The New York Times Called Galleri a Failed Test. I Think They Got It Wrong.
I took my first Grail Galleri multi-cancer detection test this year in preparation for helping with the R42 Group Longevity Retreat. I think this test is worthwhile for the peace of mind it brings. Also, anecdotally, it detected serious cancers in several people in my circles earlier than they would have otherwise been detected. This quite possibly could save or extend their lives substantially. I personally will be incorporating Galleri into my life and I hope that it does become reimbursed by insurance in the US.
Last week, Grail announced topline results from the NHS-Galleri trial, the largest randomized controlled trial ever conducted on a multi-cancer early detection (MCED) blood test. The trial enrolled 142,000 adults aged 50 to 77 across the UK’s National Health Service and followed them through three rounds of annual screening over three years.
The headlines were brutal. Grail’s stock dropped roughly 50% in a single trading session. The New York Times ran a piece titled “Grail’s Cancer Detection Test Fails in Major Study,” in which Dr. Richard Houlston, a cancer genetics researcher at the Institute of Cancer Research in Britain, was quoted saying the study results show that Galleri is “not a very good test” and that the findings don’t support rollout within the American health care system.
There is a more layered story here, and it requires separating the performance of the test from the design of the trial, and both from the question of what screening is actually supposed to accomplish.
How Galleri Works
Galleri is a blood-based multi-cancer early detection (MCED) test, sometimes referred to as a liquid biopsy. The test requires a standard venous blood draw of approximately 20 mL. From that sample, the test isolates cell-free DNA (cfDNA), which are small fragments of DNA that all cells, including tumor cells, shed into the bloodstream as they turn over and die.
What makes Galleri different from tests that look for specific genetic mutations is that it analyzes methylation patterns on this cell-free DNA. Methylation is a chemical modification in which methyl groups attach to specific positions on DNA, and these patterns differ between normal tissue and cancerous tissue. Tumor cells produce distinctive methylation signatures that differ not only from healthy tissue but also vary by the tissue of origin. By using targeted bisulfite sequencing across more than 100,000 methylation regions and a machine learning classifier trained on data from thousands of cancer and non-cancer samples, Galleri can detect the presence of a cancer signal and predict the likely organ of origin, a feature Grail calls Cancer Signal Origin (CSO) prediction.
The test screens for signals shared across more than 50 cancer types, including many for which no standard screening exists, such as pancreatic, ovarian, liver, esophageal, and stomach cancers. When a cancer signal is detected, the CSO prediction guides the diagnostic workup, directing clinicians toward the organ system most likely harboring the tumor rather than requiring an unfocused search. The test is intended for adults aged 50 and older and is designed as an adjunct to, not a replacement for, existing recommended cancer screenings such as mammography, colonoscopy, and low-dose CT for lung cancer.
Galleri is currently available in the United States with a prescription at a list price of $949. It has been sold commercially since 2021 as a laboratory-developed test (LDT) under CLIA certification, though very few insurers cover it. Grail submitted its FDA premarket approval (PMA) application in January 2026.
Understanding the Test’s Accuracy
These numbers are central to the debate about whether Galleri is ready for widespread use.
Sensitivity refers to the test’s ability to correctly identify people who actually have cancer. In the PATHFINDER 2 study (35,878 participants, results presented at ESMO 2025), Galleri’s overall sensitivity across all cancer types was 40.4%. For the 12 cancer types responsible for approximately two-thirds of cancer deaths in the US, sensitivity was 73.7%.
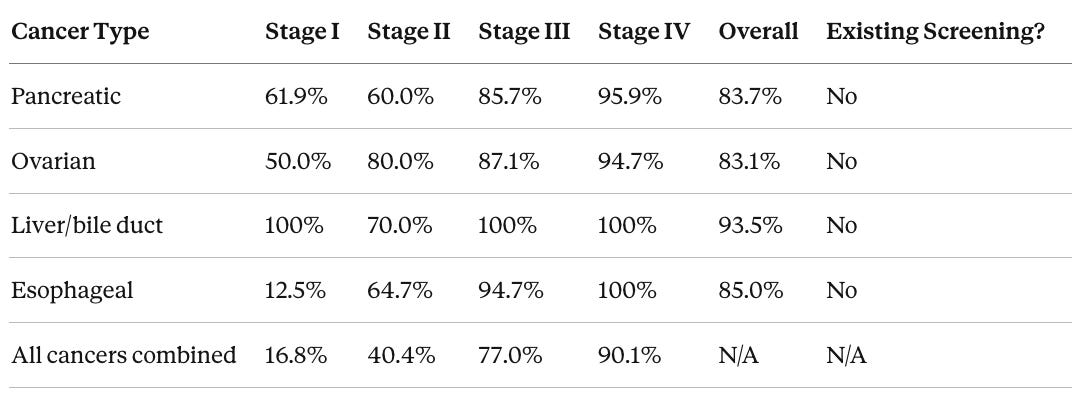
The stage-specific numbers are central to both the criticism and the defense of this test. In the CCGA clinical validation study, overall sensitivity by stage was approximately 16.8% for Stage I, 40.4% for Stage II, 77.0% for Stage III, and 90.1% for Stage IV. That Stage I number is legitimately low and frequently cited by critics. However, it aggregates across all 50+ cancer types, including cancers like kidney and breast that shed relatively little cfDNA. For the cancers Galleri is most uniquely positioned to detect, the ones with no existing screening, early-stage sensitivity is substantially higher.
Source: CCGA clinical validation study; Grail cancer-specific data
This pattern makes biological sense. More aggressive tumors shed more cfDNA into the bloodstream even at early stages. The cfDNA-based approach is inherently biased toward detecting the cancers most likely to kill you, which may actually reduce the risk of overdiagnosis that plagues some existing screening programs. In PATHFINDER 2, more than half (53.5%) of cancers detected by Galleri were Stage I or II, and more than two-thirds (69.3%) were Stages I through III.
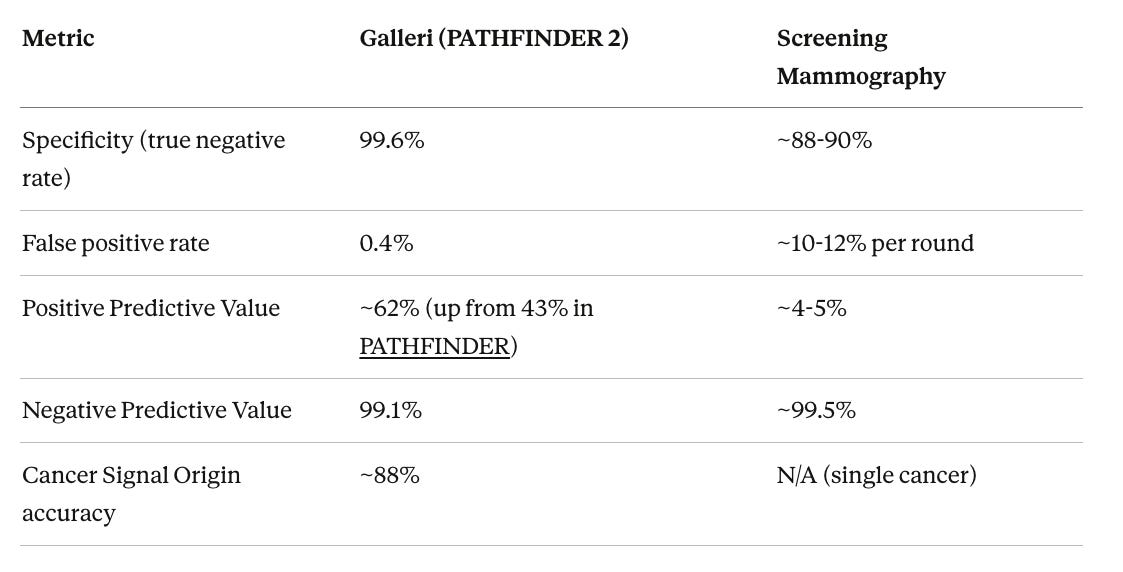
The other performance metrics from PATHFINDER 2 are equally important context for evaluating this test, particularly when compared to an established screening tool like mammography.
Positive Predictive Value (PPV) is the metric that matters most to any individual who receives a positive result. If the test says a cancer signal was detected, what is the probability that cancer is actually present? Galleri’s PPV of approximately 62% means roughly 6 out of every 10 people who receive a Cancer Signal Detected result are confirmed to have cancer. For mammography, the vast majority of positive results do not turn out to be cancer. The important caveat on the NPV side is that a negative Galleri result does not guarantee the absence of cancer, particularly early-stage cancers that may not yet shed sufficient cfDNA to be detected.
The Primary Endpoint Was Not Met
The trial was designed to demonstrate a statistically significant reduction in combined Stage III and Stage IV cancer diagnoses in the intervention arm compared to standard of care alone, focused on a prespecified group of 12 cancer types that together account for approximately two-thirds of cancer deaths in England and the United States. These 12 types include pancreatic, ovarian, lung, liver/bile duct, esophageal, stomach, colorectal, bladder, head and neck, anal, lymphoma, and myeloma.
That combined Stage III-IV reduction did not reach statistical significance. Grail has not disclosed the specific numbers yet, stating that detailed results will be presented at the ASCO 2026 Annual Meeting.
This is the fact of the matter, and it needs to be stated plainly. In the language of clinical trials, the study was negative on its primary endpoint.
The Secondary Findings Tell a Different Story
Where it gets more interesting is in the secondary endpoints. According to Grail’s press release, the trial demonstrated several clinically meaningful signals.
Stage IV cancer diagnoses in the 12 pre-specified deadly cancers decreased with each year of sequential screening, with greater than 20% reduction in Stage IV diagnoses observed in the second and third screening rounds. This is a critical observation. A Stage IV reduction that intensifies over successive screening rounds suggests that the test is catching cancers that would otherwise have been found at the most advanced and lethal stage.
Adding Galleri to standard-of-care screening produced a four-fold improvement in overall cancer detection rate compared to standard-of-care screening alone for breast, colorectal, cervical, and high-risk lung cancers. Galleri increased cancer detection seven-fold when added to USPSTF-recommended screenings, and approximately three-quarters of the cancers it detected were types without any recommended screening test. There was also a substantial increase in absolute Stage I and II diagnoses among the 12 deadly cancer types in the intervention arm.
This pattern of findings, where Stage IV cancers decrease while Stage I-II diagnoses increase, represents the “stage shift” that MCED screening aims to produce.
Why the Primary Endpoint May Have Been the Wrong Question
The trial chose combined Stage III-IV reduction as its primary endpoint. A paper by the trial investigators published in JNCI defended this choice while acknowledging its limitations, noting that NHS-Galleri is the first double-blind cancer screening RCT ever conducted and that it uses late-stage cancer incidence rather than mortality as its primary outcome, a pragmatic decision since mortality data would take many more years.
But there was a higher than anticipated incidence of Stage III cancers in the trial, which inflated the combined Stage III-IV denominator and may have obscured the Stage IV reduction. Stage III and Stage IV are meaningfully different clinical categories, with dramatically different five-year survival rates for most solid tumors. A trial that shifts cancers from Stage IV to Stage III is demonstrating a survival-relevant benefit that would not register as a “win” on the combined endpoint.
Ruth Etzioni, a biostatistician at the University of Washington, commented that she was not ready to call the trial a failure and that it may have been too short. Eric Topol of the Scripps Research Translational Institute argued that the trial should have enrolled participants based on cancer risk factors rather than screening a general population, which would likely have produced a stronger signal by concentrating the study on individuals more likely to develop cancer during the study period.
There is also a signal dilution problem baked into the endpoint itself. The 12 pre-specified cancer types include colorectal cancer and lung cancer, both of which already have established screening pathways (colonoscopy and low-dose CT) that are already reducing late-stage diagnoses. For these cancers, Galleri’s incremental contribution to preventing Stage III-IV disease is inherently smaller. But pancreatic, ovarian, liver, and esophageal cancer have no standard screening whatsoever. For these cancers, Galleri is the entire detection mechanism. When the endpoint combines all 12 types into a single metric, the cancers where Galleri has the most additive value get diluted by the cancers where existing screening already captures much of the late-stage prevention signal. If the endpoint had been restricted to cancers with no existing screening, the stage-shift signal may well have looked different.
The NYT Coverage Lacked Context
The New York Times article by Rebecca Robbins and Gina Kolata correctly reported that the trial missed its primary endpoint and accurately noted several important facts about Galleri’s regulatory and commercial status. But the framing was misleading in several ways.
The piece stated that diagnosing cancer sooner “does not always mean people live longer,” citing a 2024 JAMA article. This is technically accurate in the abstract, since lead-time bias and over-diagnosis are real phenomena in screening. But it is also a deeply incomplete framing when applied to cancers like pancreatic, ovarian, and liver cancer, where earlier detection is essentially the only lever that meaningfully changes survival. Pancreatic cancer detected at the localized stage has a five-year relative survival rate of approximately 44%. Detected at Stage IV, it drops to roughly 3%. The suggestion that earlier detection of these cancers might not improve outcomes is not supported by the oncology literature.
The article also raised concerns about false reassurance, that a negative Galleri result might cause patients to skip their mammogram or colonoscopy. This is worth examining empirically rather than treating as an assumed risk. The PATHFINDER 2 study explicitly measured whether Galleri testing affected adherence to standard-of-care screening through participant-reported outcomes, and no evidence of reduced adherence has been reported.
The Times also characterized Galleri as being sold through a “regulatory loophole,” referring to its availability as a laboratory-developed test (LDT) under CLIA certification without FDA premarket approval. This is technically accurate but misleadingly pejorative. The LDT pathway has been the standard regulatory framework for thousands of clinical laboratory tests for decades. Many of the genetic tests, companion diagnostics, and advanced biomarker panels used in precision medicine today entered clinical practice through this pathway.
Medicare Coverage Is Now Law, But Conditional
At $949 per test with almost no insurance coverage, Galleri has been functionally available only to those who can afford it. Cancer mortality is disproportionately concentrated in lower-income populations and communities with limited access to preventive care, precisely the populations least able to pay out of pocket for an uninsured screening.
On February 3, 2026, President Trump signed the Nancy Gardner Sewell Medicare Multi-Cancer Early Detection Screening Coverage Act into law as part of the Consolidated Appropriations Act of 2026. Named for the mother of Rep. Terri Sewell (D-AL) who died of pancreatic cancer in 2021, the law creates a pathway for Medicare to cover MCED tests beginning in 2028, once they receive FDA approval and CMS determines the coverage is appropriate. It does not automatically cover Galleri or any specific test.
Medicare coverage would remove the cost barrier for roughly 67 million beneficiaries and likely catalyze private insurer coverage decisions, since commercial payers frequently follow Medicare’s lead. FDA approval combined with Medicare coverage would transform Galleri from a self-pay screening available to the affluent into a standard preventive benefit available to the broader population.
But the missed primary endpoint complicates the timeline. TD Cowen analysts have characterized it as a major setback for both FDA approval and Medicare coverage. Grail is extending follow-up by 6-12 months, expecting full PATHFINDER 2 data later this year, and enrolling 50,000 participants in a third study, REACH, in collaboration with Medicare.
What This Means for an Individual
The population-level data can also be translated into individual terms. A 50-year-old has roughly a 35% chance of being diagnosed with cancer over the next 20 years. About 70-75% of those potential cancers are types with no standard screening, meaning the current detection strategy is to wait for symptoms to appear. For cancers like pancreatic, ovarian, liver, and stomach cancer, symptoms typically appear at a stage where five-year survival is in the single digits.
At $949 per year, 20 years of Galleri screening costs roughly $19,000. Galleri’s sensitivity for the 12 deadliest cancers is 73.7%, and for several of the unscreenable cancers it exceeds 80%. If Galleri catches one of these cancers at Stage I instead of Stage IV, the survival difference is large.
Source: SEER Cancer Statistics
Combining the probability of developing an unscreenable cancer (~10% over 20 years), the probability of Galleri detecting it (~78% weighted average for deadly cancers), and the probability that earlier detection meaningfully changes the outcome (~40-50%), annual Galleri screening gives a given individual roughly a 3-4% chance of altering what would otherwise be a fatal diagnosis. Whether that probability justifies $19,000 over two decades is a personal calculation, but for context, the annual probability of a house fire is roughly 0.25%, yet most people pay homeowner’s insurance premiums for decades without question, insuring against a risk substantially smaller than the one Galleri is designed to catch.
Where This Leaves Us
The NHS-Galleri trial did not meet its primary endpoint. This matters for regulatory and reimbursement decisions. But the trial also demonstrated a clinically meaningful reduction in Stage IV diagnoses over successive screening rounds, a four-fold increase in overall cancer detection, and increased early-stage detection of deadly cancers.
For cancers like pancreatic and ovarian cancer, where no screening exists and where stage at diagnosis is the primary determinant of survival, the relevant question is not whether Galleri is a perfect test. The relevant question is whether adding an imperfect test to a landscape of zero detection produces net clinical benefit. The emerging data strongly suggest that it does. The field needs longer follow-up, mortality data, and larger risk-stratified studies. These are coming.
And as the next generation of these tests learn to read not just DNA methylation but proteins, RNA, and other molecular signals in a single blood draw, and as those technologies continue to drop in cost, today’s version of Galleri is likely to look like the earliest chapter of a much longer story.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso