
NeuroAge is not a specific risk factor for Alzheimer’s Disease but instead tests brain aging, which is also a risk factor for Parkinson’s Disease, other forms of dementia, and normal memory decline in people without any neurological disease diagnosis.
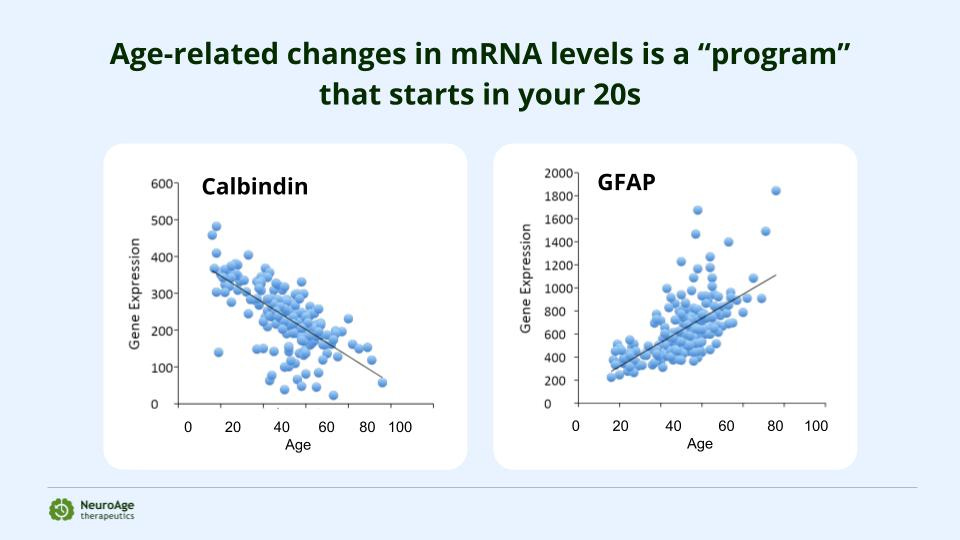
These changes start around age 25, which means we can start testing for them ~40-50 years before the onset of Alzheimer’s Disease.
We can then combine these changes into one score that we call “NeuroAge”.
People with NeuroAges that are five years younger than their chronological age are 6X less likely to have Alzheimer’s, have less pre-Parkinson’s symptoms, and have better memories and cognition overall. About 40% of your brain aging rate is due to lifestyle, which means that people can improve their NeuroAge with improved exercise, sleep, and other interventions.
We can predict levels of about 1000 genes that have RNA levels that change with age in the brain from 52 that change with age in the blood.
The NeuroAge blood biomarkers test is what is known as an aging clock, similar to methylation-based clocks, like those offered by companies like TruAge, but instead of epigenetics, we use RNA levels. Further, it’s an organ-specific aging clock testing the age of your brain specifically rather than your whole body. The advantage of an RNA-based clock instead of a methylation-based clock is two-fold 1. it’s more accurate as the methylation-based clock are notoriously bad at predicting diseases of the brain and 2. it is less of a black box. If you know which gene and which direction of change in RNA levels of that gene is protective for brain aging, then it’s possible to identify new drug targets. Epigenetic marks have an unclear association with genes or directions of changes.
Many of the gene RNA levels that we track are new drug targets that we or others are developing to create better therapeutics for neuro disorders. Alzheimer’s and other neuro disorders have a largely unexplored landscape of therapeutics. Clinical trials have been so expensive to run for Alzheimer’s and the field has been so myopically focused on amyloid and Tau that many newer drugs have yet to be tested. These new therapeutics have the potential to be much better than the current drugs in terms of efficacy, ease of use, and safety and can be used in conjunction with amyloid and Tau drugs.
Our long-term vision is not only to help people track their brain aging and reverse it with personalized lifestyle interventions, but ultimately to provide our users with new personalized therapeutics custom to their biomarkers.
It is a very exciting time for brain health. We are on the precipice of having all the tools that we need to stay sharp for our lifetimes.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso