mTOR: Genetic Variants, Rapamycin, Longevity, and Brain Health
The mammalian target of rapamycin (mTOR) pathway is arguably the most studied and therapeutically actionable longevity pathway in biology. This ancient nutrient-sensing pathway integrates signals from growth factors, nutrients, energy status, and stress to coordinate cellular growth, metabolism, and aging.
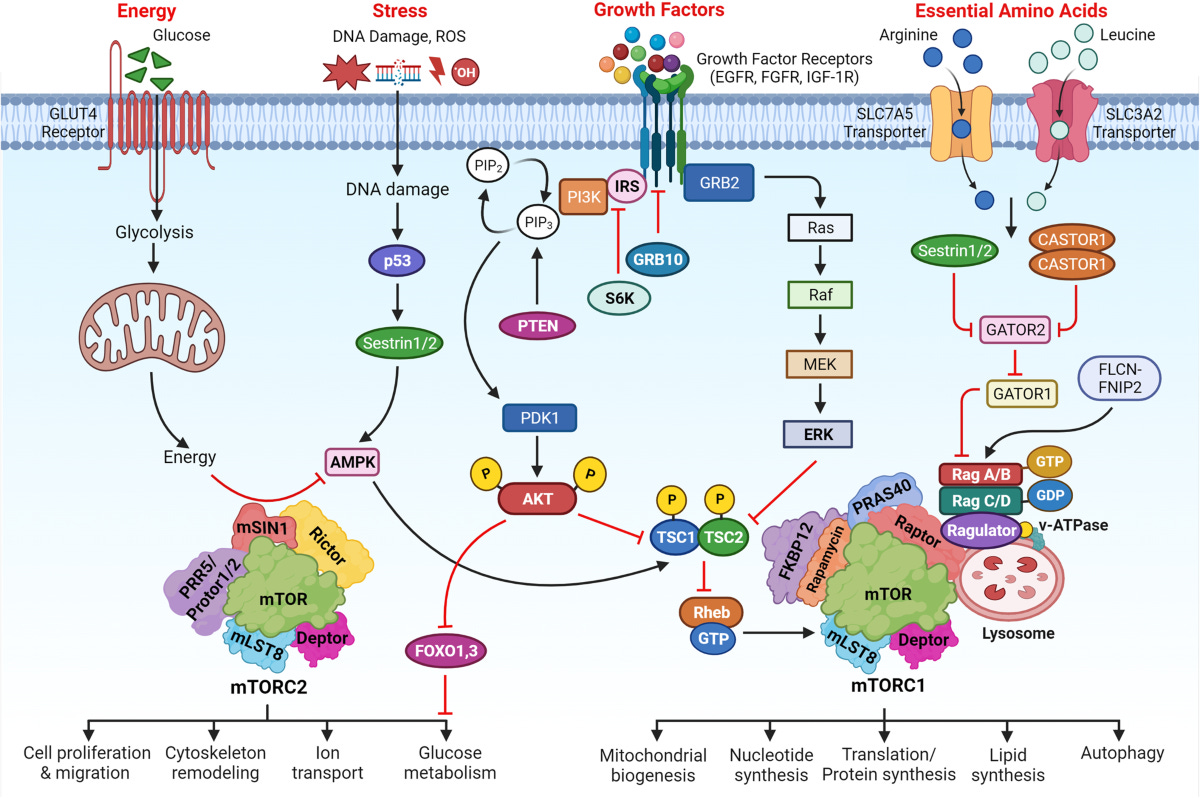
The story of mTOR is one of the most compelling in longevity science. It’s one of the few pathways where we already have FDA-approved drugs (rapamycin and its analogs) that extend lifespan in multiple species and are being used off-label by longevity enthusiasts.
But the mTOR story is more nuanced than “inhibition equals longevity.” This pathway represents a fundamental trade-off in biology: between growth and reproduction on one side, and maintenance and longevity on the other. Understanding where you sit on this spectrum through genetics, biomarkers, and personal health status is key to optimizing this pathway for healthy aging.
What is mTOR and Why Does It Matter?
mTOR is a serine/threonine protein kinase that exists in two distinct complexes: mTORC1 and mTORC2. Think of mTOR as your cell’s master growth controller—a molecular switch that determines whether your cells should be in “growth and building mode” or “maintenance and repair mode.”
When mTOR is activated (high nutrient availability, growth signals):
Protein synthesis increases
Cell growth and proliferation accelerate
Autophagy (cellular cleanup) is suppressed
Mitochondrial function shifts toward biosynthesis
Anabolic processes dominate
When mTOR is inhibited (nutrient scarcity, stress signals):
Autophagy is activated (cells clean up damaged components)
Stress resistance increases
Mitochondrial quality control improves
Cellular maintenance and repair processes activate
Longevity pathways engage
The discovery of mTOR began in the 1970s with rapamycin, a compound isolated from soil bacteria on Easter Island (Rapa Nui—hence “rapamycin”). Originally developed as an antifungal, then as an immunosuppressant for organ transplants, rapamycin’s anti-aging properties were discovered later when researchers identified its molecular target: mTOR.
mTOR Genetic Variant: rs2295080
The rs2295080 SNP is located in the mTOR gene and represents a single nucleotide change (G>T) that affects mTOR function. This variant has emerged as one of the most studied mTOR polymorphisms in relation to human longevity and disease.
The G allele (protective variant) is associated with:
Reduced mTOR signaling activity and lower transcriptional activity - the T allele enhances mTOR transcriptional activity approximately 0.5-fold over the G allele, resulting in reduced mTOR mRNA expression
Decreased risk of multiple cancers including gastric, colorectal, breast, and genitourinary cancers
23% decreased risk of gastric cancer (OR = 0.77, 95% CI = 0.65-0.92)
Enhanced cellular stress resistance
Protective against atherosclerosis in diabetic women
The T allele (risk variant) is associated with:
Potentially faster growth and development in early life
Higher baseline mTOR signaling activity
Mixed effects on cognition depending on age and context
Direct evidence linking mTOR genetic variants to exceptional longevity is lacking. However, studies of centenarian genetics have identified mTOR pathway variants among the genes contributing to extreme longevity.
Interestingly, studies have shown complex patterns across populations. However, the consistent finding across cancer studies is that the TT genotype (higher mTOR activity) is associated with increased cancer risk, while the G allele provides protection. This highlights an important theme in mTOR biology: the optimal level of mTOR activity may depend on age, tissue, metabolic state, and genetic background.
Curious whether you have the mTOR longevity variant? If you’ve had your NeuroAge genetic resilience analyzed, you can check for rs2295080 in the longevity genes section of your report.
mTOR and Longevity
If there’s one pathway where the longevity evidence is overwhelming, it’s mTOR inhibition. This is perhaps the most reproduced finding in aging biology.
Animal Lifespan Studies
Yeast: mTOR inhibition extends replicative lifespan by 30-40%
Worms (C. elegans): Genetic reduction of mTOR signaling extends lifespan by 25-30%
Flies (Drosophila): mTOR inhibition extends lifespan by 10-20% in both males and females
Mice: This is where the evidence becomes most compelling for mammals:
Rapamycin extends lifespan by 9-14% in male mice and 13-18% in female mice when started in middle age
Even when started very late in life (20 months, equivalent to age 60 in humans), rapamycin extends remaining lifespan
Rapamycin extends both median and maximum lifespan
The Interventions Testing Program (ITP), a rigorous multi-site study, has now replicated these findings across multiple cohorts and dosing regimens
Dogs: The Dog Aging Project is currently testing rapamycin in companion dogs (the TRIAD trial). Early results show improved cardiac function and potential healthspan benefits. Dogs share more similar physiology to humans than mice, making this trial particularly relevant.
Marmosets: Ongoing trials in these small primates may provide data more translatable to humans.
Rapamycin in humans: Thousands of organ transplant patients have taken rapamycin for decades as an immunosuppressant. Interestingly, these patients show reduced cancer incidence compared to patients on other immunosuppressants and lower rates of some age-related diseases (though confounded by immunosuppression). Importantly, these patients tolerate long-term rapamycin, providing safety data.
mTOR and Brain Health: A Complex Relationship
Here’s where the mTOR story gets more complicated. Unlike pathways such as PCSK9 where lower is simply better for longevity, mTOR presents a context-dependent paradox in the brain.
The mTOR balance required for brain health
Too much mTOR activity (chronic overactivation):
Impairs autophagy, leading to accumulation of damaged proteins and organelles
Promotes tau phosphorylation and aggregation in Alzheimer’s disease
Contributes to neuroinflammation
Associated with accelerated brain aging
Linked to neurodegenerative diseases
Too little mTOR activity (chronic suppression):
Could reduce protein synthesis needed for long-term memory formation
Might decrease neurotrophic support
Could impair myelination
The key insight: The brain needs variable mTOR activity—activation during learning and memory formation, followed by inhibition to clear damaged proteins through autophagy. Chronic activation or chronic suppression may both be detrimental.
mTOR in Alzheimer’s Disease
The evidence suggests mTOR overactivation contributes to Alzheimer’s pathology:
Brain tissue studies in Alzheimer’s patients show:
Elevated mTOR activity in vulnerable brain regions: Evidence from postmortem human AD brains indicates that levels of phospho-mTOR and downstream targets p70S6K and eIF4E are increased compared to age-matched controls, suggesting higher mTOR activity in AD brains
Increased mTORC1 signaling correlates with tau pathology burden: Phosphorylated p70S6K levels correlated with Braak’s stage and the levels of total and P-tau
Hyperactive mTOR is found in neurons with tau tangles: Confocal microscopy shows that activated p70S6K levels are higher in neurons that later develop neurofibrillary tangles
Preclinical studies in Alzheimer’s mouse models show that rapamycin:
Reduces amyloid-beta (Aβ) and tau pathology: Rapamycin treatment enhanced Amyloid clearance and reduced deposition in Alzheimer’s mouse models
Enhances autophagy and clearance of toxic protein aggregates: Rapamycin inhibits mTOR and activates autophagy, which enhances clearance and degradation of amyloid and tau
Improves cognitive function: Meta-analysis showed that rapamycin therapy shortened escape latency by 15.60 seconds and increased traversed platforms by 1.53 times in Alzheimer’s mouse models
Reduces neuroinflammation: Rapamycin decreased pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) and increased anti-inflammatory IL-10 secretion
Preserves synaptic function: Rapamycin increased synaptic plasticity-related proteins synaptophysin and PSD-95
Mechanism
mTOR inhibition promotes autophagy, which helps clear misfolded proteins including Aβ and tau. It also reduces inflammation and oxidative stress, both contributors to neurodegeneration.
Important Caveat: Timing and Lysosomal Function
However, there’s an important caveat: Some studies suggest that rapamycin may only be effective when administered early in disease progression, before extensive lysosomal damage occurs:
Early treatment efficacy: Rapamycin is effective only if administered prior to formation of pathological features in an Alzheimer’s mouse model. Studies show rapamycin prevents tau accumulation when given early (2-5 months) but not later (3.5-5 months) in disease progression
Lysosomal damage concern: Later in Alzheimer’s disease, the brain’s lysosomal system is severely damaged and treatment with rapamycin is likely to exacerbate this damage. Treatment would increase lysosomal cargo generation without fixing underlying lysosomal dysfunction
Recommendation for early intervention: Clinical trials should focus on MCI patients or middle-aged populations with early amyloid plaque accumulation
The Pulsatile/Intermittent Rapamycin Hypothesis
This has led to the “pulsatile rapamycin” hypothesis—intermittent dosing may provide benefits while avoiding potential cognitive side effects:
Intermittent dosing approaches: Rapamycin extends lifespan when administered continuously, intermittently, or transiently, and side effects are dose-dependent and reversible
Safety of intermittent dosing: Prolonged rapamycin use may cause glucose dysregulation via mTORC2 inhibition, prompting investigations of intermittent dosing safety profiles
Clinical trials testing intermittent protocols: The ERAP trial is evaluating weekly 7 mg rapamycin doses for six months in early-stage AD patients
Human Clinical Trial Evidence
Despite compelling preclinical data, rapamycin has been dramatically underexplored in human AD trials. In 2019, Kaeberlein and Galvan argued that the robust animal evidence, combined with rapamycin’s established safety profile, warranted immediate clinical testing in AD patients.
In 2025, the first Phase 1 clinical trial of rapamycin in Alzheimer’s disease was published. Ten participants with mild cognitive impairment or early-stage dementia received 1 mg/day rapamycin for eight weeks. The results were mixed:
Key findings:
Rapamycin was not detectable in cerebrospinal fluid (CSF) before or after treatment, raising questions about brain penetration at this dose
Several Alzheimer’s disease biomarkers increased rather than decreased: CSF phosphorylated tau-181, glial fibrillary acidic protein (GFAP), and neurofilament light all significantly increased
Multiple inflammatory markers in plasma also increased
No significant cognitive changes were observed during the 8-week treatment period
The treatment was generally well tolerated with mostly mild adverse events
Interpretation: These surprising results suggest that low-dose rapamycin (1 mg/day) may not achieve therapeutic CSF levels, and the increases in pathological biomarkers raise important questions. The authors note that higher doses, different formulations, or longer treatment duration may be necessary to see beneficial effects. Alternatively, the biomarker increases could represent compensatory responses or indicate that rapamycin’s effects in humans differ from animal models. It will be interesting to follow larger trials with varied dosing regimens and longer follow-up to better understand rapamycin’s potential in human Alzheimer’s disease.
mTOR in Other Neurodegenerative Diseases
Parkinson’s Disease:
mTOR overactivation impairs autophagy of damaged mitochondria and α-synuclein aggregates
Rapamycin shows neuroprotective effects in Parkinson’s models
Enhances clearance of toxic protein aggregates through autophagy
Huntington’s Disease:
mTOR inhibition reduces mutant huntingtin protein aggregation
Rapamycin improves motor function in animal models
ALS (Amyotrophic Lateral Sclerosis):
Evidence is mixed, with some studies showing benefit and others showing no effect
May depend on disease stage and genetic background
Cognitive Function in Aging
Studies in older adults without dementia show complex patterns:
Observational human data is limited but suggests:
Very low mTOR activity (through chronic caloric restriction) may not be optimal for cognition in older adults
Moderate mTOR activity may support cognitive reserve
High mTOR activity is associated with worse cognitive aging
Animal studies in aged mice show:
Rapamycin can improve memory in various paradigms including spatial learning and passive avoidance tasks
Benefits appear greater when started in middle age rather than very late life - one study showed rapamycin improved cognition when started at 2 months but not when started at 15 months in mice with pre-existing deficits
Intermittent dosing may be superior to continuous dosing for cognitive outcomes
Rapamycin restores memory even when treatment begins after disease onset in AD models
Studies in aged rats show rapamycin prevents deficits in learning and memory, neurovascular uncoupling, and restores cerebral perfusion
mTOR Therapeutics: From Rapamycin to Next-Generation Rapalogs
The mTOR inhibitor landscape spans from repurposed drugs to novel compounds in development.
First Generation: Rapamycin (Sirolimus)
FDA approved: 1999 (for organ transplant rejection prevention)
Mechanism: Binds to FKBP12 protein, which then inhibits mTORC1 (but not fully mTORC2)
Current approved uses:
Immunosuppression after organ transplantation
Coronary artery stent coatings
Certain cancers (lymphangioleiomyomatosis, specific kidney cancers)
Off-label use for longevity:
Growing community of physicians and patients using rapamycin for aging
Typical longevity protocols: 3-8 mg once weekly or 1-2 mg 2-3× weekly
Intermittent dosing appears to minimize side effects while maintaining benefits
Known side effects at immunosuppressive doses:
Increased infection risk (at high doses used for transplant)
Mouth ulcers (aphthous ulcers)
Hyperlipidemia (elevated cholesterol and triglycerides)
Impaired wound healing
Potential glucose intolerance
At lower longevity doses: Many side effects are reduced, but mouth ulcers remain the most common complaint. Most can be managed with oral rinses.
Second Generation: Rapalogs
These are rapamycin derivatives designed to improve pharmacokinetics or tissue selectivity.
Everolimus (RAD001)
FDA approved for various cancers and organ transplantation
Shorter half-life than rapamycin
Used in longevity protocols by some physicians
Similar side effect profile
Temsirolimus (CCI-779)
FDA approved for renal cell carcinoma
Intravenous administration
Not practical for longevity use
These drugs all work through similar mechanisms—FKBP12 binding and mTORC1 inhibition.
Third Generation: Selective mTOR Inhibitors
RapaLink-1 (preclinical)
Designed to inhibit both mTORC1 and mTORC2
Overcomes feedback mechanisms that limit first-generation drugs
Still in research phase
Bi-steric mTOR inhibitors (preclinical)
Novel mechanism to achieve more complete mTOR inhibition
Potential for improved efficacy with fewer side effects
Years away from clinical use
Tissue-selective mTOR inhibitors
The goal: inhibit mTOR in liver/muscle/fat for metabolic benefits while sparing immune system
Multiple approaches in early development
Could represent the ideal longevity intervention if successful
Rapamycin: Who Should Consider It and Who Shouldn’t?
This is the practical question for patients interested in longevity interventions.
The Case For Considering Rapamycin (Under Medical Supervision)
Strong candidates might include:
Middle-aged to older adults (45+) in good health seeking preventive aging interventions
People with family history of cancer (given cancer prevention effects in animals)
Those with early signs of metabolic syndrome (though monitoring is crucial)
Individuals with genetic variants suggesting high mTOR activity (like rs2295080 TT genotype)
People already optimizing other longevity pillars (diet, exercise, sleep) looking to add pharmacological intervention
Potential benefits based on animal data and limited human data:
Enhanced autophagy and cellular cleanup
Reduced cancer risk
Improved immune function (the “immunological rebound” phenomenon)
Better metabolic health markers in some individuals
Potential cognitive protection (though data is mixed)
Possible lifespan extension
The Case AGAINST or for Caution with Rapamycin
People who should avoid or be very cautious:
Those with active infections or chronic infectious diseases
People with poor wound healing or upcoming surgery
Individuals with diabetes or prediabetes (requires careful monitoring)
Those with significant dyslipidemia
Anyone not willing to do regular monitoring labs
People taking multiple medications without physician oversight
Those with autoimmune conditions (effects are complex)
Remaining uncertainties:
Optimal human dosing remains unknown (though weekly 3-8 mg is most common in longevity protocols)
Long-term safety data in healthy humans is limited
Effects on cognition need better human data
Individual variation in response (pharmacogenomics) is not well characterized
Interaction effects with other longevity interventions unclear
The Monitoring Protocol
If working with a physician on rapamycin for longevity:
Before starting:
Comprehensive metabolic panel
Lipid panel
HbA1c and fasting glucose
Complete blood count
Liver and kidney function
Regular monitoring (every 3-6 months):
Same panels as above
Watch particularly for: elevated lipids, increased glucose, cytopenias
Adjust dose or discontinue if problematic changes occur
Clinical assessment:
Watch for mouth ulcers (most common side effect)
Monitor wound healing
Track infections
Assess energy levels and cognitive function
How to Modulate mTOR Naturally: Diet, Exercise, and Supplements
While rapamycin is the most potent mTOR inhibitor, several lifestyle and dietary interventions modulate this pathway.
How to Modulate mTOR Naturally: Diet, Exercise, and Supplements
While rapamycin is the most potent mTOR inhibitor, several lifestyle and dietary interventions modulate this pathway.
Dietary Approaches to Reduce mTOR
Caloric Restriction:
The most well-established way to reduce mTOR activity
Caloric restriction operates at least in part by decreasing mTOR signaling and extends lifespan in mice
Sustained caloric restriction extends lifespan in multiple species
Challenge: adherence and potential negative effects (muscle loss, low energy)
Protein Restriction (mouse studies):
mTOR is particularly sensitive to amino acids, especially leucine, methionine, and arginine
Reducing protein intake decreases mTOR activation
Plant proteins cause less mTOR activation than animal proteins (lower in leucine)
Very low protein may impair muscle maintenance—balance is key
Intermittent Fasting:
Time-restricted eating (16:8 or 14:10 protocols) cyclically inhibits mTOR
Prolonged fasts (24-72 hours) provide deeper mTOR inhibition
May provide benefits of mTOR inhibition while allowing adequate protein on feeding days
Potentially superior to continuous restriction for maintaining muscle
Fasting-Mimicking Diet (FMD):
Designed by Dr. Valter Longo to inhibit mTOR while being more tolerable than water fasting
May be optimal for those unable to do complete fasting
Low Methionine Diet:
Methionine is a key amino acid that activates mTOR
Animal studies show methionine restriction extends lifespan similarly to caloric restriction
Practical approach: reduce animal protein, especially red meat
Exercise and mTOR: Context Matters
Resistance training:
This is beneficial for muscle growth and maintenance
Creates pulsatile mTOR activation followed by inhibition
Especially important in older adults to prevent sarcopenia
Endurance exercise:
Promotes autophagy and mitochondrial quality
Complements resistance training
The ideal: Combine both modalities—resistance training for pulsatile muscle mTOR activation, endurance training for chronic systemic mTOR reduction.
Natural Compounds That Affect mTOR
EGCG (from green tea):
Also activates AMPK (which inhibits mTOR)
Typical dose: 300-500 mg daily of standardized extract
Generally safe with few side effects
May provide mild benefits but far less potent than rapamycin
Curcumin:
Shows mTOR inhibitory effects in cancer cell lines
Effects in healthy humans unclear
Bioavailability is poor without piperine or liposomal formulations
Anti-inflammatory effects may be more significant than direct mTOR effects
Berberine:
Activates AMPK, which inhibits mTOR
Evidence for metabolic benefits in humans (glucose control)
Typical dose: 500 mg 2-3× daily
May be useful for metabolic health independently of direct mTOR effects
Note: These compounds show mTOR-modulating effects in cells and sometimes animals, but the magnitude is much smaller than rapamycin. They should be viewed as complementary to lifestyle interventions, not replacements for them and not equivalent to pharmaceutical mTOR inhibition.
Conclusion
mTOR represents perhaps the most therapeutically actionable aging pathway we currently have. The evidence spans from yeast to mammals, and we already have safe, FDA-approved drugs (rapamycin and rapalogs) that can modulate this pathway.
Key takeaways:
On Genetics:
The rs2295080 G allele (reduced mTOR activity) is the protective variant associated with longevity and decreased cancer risk across multiple populations
The T allele is associated with higher mTOR activity and increased cancer risk
Your genotype can inform personalized mTOR optimization strategies
Check your NeuroAge genetic resilience dashboard for this variant to guide your approach
On Longevity:
mTOR inhibition robustly extends lifespan in every organism tested, from yeast to mice
The effect size is substantial (10-18% lifespan extension in mice)
Genetic evidence in humans supports longevity association with reduced mTOR
Effects work through multiple mechanisms: enhanced autophagy, reduced cancer risk, improved stress resistance
On Brain Health:
The relationship is complex and context-dependent
Chronic mTOR overactivation contributes to Alzheimer’s and neurodegeneration
But some mTOR activity is needed for synaptic plasticity and memory
Pulsatile or intermittent mTOR inhibition may be optimal for brain health
Rapamycin shows neuroprotective effects in animal models
On Therapeutics:
Rapamycin is already available and used by a growing longevity community
Typical longevity doses: 3-8 mg weekly (much lower than immunosuppressive doses)
Side effects at low doses are generally manageable (mouth ulcers most common)
Requires physician supervision and monitoring
Next-generation selective inhibitors may improve benefit/risk ratio
On Natural Approaches:
Caloric restriction and protein restriction reduce mTOR activity
Intermittent fasting provides pulsatile mTOR inhibition
Exercise creates beneficial fluctuations in mTOR
Natural compounds (EGCG, curcumin, berberine) have modest effects
Combining approaches may be synergistic
The Paradox:
mTOR activation is needed for muscle growth, wound healing, immune function
mTOR inhibition is needed for longevity, autophagy, cancer prevention
The solution: pulsatile or cyclical approaches that get benefits of both states
Avoid chronic extremes in either direction
Personalization Matters:
Optimal mTOR modulation depends on age, genetics, metabolic status, and goals
Use genetic information (rs2295080), biomarkers (IGF-1, insulin), and functional status (muscle mass, cognition) to guide decisions
Work with knowledgeable physicians if considering rapamycin
If you’ve had your genetics analyzed through NeuroAge, check your mTOR variant status. Combined with metabolic biomarkers and personal health goals, this information can help you and your healthcare provider potentially develop a personalized mTOR optimization strategy whether through diet and lifestyle interventions alone or potentially including pharmaceutical options like rapamycin if this makes sense for you.
If brain health is your primary goal, as it is for me with my family history and genetic risk for Alzheimer’s, I think the jury is still out on rapamycin. The data looks strong in mice but it’s unclear whether rapamycin crosses the blood brain barrier. It’s also unclear if we understand well enough how to modulate it to have the benefits and none of the harms for the brain.
The mTOR pathway beautifully illustrates the complexity of longevity biology: it’s not simply about maximizing or minimizing any single pathway, but rather about finding the right balance for your individual biology and life stage. As we gather more human data and develop more sophisticated interventions, our ability to personalize mTOR modulation will only improve.
Stay tuned for updates as clinical trials progress and next-generation mTOR therapeutics move through development.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso



