The Bredesen Clinical Trial for Alzheimer's Treatment (New Study)
A critical read of the paper and the evidence for the ReCODE protocol
Dr. Dale Bredesen is the author of the 2017 NY Times best-selling book, “The End of Alzheimer’s”, former President of the Buck Institute for Research on Aging, and Founder and CSO of Apollo Health, which has a lifestyle-based Alzheimer’s treatment program called ReCODE.
For nearly a decade, Bredesen’s approach to Alzheimer’s disease has generated both excitement and controversy. Beginning with his 2014 case report describing cognitive improvement in 9 of 10 patients using a multi-pronged protocol he called “MEND” (Metabolic Enhancement for Neurodegeneration), and followed by additional case series in 2016 and 2018, Bredesen’s work attracted significant attention from patients desperate for alternatives to failed pharmaceutical trials.
However, case studies and uncontrolled observations cannot establish causality. Critics reasonably demanded randomized controlled trials. Patients might have improved due to placebo effects, regression to the mean, intensive attention from a medical team, or natural disease fluctuation rather than the specific interventions. The scientific community waited for rigorous testing.
The December 2025 preprint discussed here is the first randomized controlled trial of the Bredesen approach. While still awaiting peer review, it provides the controlled comparison data needed to move beyond anecdote. The results merit serious examination, both for what they show and for what remains uncertain. Regardless of where one stands on precision medicine for neurodegeneration, this trial advances the conversation beyond “N of 1” reports into territory where evidence-based evaluation becomes possible.
Touted as being more effective than Lecanamab (a recently approved Alzheimer’s medication), the trial showed large improvements in some aspects of cognition (notably not on Alzheimer’s cognitive tests), but no improvement in brain volume, most aging clocks tested, or Alzheimer’s blood biomarkers. The trial’s limitations, particularly the lack of blinding and the inability to attribute effects to specific components, mean it cannot definitively answer whether this approach works or why. It also did not test Bredesen’s full supplement stack leaving open questions about its efficacy. It is, nevertheless, a significant step forward.
Treatment Versus Prevention: A Critical Distinction
Treatment refers to interventions applied after cognitive symptoms have manifested (MCI or dementia stage), while prevention refers to interventions applied before clinical symptoms appear. This distinction matters because interventions that fail in treatment trials may still be effective for prevention. By the time of clinical diagnosis, patients may have lost 20-40% of neurons in affected regions, and lifestyle interventions can support living neurons but may not be able to regenerate dead neurons.
Several interventions reviewed in this document show this pattern with benefit in cognitively normal or at-risk populations but null or even harmful effects in established dementia. The Bredesen trial is a treatment trial, enrolling patients who already have MCI or early dementia. The evidence reviewed throughout this document should be interpreted through this lens.
The Bredesen Precision Medicine Trial
Overview of the Study
In December 2025, Dale Bredesen and colleagues published a preprint describing a 9-month randomized controlled trial comparing a personalized precision medicine protocol to standard of care in 73 patients with MCI or early dementia. Posted on Preprints.org and not yet peer-reviewed, it represents the first RCT of this approach.
The trial randomized 50 patients to the precision medicine arm (the Intervention Group) and 23 to standard of care (the Control Group). Participants were aged 45 to 76 with Montreal Cognitive Assessment (MoCA) scores of 18 or higher. The 2:1 randomization was based on effect sizes observed in the earlier proof-of-concept trial.
The Intervention
The precision medicine protocol combined multiple interventions delivered by a team including a physician, health coach, nutritionist, and physical trainer:
Diet: A plant-rich, high-fiber, mildly ketogenic diet with 12 to 16 hour overnight fasting, emphasizing leafy greens, unsaturated fats, wild-caught low-mercury fish, and avoidance of processed foods, simple carbohydrates, gluten, dairy, and alcohol. Blood ketone targets of 1.0 to 4.0 mM beta-hydroxybutyrate were monitored.
Exercise: At least 45 minutes of aerobic exercise six days per week, strength training twice weekly, and high-intensity interval training at least twice weekly.
Sleep: Optimization targeting 7 to 8 hours nightly, tracked by Oura ring, with CPAP or dental appliance treatment for identified sleep apnea or upper airway resistance syndrome.
Stress Management: HeartMath Inner Balance HRV biofeedback for at least 10 minutes daily.
Brain Training: BrainHQ cognitive training for at least 15 minutes daily.
Hormones and Supplements: Bioidentical hormone replacement (required for participation), thyroid optimization, and supplements to correct identified deficiencies in vitamin D, omega-3s, B vitamins, CoQ10, and minerals.
Targeted Treatments: Based on individual testing, patients received treatment for identified chronic infections, detoxification protocols for identified toxins, gut healing protocols for identified dysbiosis, and anti-inflammatory interventions including low-dose naltrexone for autoimmunity.
Adjunctive Therapies: Most sites included photobiomodulation, and three sites added hyperbaric oxygen, exercise with oxygen therapy, cranio-electrical stimulation, or neurofeedback.
Reported Results
The authors report statistically significant improvements in the precision medicine group compared to controls:
Cognitive Measures:
Neurocognitive Index (CNS Vital Signs): Improved from 92.0 to 106.2 in the Intervention Group versus declined from 96.9 to 92.4 in the Control Group (p<0.001)
Composite Memory: Improved 13.12 points in the Intervention Group versus declined 5.39 points in the Control Group (p<0.001)
Executive Function: Improved 16.86 points versus 1.82 points (p=0.001)
BrainHQ Composite: Improved 25.1% versus 10.4 points (p=0.011)
Clinical Measures:
AQ-Change (informant-rated): Improved 8.74 points in the Intervention Group versus declined 2.36 points in the Control Group (p<0.001). AQ (Alzheimer’s Questionare) is a 21-item dementia assessment filled out by a family member or caregiver rather than the patient and is essentially measuring whether the they perceive that the patient’s everyday cognitive functioning has improved or worsened over the trial period. It’s worth noting that informant-rated measures can be susceptible to expectancy effects in open-label trials. If the family member knows their loved one is receiving an intensive intervention, they may perceive improvements that aren’t reflected in objective testing.
MoCA (a standard Alzheimer’s cognitive test) improved in both groups (3.79 versus 2.57 points), but the between-group difference was not statistically significant (p=0.154).
Metabolic Parameters: Significant improvements in BMI, blood pressure, HbA1c, HOMA-IR, homocysteine, and triglycerides.
Epigenetic Aging Clocks: The trial reported results from multiple epigenetic aging clocks, but the presentation raises concerns about selective reporting. The authors highlighted OMICmAge, a newer multi-omics clock, which showed a decrease of approximately 1.3 years in the Intervention Group (p=0.016). However, several other established clocks were also measured:
GrimAge (widely considered the best predictor of mortality and healthspan): Results not reported
PhenoAge (trained on mortality-related biomarkers): Results not reported
Horvath clock (the original pan-tissue clock): Results not reported
DunedinPACE (measures pace of aging): Results not reported
This is methodologically concerning. A 2025 comparison of 14 clocks in 18,859 individuals confirmed that second-generation clocks (GrimAge, PhenoAge) significantly outperform first-generation clocks for predicting disease outcomes. GrimAge specifically has been shown to predict 8 of 9 age-related clinical outcomes including walking speed, frailty, polypharmacy, and mortality, while Horvath and Hannum clocks were “not predictive of health.”
The fact that the trial chose to emphasize OMICmAge rather than the more established and validated GrimAge suggests the possibility that GrimAge did not show significant improvement. Without transparent reporting of all clock results, the epigenetic findings cannot be properly evaluated.
MRI Volumetrics: No significant between-group differences in brain volume changes.
Alzheimer’s Blood Biomarkers: P-tau 217 declined significantly within the Intervention Group (p=0.028), but between-group differences were not significant. No significant changes in amyloid-beta 42/40 ratio, GFAP, or NfL.
Critical Evaluation
Strengths
This represents an important advance beyond case series, providing controlled comparison data. The trial employed multiple outcome measures, blinded cognitive assessments for MoCA, biomarker confirmation of AD pathophysiology (68 of 70 participants had abnormal plasma amyloid-beta 42/40 ratios), and showed large effect sizes with a favorable safety profile.
Limitations
Open-label design: Subjects, physicians, and health coaches all knew group assignment. Only neuropsychological raters were blinded. This makes it difficult to separate true treatment effects from placebo and expectancy effects.
Control group design: The standard of care group received observation rather than an active intervention, making this a comparison of intensive multimodal intervention versus minimal contact rather than precision medicine versus standard dementia care.
MoCA did not differentiate groups: The most widely-used clinical cognitive measure showed no significant between-group difference.
No structural brain improvements: MRI volumetrics showed no significant differences, and both groups showed continued gray matter loss.
Mixed biomarker results: Between-group differences in tau and other biomarkers were not significant.
Incomplete epigenetic clock reporting: Multiple epigenetic clocks were measured, but only OMICmAge results were reported. Results from GrimAge, the clock most validated for predicting mortality and healthspan, were not included. This makes it difficult to assess the consistency of epigenetic findings across different measures.
Attribution difficulties: With 15 to 20 simultaneous interventions, it is difficult to determine which components drove any observed effects.
The supplement stack was not actually tested: The ReCODE protocol includes an extensive supplement regimen with over 20 compounds including curcumin, ashwagandha, bacopa monnieri, gotu kola, magnesium threonate, ubiquinol (CoQ10), PQQ, resveratrol, nicotinamide riboside, omega-3s, choline, liposomal glutathione, probiotics, vitamin D3+K2, B vitamins, alpha-lipoic acid, NAC, zinc, huperzine A, lion’s mane mushroom, rhodiola, and ALCAR, among others. Participants took these supplements as part of the bundled intervention.
Because everything was combined including diet, exercise, sleep optimization, stress management, brain training, hormone replacement, AND all these supplements, we cannot attribute any effect to the supplement stack specifically. If the trial showed benefit, it might be entirely from exercise and diet with zero contribution from supplements. Or one supplement might be responsible while the others are inert. Or some might even be harmful, with their negative effects masked by benefits from other components.
Many of these supplements have minimal or no RCT evidence for cognitive benefit in humans.
Some supplements do have evidence (omega-3s for MCI, B vitamins for elevated homocysteine), but these were bundled with many unproven compounds. Because everything was combined, positive outcomes cannot be attributed to any individual supplement, yet these results may be cited to support the full protocol.
A more informative design would have tested: (1) lifestyle interventions alone, (2) lifestyle + evidence-based supplements only (omega-3, B vitamins if indicated), and (3) lifestyle + full supplement stack. Without such comparisons, we cannot determine whether any supplements contributed to outcomes or whether patients could achieve similar results with fewer interventions.
Participants were metabolically healthy at baseline: Examining baseline values, HbA1c was 5.5% (normal), fasting glucose 96 mg/dL (normal), HOMA-IR 1.4 (normal), triglycerides 77 mg/dL (optimal). This suggests the cognitive improvements were not driven by correcting metabolic dysfunction, since participants were not metabolically dysfunctional to begin with.
Bottom Line
This trial provides the most rigorous evidence to date for the Bredesen approach, showing large cognitive improvements compared to standard care. However, the open-label design, negative MRI results, largely negative biomarker findings, and inability to attribute effects to specific interventions leave important questions unanswered. The cognitive improvements could reflect true disease modification, intensive support and engagement effects, or some combination. Larger, blinded trials with active comparators would help clarify these questions.
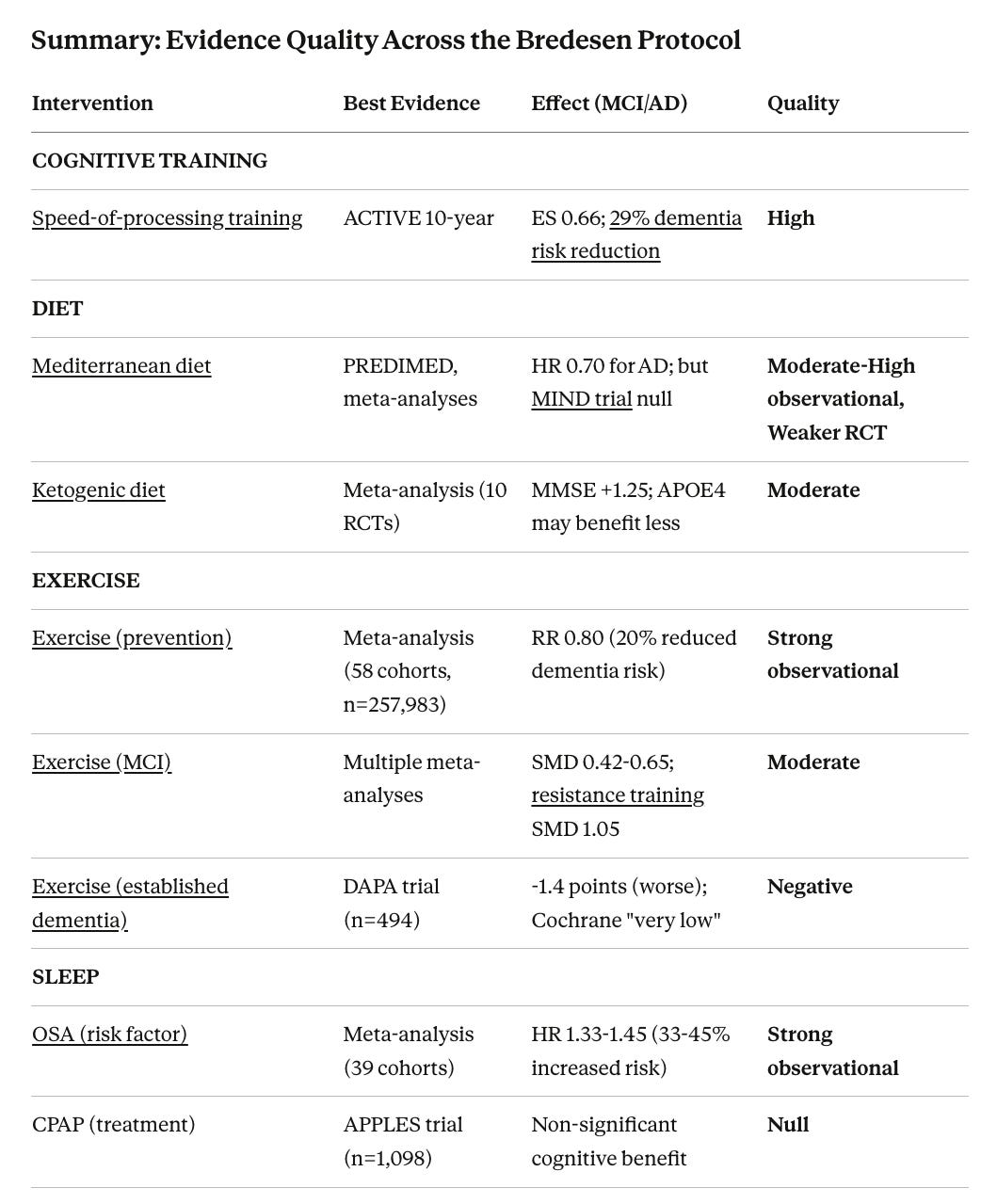
Evidence for Protocol Components
Below I briefly review the existing evidence for the Alzheimer’s treatment protocol utilized in the trial.
Dietary Interventions
Mediterranean Diet
The Mediterranean diet has the most robust dietary evidence for brain health. A 2024 meta-analysis of 23 studies found hazard ratios of 0.70 for Alzheimer’s disease (30% risk reduction) and 0.89 for all-cause dementia. The PREDIMED trial showed effect sizes of 0.39 to 1.29 for cognitive composites.
However, intervention evidence is less consistent. The 2023 MIND diet trial (n=604) found no significant cognitive difference between groups. The Australian MedLey study was also negative. These results raise questions about whether observational benefits translate to intervention effects.
Evidence Pattern: Strong observational evidence for prevention, weaker intervention evidence for treatment.
Ketogenic Diet
A 2024 meta-analysis of 10 RCTs (n=691 AD patients) found MMSE improvement of 1.25 points and ADAS-Cog improvement of 3.43 points. The BENEFIC trial demonstrated improvements across multiple cognitive domains in MCI patients using a ketogenic drink. APOE-epsilon-4 carriers may derive less benefit and some may even have an increase in inflammatory and cardiovascular risk markers. Read “Keto or Ket-no for brain health?”
Quality Rating: Moderate for MCI, emerging for AD
Exercise
Exercise evidence reveals critical disease-stage dependency. The distinction between MCI and established dementia populations is essential.
For Prevention (strong observational evidence):
A 2022 meta-analysis of 58 prospective cohort studies (n=257,983) found physical activity associated with 20% lower risk of all-cause dementia (RR 0.80, 95% CI 0.77-0.84), 14% lower risk of Alzheimer’s disease (RR 0.86), and 21% lower risk of vascular dementia (RR 0.79). These associations persisted even with follow-ups of 20 years or longer. Earlier meta-analyses found relative risks of 0.72 for dementia in those with midlife exercise.
A 2025 Nature Medicine study using pedometer-measured step counts in cognitively unimpaired older adults with elevated amyloid found higher physical activity was associated with slower tau accumulation and cognitive decline. Dose-response analysis showed benefits plateauing at 5,000-7,500 steps per day.
In MCI populations: Meta-analyses consistently show small-to-moderate effects on global cognition (SMD 0.42 to 0.65 for aerobic exercise). Network meta-analyses find multi-component exercise shows the strongest effects (SMD 0.99 to 1.09).
In established dementia: The DAPA trial (n=494), the largest RCT in established dementia, found that moderate-to-high intensity exercise showed slightly worse cognitive outcomes than usual care (ADAS-cog 25.2 vs 23.8 at 12 months, p=0.03). The exercise program improved physical fitness but showed “no noticeable improvements in other clinical outcomes.” The investigators concluded: “Future work should concentrate on approaches other than exercise to influence cognitive impairment in dementia.”
The Cochrane Review rated evidence quality as “very low” with non-significant effects on cognition. The EXERT trial in amnestic MCI similarly showed no 12-month benefit.
Resistance training may be particularly effective in MCI, with network meta-analysis showing SMD 1.05 for global cognition.
Evidence Pattern: Moderate benefit in MCI, null to negative results in established dementia. This is a critical finding: exercise that appears protective in earlier disease stages does not help and may possibly worsen outcomes in established dementia.
Sleep
CPAP for Sleep Apnea
Prevention Context (strong observational evidence):
Obstructive sleep apnea is increasingly recognized as a modifiable dementia risk factor. A 2022 systematic review and meta-analysis found sleep apnea associated with 43% increased risk of any neurocognitive disorder (HR 1.43), 28% increased AD risk (HR 1.28), and 54% increased Parkinson’s disease risk (HR 1.54). A 2025 meta-analysis of 39 cohort studies confirmed OSA increases all-cause dementia risk by 33% (HR 1.33) and AD risk by 45% (HR 1.45).
A UK Biobank study found individuals with OSA had over 4-fold higher risk of all-cause dementia (HR 4.24) and nearly 6-fold higher AD risk (HR 5.67), with earlier OSA diagnosis (before age 52) conferring even higher risk. Observational data suggest CPAP adherence is associated with lower odds of incident dementia diagnoses (OR 0.78 for AD, OR 0.69 for non-specified dementia).
Treatment Evidence (null):
The APPLES trial (n=1,098), the largest sham-controlled CPAP study, found no significant cognitive benefit on primary outcomes at 6 months. Meta-analyses show CPAP improves only executive function in severe OSA (SMD 0.93 for Trail Making Test-B), with no consistent effects on other domains.
An American Thoracic Society workshop report noted that observational studies suggest CPAP may delay MCI by up to 10 years, but this finding was based on only 35 patients. The report concluded that “the impact of CPAP in improving cognitive function and preventing dementia is unclear” and called for large RCTs.
Evidence Pattern: Strong observational association for prevention (33-45% increased dementia risk with OSA), but no RCT-demonstrated cognitive benefit from treatment. The disconnect may reflect the need for earlier or longer-duration treatment than studied in trials.
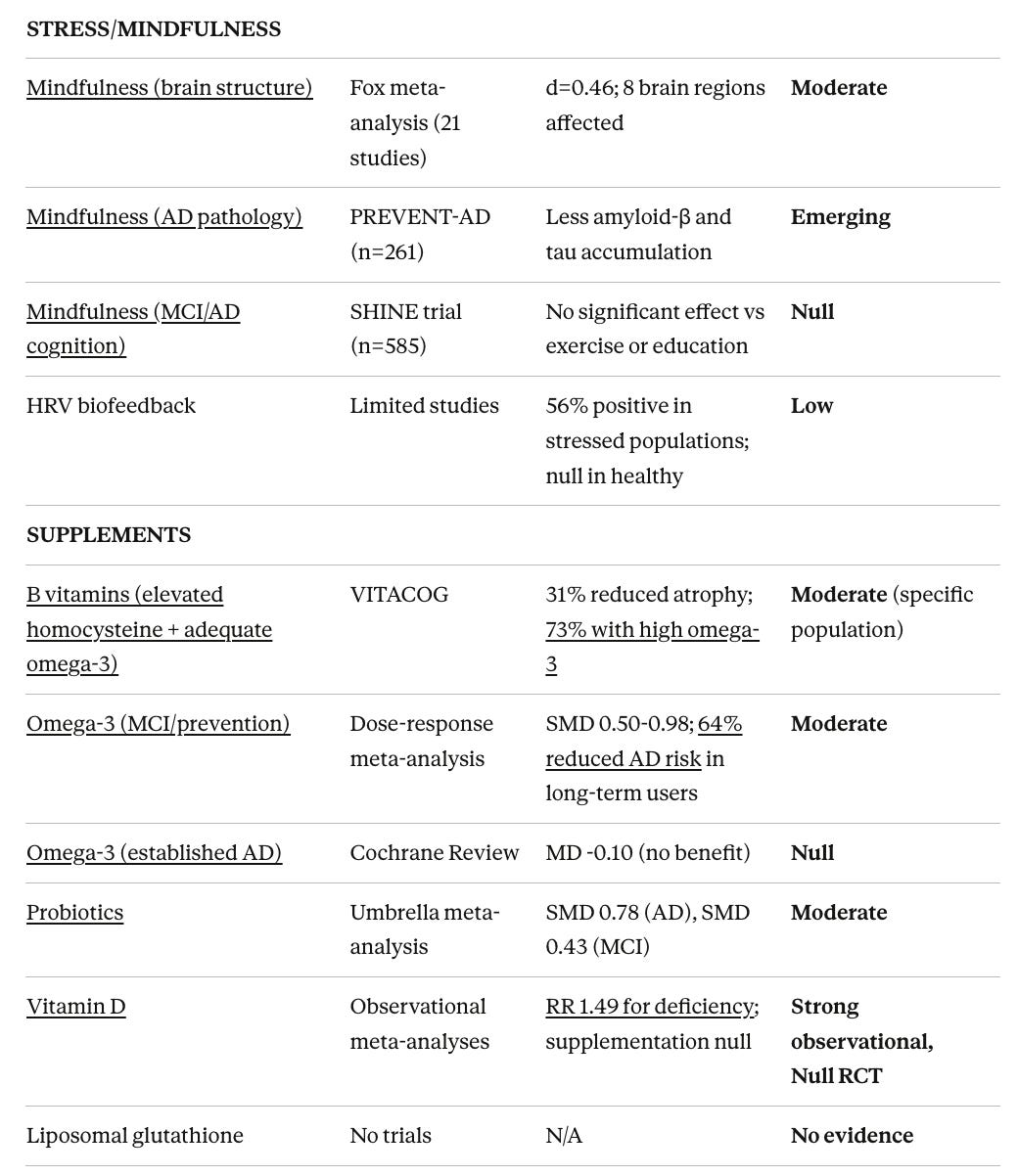
Stress Management
Mindfulness and Meditation
The evidence for mindfulness requires careful parsing by outcome type, as the pattern differs substantially between brain structure changes, cognitive outcomes in healthy populations, and cognitive outcomes in MCI/dementia.
Prevention Context (emerging evidence):
A 2022 PREVENT-AD study in 261 nondemented older adults with family history of AD found trait mindfulness was associated with less cognitive decline and less amyloid-β and tau accumulation in the brain. This suggests mindfulness may influence core AD pathology, not just symptoms.
Long-term meditators have been shown to be physiologically 12 years younger than chronological age in some measures. Studies show expert meditators have enhanced brain integrity in regions vulnerable to AD. The European Medit-Ageing project is conducting large-scale RCTs to test whether meditation training can reduce dementia risk factors in aging populations.
Brain Structure Changes (strong evidence):
The Fox et al. 2014 meta-analysis of 21 morphometric neuroimaging studies (~300 practitioners) found consistent structural differences across 8 brain regions, with a moderate global effect size (Cohen’s d = 0.46). Regions showing changes included prefrontal cortex, anterior cingulate cortex, insula, hippocampus, and somatosensory areas. Both gray matter volume and white matter integrity show differences in meditators.
Studies on white matter using diffusion tensor imaging (DTI) have shown improvements. Tang and colleagues demonstrated white matter changes after just 2-4 weeks of training. Longitudinal studies suggest meditation may slow age-related white matter decline.
Cognitive Outcomes in General/Healthy Populations (small positive effects):
Meta-analyses show small but significant effects: overall g=0.15 to 0.36. Working memory shows the most consistent benefit (g=0.23 to 0.42). Effects are modest but real in cognitively normal individuals.
Cognitive Outcomes in MCI/Dementia (null effects):
Here the evidence weakens substantially. The SHINE trial (n=585, 18 months) found no significant cognitive differences between MBSR, exercise, and health education. A meta-analysis specifically in MCI and dementia populations found no significant effects on depression, anxiety, quality of life, memory, or overall cognitive function.
Interpretation:
The pattern suggests mindfulness produces measurable brain changes in healthy populations that may represent cognitive reserve or neuroprotection. However, once cognitive impairment is established, these mechanisms do not translate to measurable cognitive improvement. This is consistent with the broader pattern of prevention versus treatment effects seen across interventions.
The intervention may be valuable for stress reduction and quality of life without producing cognitive disease modification in symptomatic patients. For individuals without cognitive symptoms, the brain structure evidence suggests potential protective value.
Quality Rating: Moderate for brain structure (healthy populations), Small positive for general cognitive effects, Null for MCI/dementia cognitive outcomes
HRV Biofeedback
A systematic review found 56% of studies reported positive effects on executive functions, but benefits were concentrated in stressed or vulnerable populations. A 5-week RCT in healthy adults found no significant improvement in inhibitory control, working memory, or processing speed.
HeartMath specifically has limited independent research. Most cognitive studies come from the HeartMath Institute itself. No trials exist in MCI or dementia populations.
Quality Rating: Low (limited independent evidence, no MCI/AD data)
Brain Training
The ACTIVE trial (n=2,832) provides the most compelling cognitive training evidence. At 10-year follow-up:
Speed-of-processing training showed large effect size (ES 0.66) with durable benefits
Reasoning training showed moderate effects (ES 0.23)
Memory training showed no durable effects
Most importantly, speed training showed 29% reduction in dementia risk (HR 0.71, p=0.049). Each additional training session was associated with 10% lower hazard for dementia. This is one of the only interventions with RCT evidence for dementia risk reduction.
BrainHQ builds on ACTIVE’s Useful Field of View paradigm. A comprehensive meta-analysis of computerized cognitive training (52 RCTs, n=4,885) found overall effect g=0.22 (small but significant).
Quality Rating: High for speed-of-processing training specifically (unique dementia risk reduction evidence), Lower for general cognitive training
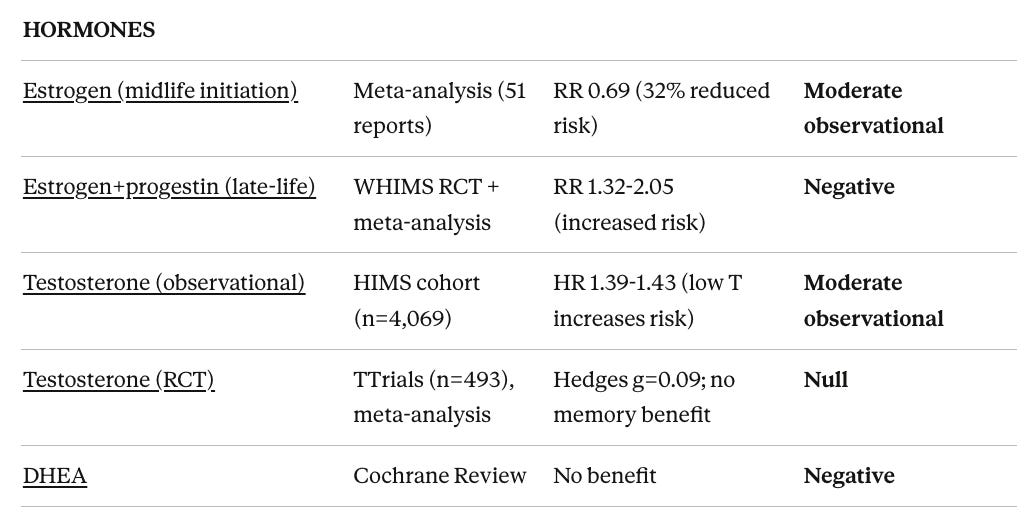
Hormone Replacement
Estrogen/HRT
A 2023 systematic review and meta-analysis provides the most comprehensive analysis of HT effects on dementia risk, pooling 51 reports (6 RCTs, 45 observational studies) including over 768,000 dementia cases and 5.5 million controls.
RCT Evidence (late-life initiation, ages 65+):
The Women’s Health Initiative Memory Study (WHIMS, n=7,479) found combined hormone therapy (EPT) doubled dementia risk (HR 2.05) in women aged 65 to 79, while estrogen-only therapy (ET) showed non-significant 49% increased risk. Meta-analysis of WHIMS reports shows overall HT increased dementia risk by 38% (RR 1.38), driven by EPT (RR 1.64, p=0.002) rather than ET (RR 1.19, p=0.18).
Observational Evidence (mixed timing):
Pooled observational data showed HT associated with 22% reduced AD risk (RR 0.78, p=0.013) and 19% reduced all-cause dementia risk (RR 0.81, p=0.007). By formulation:
Estrogen-only (ET): 14.5% reduced dementia risk (RR 0.855, p=0.002)
Estrogen-plus-progestogen (EPT): Non-significant 9% reduction (RR 0.91, p=0.251)
The Timing Hypothesis (critical distinction):
The meta-analysis stratified by initiation timing reveals the prevention vs treatment pattern:
Midlife initiation:
Overall HT: 13% reduced dementia risk (RR 0.87, p=0.002)
ET specifically: 32% reduced risk (RR 0.685, p=0.010)
EPT: 23% reduced risk (not significant, RR 0.775, p=0.309)
Late-life initiation:
Overall HT: 7.5% increased dementia risk (RR 1.075, p<0.001)
ET: 7% increased risk (not significant)
EPT: 32% increased risk (not significant, RR 1.32)
Duration effects: Long-term use (>5 years) showed 18% reduced risk (RR 0.82, p<0.001), while short-term use showed non-significant effects.
A 2025 Lancet Healthy Longevity review of over 1 million participants found no significant overall association, but confirmed timing-dependent effects consistent with the meta-analysis above.
RCTs testing the timing hypothesis (KEEPS-Cog, ELITE) showed no cognitive benefit in either early or late postmenopause groups, though these were not powered for dementia outcomes.
Summary: The evidence supports a critical window for therapeutic benefit. Midlife estrogen-only therapy shows the strongest protective association (32% risk reduction), while late-life combined therapy shows increased risk. This aligns with the “healthy cell bias” hypothesis: estrogen is neuroprotective when brain cells are healthy but potentially harmful when neurodegeneration has begun.
Quality Rating: Negative for late-life EPT initiation, Moderate observational support for midlife ET, Null for early initiation in cognitive RCTs
Testosterone (Men)
Observational Evidence:
The Health in Men Study (Ford et al. 2018), a prospective study of 4,069 Australian men aged 71-88 followed for 10.5 years, found strong associations between low testosterone and dementia risk:
Lowest quartile of total testosterone: 39% increased dementia risk (HR 1.39, 95% CI 1.04-1.85)
Lowest quartile of free testosterone: 43% increased risk (HR 1.43, 95% CI 1.08-1.90)
Risk increased with each standard deviation decrease in testosterone (HR 1.14-1.18)
A meta-analysis of 7 prospective cohort studies found low plasma testosterone significantly associated with 48% increased AD risk in older men (RR 1.48, 95% CI 1.12-1.96). Additional support comes from UK Biobank data (n=159,411 men aged 50-73).
RCT Evidence:
The Testosterone Trials (TTrials) Cognitive Function Trial (n=493 men aged ≥65 with age-associated memory impairment and low testosterone) found no improvement in cognitive function after 1 year of testosterone treatment:
No improvement in verbal memory (primary outcome)
No improvement in visual memory, executive function, or spatial ability
Concerning: cardiovascular sub-study showed testosterone increased non-calcified coronary plaque volume
A 2022 comprehensive review concluded that while observational studies consistently show associations between low testosterone and dementia risk, intervention studies have provided inconsistent results, with the largest RCT showing no cognitive benefit.
Summary: This mirrors the estrogen pattern perfectly. Strong observational evidence links low testosterone to dementia risk, but RCT evidence for testosterone replacement improving cognition is null. The authors of the HIMS study note: “data on the role of testosterone treatment in preventing dementia is lacking.” Lower testosterone may be a biomarker of brain aging rather than a therapeutic target.
Quality Rating: Moderate observational support for association, Null RCT evidence for treatment
Testosterone
Meta-analysis of 23 RCTs shows no significant cognitive benefit. The Testosterone Trials (n=493) found no improvement in any cognitive domain.
Quality Rating: Null
DHEA
A Cochrane Review concluded “no current evidence for improvement in memory or other aspects of cognitive function.”
Quality Rating: Negative
Supplements
Vitamin D
Vitamin D exemplifies the disconnect between observational evidence and intervention results.
Observational Evidence (strong):
A 2025 meta-analysis of 22 studies (n=53,122) found participants in the lowest vitamin D category had 49% higher dementia risk (RR 1.49). Dose-response analysis showed each 10 nmol/L increase associated with 1.2% lower risk.
The UK Biobank study (n=269,229, 14-year follow-up) found:
Vitamin D deficiency: 19-25% increased risk of all-cause dementia, AD, and vascular dementia
Regular vitamin D supplement users: 17% lower AD risk
Effect stronger in younger individuals (55-64) than older (65-69)
Effect stronger in those with obesity
A 2024 meta-analysis found vitamin D deficiency was a risk factor for dementia, AD, and cognitive impairment with nonlinear dose-response relationships.
Intervention Evidence (null):
The VITAL-Cog trial (n=4,218) found no overall cognitive benefit from 2000 IU daily supplementation over 2-3 years.
The Finnish Vitamin D Trial (n=2,492) found neither 1,600 IU nor 3,200 IU daily reduced dementia incidence over 4.2 years of supplementation or 7.7 years of follow-up.
Possible Explanations:
Reverse causation (early dementia causes vitamin D deficiency rather than vice versa)
Confounding (vitamin D deficiency marks other risk factors)
Timing (intervention may need to start decades earlier)
Severity threshold (benefits may be limited to correcting severe deficiency rather than optimization)
Mendelian randomization studies suggest maintaining adequate vitamin D status in people over 60 may slow cognitive decline, but note that RCTs are needed for confirmation.
Quality Rating: Strong observational association, Null for intervention trials
Omega-3 Fatty Acids
Observational and Dietary Evidence (strong):
A meta-analysis of 48 longitudinal studies (n=103,651) found dietary omega-3 intake could lower dementia/cognitive decline risk by ~20%, especially for DHA (RR 0.82). Each 0.1 g/d increment of DHA or EPA intake was associated with 8-10% lower risk. Fish intake of 2 portions per week was associated with 30% reduced AD risk in dose-response analyses.
In the ADNI cohort (n=1,135), long-term omega-3 supplement users showed 64% reduced AD risk (HR 0.36) over 6-year follow-up. Multiple meta-analyses confirm that elevated erythrocyte DHA and plasma EPA levels predict lower cognitive decline risk.
Intervention Evidence (stage-dependent):
A 2025 dose-response meta-analysis of 58 RCTs found each 2000 mg/d omega-3 supplementation showed significant improvement in attention (SMD 0.98), perceptual speed (SMD 0.50), and language.
An overview of systematic reviews of 14 RCTs found 7 of 8 systematic reviews reported MMSE improvement with omega-3 supplementation.
The Cochrane Review in AD patients found no benefit (MD -0.10 for ADAS-Cog).
Emerging consensus: Omega-3 supplementation shows benefit in MCI and prevention contexts but not in established AD. A 2023 review concluded: “Supplementation with DHA in RCTs in those with mild cognitive impairment showed benefit on cognitive decline, whereas there was no benefit in Alzheimer’s disease.”
Quality Rating: Moderate for MCI/prevention, Null for established AD
B Vitamins
The VITACOG trial provides the strongest supplement evidence. In MCI patients with elevated homocysteine:
B vitamins (folic acid 0.8mg + B12 0.5mg + B6 20mg) reduced brain atrophy by 31% (53% in high-homocysteine subgroup)
Regional analysis showed 9-fold reduction in gray matter atrophy in AD-vulnerable regions
Cognitive benefits occurred only in those with elevated baseline homocysteine
Critically, benefits were dependent on adequate omega-3 status. Those with low omega-3 showed no B vitamin benefit; those with high omega-3 showed 73% reduction in atrophy rate.
The FACIT trial showed 3 years of folic acid improved cognitive function in those with elevated homocysteine (>13 μmol/L).
However, other trials have been negative, and the Cochrane Review found no evidence for cognitive benefit in general populations.
Key insight: B vitamins help those with metabolic dysfunction (elevated homocysteine), not replete individuals. The interaction with omega-3 status suggests these nutrients work together.
Quality Rating: Moderate for elevated homocysteine + adequate omega-3, Null for general population
Probiotics
Umbrella Meta-Analysis Evidence:
An umbrella meta-analysis of 13 meta-analyses (n=3,910) found probiotics significantly improved cognitive function in both AD (SMD 0.78, large effect) and MCI (SMD 0.43, moderate effect). Probiotics also improved antioxidant capacity and reduced inflammatory markers.
Individual Meta-Analysis Results (more mixed):
A 2024 meta-analysis of 10 RCTs found non-significant effects across cognitive domains: MMSE (SMD 0.28, p=0.38), MoCA (SMD 0.51, p=0.33), memory (SMD 0.20, p=0.26). The authors noted high heterogeneity and inconsistency across studies.
A 2024 systematic review concluded: “Human studies show no consistent effects of gut microbiota interventions on cognitive function in AD.”
Key effective strains identified include L. plantarum, B. breve MCC1274 (which showed suppressed brain atrophy progression), and multi-strain formulations.
Quality Rating: Moderate (umbrella meta-analysis positive, individual studies mixed)
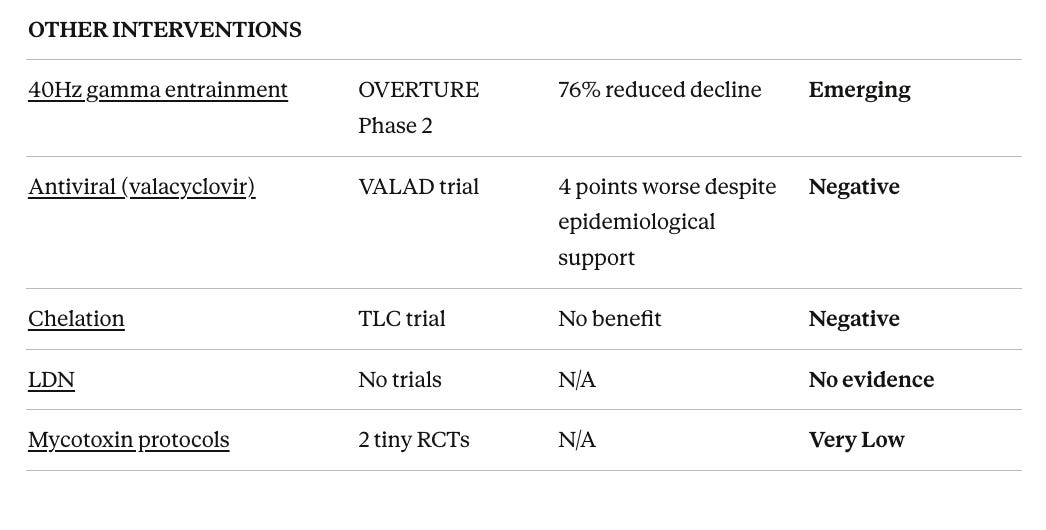
Photobiomodulation and Gamma Entrainment
40 Hz Gamma Entrainment (Cognito Therapeutics)
This intervention has the strongest translational pathway, based on extensive MIT preclinical data showing amyloid clearance and microglial activation.
The OVERTURE trial (Phase 2, n=76, sham-controlled, 6 months) showed:
76% reduction in cognitive decline (ADAS-Cog)
77% slowing of functional decline (ADCS-ADL)
69% reduction in whole brain atrophy
Preserved white matter and myelin (secondary analysis)
18-month extension data showed continued slowing of disease progression with no ARIA-like side effects.
The Phase 3 HOPE trial (n=670) completed enrollment in December 2025 and is the largest device trial for AD.
Quality Rating: Emerging (Phase 2 highly promising, awaiting Phase 3 confirmation)
Hyperbaric Oxygen Therapy
A meta-analysis of 11 RCTs in AD (n=847) found MMSE improvement of 3.08 points and ADAS-Cog improvement of 4.53 points. However, all 11 studies were conducted in China with no sham controls, representing high risk of bias.
The highest-quality evidence comes from sham-controlled post-COVID trials (n=73) showing significant cognitive improvement (d=0.46 to 0.74) with 1-year durability.
Quality Rating: Low for AD (all evidence from single country without sham controls)
Antimicrobial Treatments
Antiviral Therapy for HSV
The VALAD trial (completed 2025) definitively tested the HSV-dementia hypothesis. This Phase II RCT (n=120) found valacyclovir worsened cognitive outcomes in early AD: ADAS-Cog showed 10.86 points worsening versus 6.92 on placebo (3.93 points worse, p=0.01).
This occurred despite strong epidemiological evidence: meta-analyses show HSV-1 associated with AD risk (OR 1.38-1.80), with Taiwan database studies suggesting 90% risk reduction with antiviral treatment. However, RCT results supersede observational associations.
Quality Rating: Negative (treatment worsened outcomes)
Lyme Disease Treatment
RCTs have been consistently negative. The PLEASE trial (n=280) found extended antibiotics did not improve cognitive performance.
Quality Rating: Negative
Low-Dose Naltrexone
No clinical trials exist in MCI or AD populations. Historical AD trials used much higher doses (5-100mg) and were uniformly negative.
Quality Rating: No evidence
Detoxification Protocols
Heavy Metal Chelation
The TLC trial (n=780 children), the largest chelation RCT, found no cognitive benefit. No adult dementia trials exist.
Quality Rating: Negative
Mycotoxin Protocols
Only 2 tiny RCTs (n=8 and n=13) for cholestyramine exist. The Alzheimer’s Drug Discovery Foundation notes “no direct evidence mold exposure causes AD.”
Quality Rating: Very Low
Multi-Domain Interventions in General
The strongest evidence supporting the general Bredesen philosophy comes from multi-domain intervention trials that combine multiple lifestyle modifications.
The FINGER trial (n=1,260, 2 years) tested diet, exercise, cognitive training, and vascular risk management in at-risk older adults. Results showed 25% greater cognitive improvement and 30% reduced risk of cognitive decline. This provides proof-of-concept for multimodal approaches.
However, FINGER enrolled at-risk individuals without cognitive impairment, making it a prevention rather than treatment trial. This distinction is crucial when interpreting Bredesen trial results in MCI/dementia patients.
Why Personalization Might Matter
The Case for Assessment-First Precision Medicine
The Bredesen protocol is marketed as “precision medicine,” but its implementation is closer to “comprehensive medicine with personalized adjustments.” Nearly all participants receive the same core interventions with modifications based on identified abnormalities.
A genuinely precision approach would: (1) comprehensively assess individual risk factors and pathology drivers, (2) target interventions to those specific drivers, and (3) measure whether those specific drivers improved.
Potential advantages:
Efficiency: If testing identifies that Patient A has primary vascular risk factors while Patient B has primary inflammatory drivers, each can receive focused intervention rather than both receiving every possible treatment.
Attribution: When targeted interventions address specific identified problems, outcomes can be attributed to those interventions.
Measurability: Assessment-first approaches allow reassessment to determine whether interventions actually modified the identified risk factors.
Prevention May Be Different
Several patterns in the evidence suggest prevention and treatment require different approaches. The underlying biology may explain why with lifestyle interventions supporting mitochondrial function, reducing inflammation, and enhancing synaptic plasticity in living neurons, but being unable to regenerate the 20-40% of neurons already lost by the time of clinical diagnosis.
Disease stage matters critically. Exercise improves cognition in MCI but showed worse outcomes in the DAPA dementia trial. Hormone replacement shows 32% reduced dementia risk with midlife estrogen-only therapy but 32% increased risk with late-life combined therapy. Low testosterone strongly predicts dementia risk in men, but testosterone replacement trials show no cognitive benefit. Valacyclovir worsened outcomes in early AD despite epidemiological associations. Once substantial neurodegeneration has occurred, interventions that would have been protective may no longer be effective.
Observational associations frequently fail to translate to treatment benefits. Vitamin D deficiency is strongly associated with dementia risk (49% higher), but supplementation trials are negative. Omega-3 levels correlate with brain health, but supplementation in AD shows no benefit. Low testosterone in men increases dementia risk by 39-48%, but testosterone replacement shows no cognitive benefit in RCTs. Infections are associated with increased AD risk, but treating infections in symptomatic patients worsens outcomes.
The prevention evidence is stronger. Mediterranean diet shows 30% AD risk reduction observationally. Exercise shows consistent cognitive benefits in healthy aging. Speed-of-processing training shows 29% dementia risk reduction. These prevention-focused findings are more robust than the treatment literature.
Interventions With Evidence Not Emphasized in the Protocol
Below are a few interventions have RCT or strong observational evidence but receive little emphasis in multimodal protocols like Bredesen’s. There are many more interventions that he didn’t test that could have efficacy.
Hearing Aids
The ACHIEVE trial (n=977, ages 70-84) is one of the few RCTs showing cognitive benefit from an intervention. In participants at higher baseline dementia risk, hearing aids reduced cognitive decline by 48% over 3 years (HR 0.52). The Lancet Commission identifies hearing loss as potentially the largest modifiable dementia risk factor (8% of cases), yet it receives minimal attention in most cognitive protocols.
A 2025 Framingham study found hearing aid use in those under 70 reduced dementia risk by 61% over 20 years.
Intensive Blood Pressure Control
SPRINT-MIND (n=9,361) is the first RCT to show an intervention significantly reduces MCI. Intensive BP control (<120 mmHg vs <140 mmHg) reduced MCI by 19% (HR 0.81) and combined MCI/dementia by 15% (HR 0.85). Seven-year follow-up showed sustained benefits. While the protocol addresses vascular risk factors, aggressive BP targets specifically are not emphasized.
GLP-1 Agonists
Multiple large observational studies show GLP-1 receptor agonists (semaglutide, liraglutide) associated with 40-70% reduced dementia risk compared to other diabetes medications. A 2024 target trial emulation found semaglutide associated with 67% reduced AD diagnosis (HR 0.33 vs insulin). However, the EVOKE trials (Phase 3, n=1,800) showed no benefit for treating established AD - another example of the prevention-treatment disconnect. GLP-1s may work through weight loss, metabolic improvement, and anti-inflammatory mechanisms rather than directly on AD pathology.
Conclusions
The Trial
The Bredesen Precision Medicine Trial represents an important step forward from case reports to controlled data. The cognitive improvements were substantial (14-point NCI gain vs 4-point decline in controls), and the safety profile was favorable. However, critical limitations prevent strong conclusions:
No blinding of participants, physicians, or health coaches
No significant difference on MoCA, the most widely-used clinical measure
No significant brain volume improvements on MRI
Incomplete reporting of epigenetic clock results (only OMICmAge reported; GrimAge results not included)
Participants were metabolically healthy at baseline, undermining the “metabolic dysfunction” narrative
Impossible to attribute effects to any specific intervention
The Evidence Landscape
This review found the protocol components span from high-quality evidence to no evidence at all:
Strong evidence: Speed-of-processing cognitive training (ACTIVE trial: 29% dementia risk reduction), Mediterranean diet (HR 0.70 for AD)
Moderate evidence for specific populations: Exercise in MCI (but harmful in established dementia), B vitamins for elevated homocysteine (but only with adequate omega-3), omega-3s in MCI (but not AD), ketogenic diet, probiotics
Null or negative evidence: CPAP for cognition (despite strong sleep apnea-dementia association), vitamin D supplementation (despite strong observational links), late-life hormone replacement (increased dementia risk), testosterone replacement (no cognitive benefit despite observational associations), valacyclovir (worsened outcomes), chelation, DHEA
No evidence: Low-dose naltrexone, liposomal glutathione, many nootropic supplements in the ReCODE stack.
Prevention vs Treatment: The Central Pattern
The most consistent finding across this evidence review is that interventions effective for prevention frequently fail or cause harm in treatment:
Exercise: 20% reduced dementia risk observationally, but worse outcomes in established dementia (DAPA trial)
Estrogen: 32% reduced risk with midlife initiation, 32% increased risk with late-life initiation
Testosterone: 39-48% increased risk with low levels, but no benefit from replacement
Omega-3s: 20-64% reduced risk observationally, null in AD treatment trials
OSA treatment: Strong risk association, null cognitive benefit from CPAP
Valacyclovir: Epidemiological support for viral hypothesis, but worsened AD outcomes
This pattern has profound implications. The Bredesen approach may be most valuable for prevention in cognitively normal individuals, not for reversing established disease. The trial enrolled MCI/early dementia patients, precisely the population where many interventions fail.
Summary
The Bredesen trial advances the conversation beyond anecdote but does not prove the protocol works. The cognitive improvements could reflect true disease modification, intensive attention and support, placebo effects, or some combination. The inability to identify which components drive effects, combined with evidence that many components are ineffective or harmful, makes wholesale adoption of the full protocol premature.
For patients and clinicians, a reasonable approach is to prioritize components with independent evidence (Mediterranean diet, exercise for MCI, speed-of-processing training, B vitamins for elevated homocysteine) while remaining skeptical of expensive supplements with no RCT support. Prevention and treatment may require somewhat different strategies.
Future trials should test lifestyle interventions alone versus lifestyle plus evidence-based supplements versus the full protocol, with proper blinding and active controls. Until then, we cannot know whether the supplement stack contributes anything beyond what diet and exercise provide.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso