Keto or Ket-no for brain health?
My take on what we know from the scientific literature
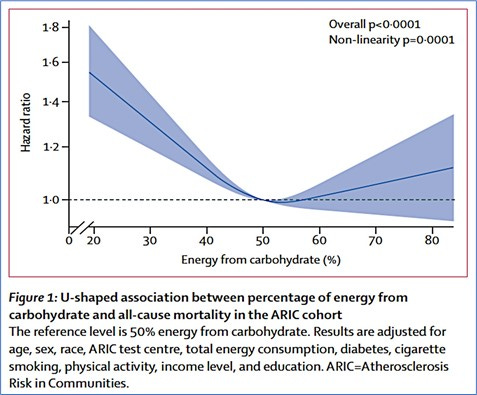
I have been experimenting with longer fasting windows and a low carb diet this Fall. Normally I fast until around 12pm everyday but have been mixing in some longer 24-36 hr fasts lately. My recent biomarker tests flagged high urine ketones (unsurprisingly given my 24hr fast), and worryingly, a substantial increase in some of my cardiovascular markers- ApoB (85→108), LDL, and CRP. This was despite optimal metabolic biomarkers- a fasting glucose of 69 and an HgbA1C of 5.3 and normal insulin levels.
Also, anecdotally, I feel bad when I’m in ketosis, foggy, lower mood, and lower energy so rising inflammatory and cardiovascular markers are consistent with my subjective experience. Other people experience ketosis more positively, with a clearer head and more energy. There are some genetic factors that may be in play that I get into later on in the article.
All of this led me to dig into whether Keto diet, which I was achieving by default through fasting and low carb diet, could be actually worsening my cardiovascular markers. I also dug into whether it is a net positive for dementia prevention given that it is the recommended diet by Dr. Dale Bredesen for his dementia patients.
Ketogenic interventions have exploded in popularity as potential treatments for cognitive decline, but the scientific evidence presents a complex picture that resists simple conclusions. After examining recent peer-reviewed research, including clinical trials, systematic reviews, and mechanistic studies, the data reveal some genuine promise but significant limitations that demand careful interpretation, especially surrounding cardiovascular risks with keto in healthy individuals that can negatively affect the brain.
A meta-analysis of 10 randomized control trials showed ketogenic interventions have modest, short-term cognitive benefits in mild cognitive impairment and early Alzheimer’s disease, with consistent improvements of 1-3 points on standardized cognitive tests. This level of improvement is questionably meaningful for quality of life or noticeable improvement in memory.
However, the evidence base suffers from small sample sizes, short durations, high dropout rates, and methodological limitations. No large-scale trials demonstrate long-term disease modification, and approximately one-third of studies show no benefit. The overall quality of evidence remains low to moderate, insufficient for broad clinical recommendations despite mechanistic plausibility.
The supporting evidence: What ketosis might offer the brain
Multiple biological mechanisms suggest ketones could benefit brain health, supported by both laboratory research and preliminary clinical data. The most compelling mechanistic evidence centers on ketone bodies, primarily β-hydroxybutyrate (βHB), serving as alternative brain fuel when glucose metabolism falters, a hallmark of Alzheimer’s disease.
PET imaging studies consistently demonstrate that brain ketone metabolism remains intact even as glucose utilization declines in AD progression. This preserved metabolic pathway could theoretically rescue energy-starved neurons. Ketones provide approximately 27% more ATP per molecule than glucose and bypass Complex I deficits in the mitochondrial electron transport chain, potentially enhancing cellular energy production. In 2024, a meta-analysis of 10 randomized controlled trials involving 691 AD patients found ketogenic diets improved cognition scores: Mini-Mental State Examination scores increased by 1.25 points (95% CI: 0.46-2.04, P=0.002) and ADAS-Cog scores improved by 3.43 points (95% CI: -5.98 to -0.88, P=0.008). Again, these are very small effect sizes that on average would not be noticeable.
Beyond energy metabolism, ketones have multiple mechanisms that are good for the brain. βHB acts as a signaling molecule that inhibits the NLRP3 inflammasome at physiologically achievable concentrations (0.5-7 mM), shifting microglia from pro-inflammatory to anti-inflammatory phenotypes. Studies from 2023-2024 show ketones increase brain-derived neurotrophic factor (BDNF) through histone acetylation and CREB pathway activation, potentially supporting neuronal survival and growth. Read more about this in my article “Exercise releases “plant food” for your brain”.
Additionally, βHB functions as an endogenous histone deacetylase inhibitor, suggesting epigenetic mechanisms that could influence gene expression patterns related to neuroprotection.
Animal models provide supportive mechanistic evidence. In an Alzheimer’s mouse model, 8-month ketone ester feeding reduced hippocampal amyloid-β and phosphorylated tau while improving cognitive performance. A 2025 study in an Alzheimer’s mouse model found medium-chain triglycerides reduced neuronal apoptosis, promoted axonal regeneration, and restored gut microbiome homeostasis. However, translation from rodent models to humans remains uncertain, particularly given that many promising Alzheimer’s treatments fail despite positive animal data.
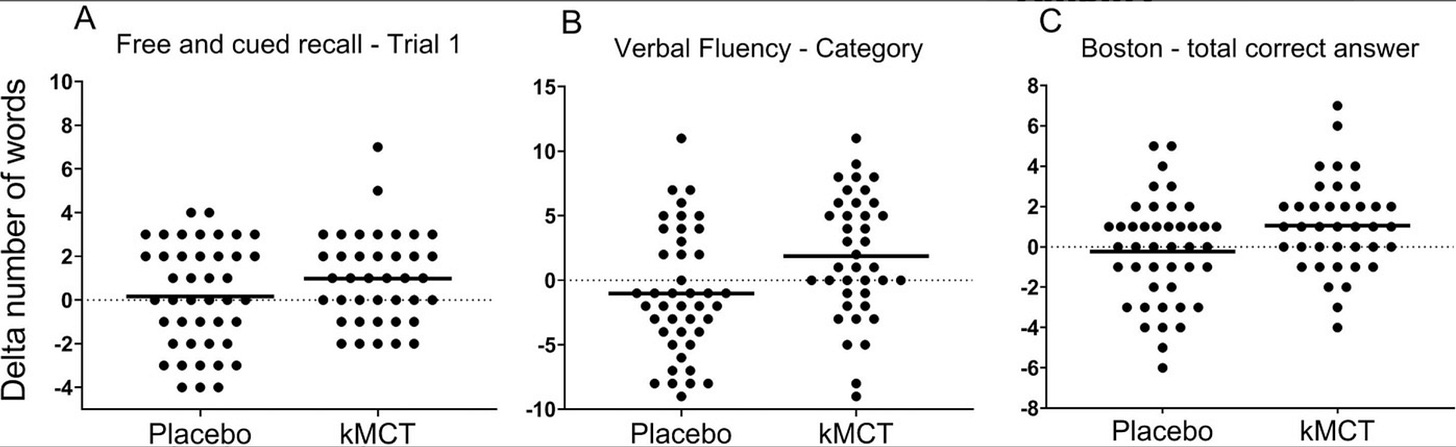
The clinical evidence shows differential effects across populations. The 2021 BENEFIC trial—among the highest-quality studies—tested ketogenic MCT drinks in 83 MCI patients over 6 months, finding significant improvements in free recall (P=0.047), verbal fluency (P=0.024), and the Boston Naming Test (P=0.033). The effect sizes were not very impressive (see figure below).
A 2022 Alberta trial followed 20 AD patients for 15 months with MCT oil, reporting that 80% stabilized or improved cognitively, with better outcomes in those maintaining longer MCT exposure and ages above 73 years. This was only statistically significant vs. placebo for one of the four cognitive assessments on the trial. Average MCT oil consumption was 1.8 tablespoons/day (25.2 g, 234 kcal). Thirty‐nine mild to moderate adverse events were reported in 17 (85%) subjects. Of these, 25 (64%) were GI and included vomiting, diarrhea, or abdominal cramping, felt to be related to the study oil.
Different ketogenic approaches show varying profiles. Dietary ketosis produces robust ketone elevation (0.6-0.95 mmol/L) but faces adherence challenges. MCT oil provides moderate ketone elevation with dose-dependent effects (optimal 25-42 g/day) but causes gastrointestinal distress in many users. Exogenous ketone supplements elevate ketones rapidly without dietary restriction, with a 2025 meta-analysis of 18 studies (875 participants) showing modest but significant cognitive benefits (standardized mean difference=0.26, 95% CI: 0.11-0.40, P=0.0007). Intermittent fasting represents another approach: a 2024 NIA-funded trial in 40 older adults with insulin resistance found 5:2 intermittent fasting improved executive function and reduced neuronal insulin resistance more effectively than a USDA healthy diet over 8 weeks.
The contradictory evidence: where ketosis falls short
The enthusiasm for ketogenic interventions must be tempered by substantial contradictory evidence and methodological concerns. Multiple well-designed studies report null results, and at least 3 of 11 human studies in a 2020 systematic review found no significant cognitive effects from ketogenic interventions.
In healthy populations, the evidence is particularly weak. A 2022 South African crossover trial in 11 healthy adults achieved sustained ketosis over 3 weeks but found no significant effects on cognitive performance, mood, or sleep quality compared to a high-carbohydrate diet. Five additional studies in healthy individuals from 2011-2019 similarly reported no cognitive improvements, with one finding a high-fat diet actually impaired both cardiac metabolism and cognitive function. This pattern suggests ketogenic interventions may benefit only metabolically compromised brains, not healthy ones.
More concerning, a 2024 case report in Neurology documented reversible memory loss, brain fog, and executive function deficits in a 48-year-old woman after 2 years of strict ketogenic diet. Formal neuropsychological testing revealed impairments in attention, anterograde memory, and task-switching that significantly improved within 2 months of discontinuing the diet. While a single case report has limited generalizability, it raises important safety questions about prolonged ketogenic diets that existing short-term trials cannot address.
Even studies showing positive results often reveal limitations upon closer inspection. The 2021 Phillips crossover trial in 26 AD patients achieved ketosis and improved quality of life and daily functioning (P<0.05) but found no significant improvement in the primary cognitive outcome (ACE-III score: P=0.24). The trial occurred during COVID-19 lockdowns, potentially introducing confounding from social isolation. Another 2020 study testing caprylic triglyceride formula (AC-1204) in mild-to-moderate AD found no ketone body increase and no cognitive improvements, representing a complete failure of the intervention.
The APOE4 genotype presents a critical complication. Multiple studies suggest APOE4 carriers—representing up to 40% of Alzheimer’s patients and the primary genetic risk factor—show reduced or absent cognitive benefits from ketogenic interventions. Two studies found benefits only in APOE4-negative patients, though three other underpowered studies found no genotype differences. This genetic heterogeneity could substantially limit the intervention’s clinical utility.
The cardiovascular risk question remains unresolved
Safety concerns extend beyond cognition. A 2024 meta-analysis of 27 RCTs with 1,278 participants found ketogenic diets significantly increased total cholesterol (+0.36 mmol/L, 95% CI: 0.15-0.57) and LDL cholesterol (+0.35 mmol/L, 95% CI: 0.20-0.50), with authors concluding the “impact on CVD risk factors warrants a cautious approach.” A 2024 review from the American College of Cardiology noted that very low-carbohydrate diets show worse cardiovascular mortality outcomes than moderate low-carb patterns, asserting ketogenic diets “do not fulfill the criteria of a healthy diet.”
The American College of Cardiology and National Lipid Association guidelines identify strict contraindications including hypertriglyceridemic pancreatitis, familial hypercholesterolemia, and caution that patients with atherosclerosis should avoid very low-carb diets liberalizing saturated fat. A 2019 ACC study linked carbohydrate intake below 44% to increased irregular heart rhythms, while a 2018 Lancet meta-analysis associated low-carb diets with early death from cardiovascular disease. For elderly individuals already at cardiovascular risk, these concerns cannot be dismissed lightly.
Inflammation as measured by CRP testing demonstrates that duration critically modulates outcomes. Studies shorter than 12 weeks show inconsistent results, with some documenting paradoxical CRP increases in the acute adaptation phase. A 2007 study found a 25% CRP increase in women after just 4 weeks on low-carb diets, while the high-carb comparison group experienced a 43% decrease. Only interventions exceeding 12.5 weeks consistently demonstrate anti-inflammatory benefits, suggesting a prolonged adaptation period before metabolic advantages manifest.
ApoB elevations are consistent and concerning in lean individuals.
Unlike the mixed CRP findings, ApoB responses show consistent directional effects that vary dramatically by baseline body composition. The pattern is striking: a 2023 meta-analysis by Joo et al. in normal-weight adults (BMI <25) found ApoB increased by 0.35 g/L (95% CI: 0.06-0.65), occurring alongside LDL-C increases of 1.08 mmol/L (42 mg/dL). Pediatric studies reveal even more pronounced effects: Kwiterowich et al.’s study of 141 children on therapeutic ketogenic diets for epilepsy documented a mean ApoB increase of +49 mg/dL at 6 months (p<0.001), with 53% developing elevated LDL-C and the ApoB/ApoA-I ratio increasing by 2 standard deviations.
Genetic variation explains substantial inter-individual heterogeneity in biomarker responses. ApoE genotype emerges as the most clinically relevant polymorphism. ApoE4 carriers (25% of the population) demonstrate heightened susceptibility to cholesterol hyperabsorption and may experience exaggerated LDL-C/ApoB increases. Peter Attia Case studies document ApoE4+ individuals developing severe dyslipidemia on high saturated fat ketogenic diets.
Polygenic variation affecting lipid metabolism, glycogen synthesis, and fat oxidation capacity modulates responses across populations. Validated genetic markers from clinical trials include LIPF rs814628-G (greater weight loss), GYS2 rs2306179-C (hepatic glycogen synthesis, greater weight loss), and CETP rs5883-T (cholesteryl ester transfer, greater weight loss response).
A Norwegian RCT in normal-weight young adults documented LDL-C increases averaging 44% but ranging from 5% to 107%—an “unpredictable individual response” that prompted researchers to recommend mandatory lipid monitoring. A Swedish study of 17 healthy young women found LDL-C increased in every single participant by 1.82 mM (p<0.001), accompanied by ApoB increases and elevations in both small dense and large buoyant LDL particles, leading investigators to conclude the diet produced a “deleterious blood lipid profile.”
The clinical significance of ketogenic diet-induced biomarker changes represents the field’s most contentious debate. Traditional lipidology provides clear evidence: elevated ApoB causally increases cardiovascular disease risk, with statin trials demonstrating proportional risk reduction with ApoB lowering. The UK Biobank study offers concerning observational data: 305 individuals on low-carb high-fat diets followed for 11.8 years showed ApoB levels of 1.09 vs 1.04 g/L in controls (p<0.001), with a major adverse cardiovascular event (MACE) rate of 9.8% vs 4.3% (adjusted HR: 2.18; 95% CI: 1.39-3.43; p<0.001). Those with LDL-C ≥5 mmol/L on LCHF diets exhibited a 6.68-fold increased risk compared to standard diet with LDL <3.5 mmol/L.
Adherence emerges as a major limitation of ketogenic interventions
Perhaps the most underappreciated limitation is poor long-term adherence. A 2024 systematic review tracking ketogenic diet adherence found compliance drops dramatically over time: 79.7% at 6 months, 66.7% at 24 months, and just 37.7% at 36 months. Dropout reasons include diet restrictiveness, difficulty preparing meals, cost, adverse effects, food refusal, and lack of motivation.
In the Henderson 2020 MCT trial, 23% of intervention participants discontinued treatment, primarily due to gastrointestinal symptoms affecting 49% of users. The BENEFIC trial reported 25% dropout, mostly from intolerance to the MCT drink. A modified Atkins diet study showed 33.3% dropout from poor compliance, while classic ketogenic diet studies report 60% dropout due to psychosocial factors, fatigue, and weight loss. If an intervention cannot be sustained beyond months, its clinical value is profoundly limited regardless of short-term efficacy.
Different ketogenic approaches show distinct tradeoffs
The field encompasses multiple intervention types with varying profiles. Classical ketogenic diets (90% fat) produce the highest ketosis but face the worst adherence challenges and require medical supervision. Modified Atkins diets (65% fat) offer better tolerability while maintaining moderate ketosis. Mediterranean-ketogenic hybrids attempt to preserve cardiovascular benefits while inducing ketosis, with a 2024 study showing favorable lipid profile changes inversely correlated with AD biomarkers.
MCT oil supplementation bypasses dietary restriction by rapidly converting to ketones in the liver. The longest MCT study (15 months) found 80% of AD patients stabilized or improved, with dose-dependent effects optimal at 25-42 g/daily. However, gastrointestinal side effects remain the limiting factor, including diarrhea, nausea, and abdominal discomfort that improve with gradual dose escalation but never fully resolve in many users.
Exogenous ketone esters and salts represent the newest approach, producing ketosis within 30-60 minutes without dietary change. A 2025 meta-analysis found modest but consistent cognitive benefits (SMD=0.26), with no differences between acute and intermediate durations or between healthy and AD populations. Ketone esters achieve higher blood ketone concentrations (2-5 mmol/L) than other methods but face challenges with taste, cost, and gastrointestinal tolerance.
Intermittent fasting induces ketosis through caloric restriction rather than macronutrient manipulation. A 2024 NIA trial found 5:2 intermittent fasting (restricting to 25% of recommended intake 2 days weekly) improved executive function and reduced neuronal insulin resistance in older adults with metabolic dysfunction. A 2020 Malaysian observational study of 99 elderly individuals with MCI found regular intermittent fasting associated with 24.3% reversion to successful aging versus 3.7% in non-fasters over 36 months, though observational design limits causal inference.
The evidence quality falls short of clinical standards
Systematic assessment reveals the evidence base suffers from fundamental methodological limitations. The most comprehensive quality assessment (Grammatikopoulou et al. 2020, Advances in Nutrition) evaluated 10 RCTs using Cochrane Risk of Bias tools and Oxford quality scores, finding most studies showed “unclear bias” across multiple domains with low Oxford scores. Sample sizes ranged from 6 to 152 participants with a mean of just 31 participants per study. Study durations averaged only 11 weeks, with no consistent follow-up beyond interventions.
No Cochrane reviews exist specifically for ketogenic interventions in dementia. No position statements from the Alzheimer’s Association or American Academy of Neurology endorse these approaches. The evidence quality for cognitive outcomes rates as low to moderate using GRADE-like criteria, downgraded for risk of bias, inconsistency, indirectness (short-term studies for chronic disease), imprecision from small samples, and likely publication bias favoring positive results.
A 2024 meta-epidemiological review of ketogenic diet studies found that of publications from 1946-2024, none met predefined inclusion criteria for methodological rigor. Only 7 articles came close, with reviewers concluding “even though some studies show positive results, we cannot consider them applicable to general population” due to small samples, short duration, and high dropout.
The heterogeneity across studies prevents definitive conclusions. Different macronutrient ratios, ketone thresholds, fat types, caloric restrictions, and cognitive assessment tools create an evidence base that resists synthesis. A 2024 review noted most of 28 meta-analyses on ketogenic diets failed to define essential parameters including calories, macronutrient ratios, types of fatty acids, and ketone body measurements, concluding studies should adopt “a single definition” to enable meaningful comparisons.
Specific cognitive domains show differential evidence strength
Not all cognitive outcomes respond equally to ketogenic interventions. General cognition measured by ADAS-Cog shows the most consistent improvements across studies, representing the strongest evidence. Episodic and secondary memory show moderate-to-strong evidence of benefit, particularly with longer interventions (6 months). Processing speed and attention demonstrate time-dependent effects, improving primarily in studies exceeding 6 months.
Conversely, executive function tests (Trail Making Test, Digit Symbol Substitution) show minimal to no improvement across studies, representing a notable gap given executive dysfunction’s prominence in dementia. MMSE scores show less sensitivity to ketogenic interventions than ADAS-Cog, possibly due to ceiling effects or test characteristics. Mood and psychological outcomes (Geriatric Depression Scale, Profile of Mood States) show no improvements in available studies.
Quality of life and activities of daily living represent arguably more clinically meaningful outcomes than cognitive test scores. The Phillips 2021 trial found significant improvements in both QOL-AD (+3.37 points, P=0.023) and ADCS-ADL (+3.13 points, P=0.0067) despite non-significant cognitive changes, suggesting functional benefits may exceed pure cognitive effects. This pattern appears consistently across ketogenic diet trials, raising important questions about outcome selection in future research.
Biomarkers provide mechanistic insights but limited prognostic value
Biomarker data support plausible mechanisms while highlighting critical gaps. Blood ketone elevations achieve statistical and clinical significance across all intervention types, with β-hydroxybutyrate concentrations positively correlating with cognitive improvements (r=0.62 in some studies). PET imaging with 11C-acetoacetate consistently demonstrates increased brain ketone uptake, with CMRKetones increasing 230% in the BENEFIC trial despite preserved glucose metabolism.
However, downstream disease biomarkers show limited evidence. Only one small study examined CSF tau and amyloid-β, with unclear results. A 2024 Wake Forest trial of modified Mediterranean-ketogenic diet found CSF changes (increased Aβ42, decreased tau) suggestive of reduced AD pathology, but sample sizes preclude definitive conclusions. No studies track disease progression biomarkers beyond 6 months, leaving the critical question of disease modification versus symptomatic benefit entirely unanswered.
Inflammatory markers show more consistent patterns. Studies from 2023-2024 report reduced C-reactive protein, IL-6, and increased anti-inflammatory cytokines with ketogenic interventions. Oxidative stress markers improve, with increased superoxide dismutase activity and glutathione levels. A 2025 study found gut microbiome changes including increased Akkermansia muciniphila and short-chain fatty acids associated with reduced systemic inflammation. These changes suggest plausible biological activity, though their relationship to clinical outcomes remains correlative rather than causal.
The weight loss confound clouds interpretation
A critical methodological issue pervades the literature: most ketogenic intervention studies report significant weight loss, introducing a major confounding variable. Krikorian et al. documented 4 kg mean weight loss after 6 weeks in MCI adults. Four of five AD mouse model studies showed significant weight loss alongside cognitive benefits.
Weight loss independently improves cognitive function through multiple mechanisms including reduced inflammation, improved insulin sensitivity, and cardiovascular benefits. Parsing ketosis-specific effects from caloric restriction effects becomes nearly impossible in studies lacking weight-matched control groups. This confound particularly affects interpretation of longer-duration studies where cumulative weight changes become substantial.
For frail elderly AD patients already at nutritional risk, weight loss presents a genuine safety concern. Multiple reviews identify potential malnutrition as a limitation requiring careful monitoring. The Phillips trial reported 2.62 kg average weight loss over 12 weeks. Whether such weight loss represents healthy metabolic adaptation or concerning nutritional compromise depends on individual patient context, yet few studies adequately assess nutritional status, body composition, or functional decline related to weight changes.
Critical research gaps prevent definitive conclusions
The field suffers from glaring evidence gaps that preclude strong clinical recommendations. No large-scale phase 3 trials exist—the largest published trial enrolled 152 participants, far below the hundreds or thousands typical for drug approval. No studies extend beyond 15 months, insufficient for a chronic progressive disease where disease-modifying effects require years to assess. The meta-analysis evidence, while suggestive, synthesizes small heterogeneous studies rather than large definitive trials.
Disease modification remains entirely speculative. No adequately powered studies measure amyloid or tau PET imaging longitudinally, CSF biomarkers serially, or clinical progression against validated criteria for disease modification. The existing evidence demonstrates only symptomatic benefit lasting weeks to months, with effects waning after intervention cessation. A 2021 meta-analysis noted benefits evident in the short term “after 12 months, benefits began to fade,” suggesting transient rather than sustained effects.
Population diversity limitations reduce generalizability. Most studies enroll predominantly Caucasian participants with limited ethnic representation. APOE4 carriers are often systematically excluded despite representing 40% of AD patients. Few studies examine effects across dementia subtypes—vascular dementia, Lewy body dementia, and frontotemporal dementia remain essentially unstudied.
Optimal intervention parameters remain undefined. No head-to-head trials compare ketogenic diet versus MCT versus exogenous ketones versus intermittent fasting. Dose-response relationships receive inadequate attention. The minimal effective ketone concentration, optimal intervention duration, and best timing in disease course lack evidence.
The mechanistic picture suggests more than simple energy rescue
While energy deficit rescue represents the most intuitive mechanism, the mechanistic data suggest ketones act through multiple pathways simultaneously. The 2024 Buck Institute study in animal models identified direct βHB effects on protein solubility and misfolded protein clearance via autophagic flux modulation, representing a distinct mechanism from energy metabolism. HDAC inhibition creates epigenetic changes affecting gene expression programs related to stress resistance and neuroprotection.
The gut-brain axis emerges as an unexpected mechanistic player. A 2025 study found MCT intervention in Alzheimer’s mice increased beneficial gut bacteria (Akkermansia), reduced intestinal permeability, elevated brain short-chain fatty acid concentrations, and ameliorated amyloid deposition. Whether gut microbiome changes mediate cognitive effects in humans remains speculative but biologically plausible given emerging connections between gut health and neurodegeneration.
These multiple mechanisms could explain why simple glucose rescue doesn’t fully account for observed effects. βHB functions simultaneously as energy substrate, signaling molecule, histone modifier, inflammasome inhibitor, and mitochondrial enhancer. This complexity makes identifying primary versus secondary mechanisms challenging but suggests genuine biological activity beyond placebo effects.
However, mechanistic plausibility doesn’t guarantee clinical efficacy. Alzheimer’s research is littered with interventions demonstrating compelling mechanisms in cell culture and animal models that fail in human trials. The mechanistic evidence for ketogenic interventions surpasses that available for many failed Alzheimer’s drugs, but this comparison provides little comfort given the field’s abysmal translation record.
Where the evidence actually supports cautious implementation
Despite substantial limitations, the evidence supports several provisional conclusions. In mild cognitive impairment and early Alzheimer’s disease, ketogenic interventions produce modest but statistically significant short-term cognitive improvements, particularly in memory and general cognition. Effect sizes (1-3 points on standardized tests, SMD=0.26 for exogenous ketones) fall below those of FDA-approved Alzheimer’s medications but remain clinically meaningful for some patients.
The interventions appear generally safe over 3-6 months with manageable side effects in most individuals. Serious adverse events are rare in published trials. Gastrointestinal symptoms represent the primary tolerability issue, managed through gradual introduction and dose adjustment. Cardiovascular effects require monitoring but appear acceptable in metabolically healthy individuals without contraindications.
Functional outcomes (quality of life, activities of daily living) may improve more consistently than pure cognitive scores, suggesting patient-centered benefits that cognitive tests might not fully capture. For individuals and families seeking non-pharmacological approaches to slow cognitive decline, ketogenic interventions represent a reasonable experimental approach with appropriate medical supervision, particularly for those with concurrent metabolic dysfunction like insulin resistance or type 2 diabetes.
Intermittent fasting emerges as potentially the most promising approach based on the 2024 NIA trial results and superior adherence compared to continuous dietary restriction. The 5:2 protocol allows normal eating 5 days weekly, dramatically improving sustainability. Benefits extend beyond ketosis to include autophagy induction, insulin sensitivity improvement, and inflammatory modulation.
Conclusion: premature for broad implementation and inappropriate for cognitively normal lean individuals and those with APOE4 alleles
The evidence landscape for ketosis and brain health shows some promising preliminary data that is insufficient for definitive conclusions. The biological mechanisms are plausible and increasingly well-characterized. Small trials show consistent direction of effect, even if effect sizes remain modest. Safety appears acceptable in appropriately selected patients. Yet fundamental evidence gaps including small samples, short duration, high dropout, absent disease modification data, and no large phase 3 trials prevent the scientific community from endorsing ketogenic interventions as standard dementia treatment or prevention.
The field requires what it has lacked: large-scale, long-term randomized controlled trials with 500+ participants followed for 2+ years, measuring both cognitive outcomes and disease biomarkers, stratified by APOE genotype, with rigorous adherence support and monitoring. Several such trials are underway, including the $3.5 million NIH-funded TAKEOFF trial testing ketone esters in 180 older adults and ongoing Paris trials examining brain metabolism via PET over 12 months in 70 early AD patients.
For motivated individuals with early cognitive decline, concurrent metabolic dysfunction, and no contraindications (APOE4), supervised trials of ketogenic approaches represent reasonable experimental interventions. For the general population seeking dementia prevention, the evidence remains too preliminary to recommend over established approaches like Mediterranean diet, exercise, cognitive engagement, and cardiovascular risk management.
In general the evidence does not support ketogenic approaches as superior to Mediterranean or other evidence-based dietary patterns for cardiovascular health or longevity. Mediterranean diets offer equivalent glycemic benefits with superior lipid profiles, nutrient adequacy, sustainability, and robust long-term cardiovascular outcomes evidence. For brain-focused applications, ketogenic diets should be considered adjunctive tools requiring precision implementation with comprehensive biomarker monitoring, genetic risk stratification, Mediterranean-style fat quality, and explicit informed consent regarding the absence of long-term safety data on hard cardiovascular endpoints.
With my APOE4 allele and poor subjective and biomarker response to keto diet, I will be sticking with the Mediterranean-style diet of my Sicilian ancestors from here on out. It has rock solid evidence for longevity and brain health and is delicious.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso