HRV, longevity, and brain health
What HRV is, how much it matters, and how to improve it
Many people reading this have been made aware of their heart rate variability (HRV), as I have been, when they started using a wearable device. My HRV is steadily improving, but is not great. I have been wondering for a while what this really means for my brain health and if it could be confounded by other things like my sex, genetics, etc. It seems that HRV is something to pay attention to and it may mean I need to prioritize lowering my stress levels even more. In this article I talk about what HRV is, the evidence linking it to disease, how to improve it, and what’s on the horizon in terms of new technology.
Your heart is supposed to be irregular
Most people assume a healthy heart beats like a metronome, but a healthy heart varies the time between beats from moment to moment, and that variation reflects how well your body is regulating itself.
Heart rate variability, or HRV, measures how much that variation exists, which is a different quantity from heart rate itself. Heart rate tells you how fast your heart beats. HRV tells you how consistently or inconsistently it spaces those beats.
Two systems are always competing for control of your heart
Your heart receives constant signals from two branches of the autonomic nervous system, the part of your nervous system that governs heart rate, breathing, digestion, and immune responses without any conscious input from you. The sympathetic branch acts as an accelerator, speeding the heart, mobilizing energy, and evolved to handle real physical threats. The parasympathetic branch, whose main channel to the heart is the vagus nerve, acts as a brake, slowing the heart and governing rest, recovery, digestion, and inflammatory control.
The sinoatrial node, the cluster of cells in the right atrium that sets your heart’s rhythm, receives both signals simultaneously. Sympathetic nerve fibers release norepinephrine, a hormone that accelerates heart rate, onto it to speed firing; vagal fibers release acetylcholine to slow it. At rest in a healthy person, the vagus is dominant and actively pressing the brake on every beat.
The beat-to-beat variation that HRV measures comes almost entirely from moment-to-moment fluctuation in how strongly the vagus presses that brake. Breathing is the largest driver, as inhaling stretches receptors that briefly reduce vagal outflow, releasing the brake so the heart speeds up a few beats, while exhaling restores vagal firing and the heart slows again. This cycling happens twelve to twenty times per minute and is what creates the rhythmic oscillation visible in HRV data.
Low HRV means the vagal signal has become weak, constant, or both. When vagal tone drops, beat intervals become more uniform because the sinoatrial node is receiving less dynamically fluctuating parasympathetic input. The underlying causes act at different points in the chain: chronic stress and poor sleep suppress vagal output at the brainstem level; inflammation degrades vagal signaling along the nerve fiber; aging and diabetes damage autonomic nerve fibers directly and reduce the sinoatrial node’s sensitivity to acetylcholine. Each mechanism reduces the dynamic, variable parasympathetic input the sinoatrial node receives.
Why this matters beyond cardiovascular health
The vagus nerve extends through the chest and into the abdomen, connecting to the gut, liver, spleen, and lungs. It also suppresses inflammation. When it fires, it releases acetylcholine at nerve endings in the spleen, which binds to receptors on macrophages and signals them to reduce pro-inflammatory output including a set of inflammatory proteins called TNF-alpha, IL-6, and IL-1. Researchers call this the cholinergic anti-inflammatory pathway. A series of animal experiments established that vagus nerve stimulation significantly reduces inflammatory protein release and improves survival in models of sepsis and other inflammatory conditions.
When vagal tone declines, this brake weakens and the result is a low-grade chronic inflammatory state that does not resolve the way acute inflammation from an infection does, providing one pathway through which autonomic dysfunction is linked not just to heart disease, but to metabolic disease, neurodegeneration, and accelerated aging. HRV, as a non-invasive index of vagal tone, is therefore a readout of this entire system.
There is also a more direct relationship between neurodegeneration and the vagus nerve itself. In Parkinson’s disease, the misfolded protein that defines the disease pathologically has been found in the brainstem nuclei that generate vagal outflow, and in the nerve network lining the gut, often before it appears in the brain regions responsible for motor symptoms. This has led to the hypothesis that Parkinson’s may originate in the gut and propagate toward the brain partly along the vagus nerve. Consistent with this, a large Danish cohort study found that people who had undergone surgical cutting of the vagus nerve for peptic ulcer disease decades earlier had a modestly lower incidence of Parkinson’s disease over long follow-up, as though severing the nerve had interrupted a route of transmission. The evidence is not definitive and the comparison groups differ in important ways, but it points toward the vagus nerve as something more than a passive readout of brain health.
In Alzheimer’s disease, the cholinergic neurons that are among the first to be lost are part of the same broader acetylcholine system as the vagal fibers that suppress peripheral inflammation, which means that declining vagal tone in aging may partly reflect the same underlying vulnerability rather than being a separate process. This matters for interpreting HRV in midlife. A declining baseline could reflect chronic stress, poor sleep, or metabolic dysfunction suppressing vagal output from a structurally intact system, or it could reflect early subclinical neurodegeneration affecting the vagal circuits themselves. Distinguishing those two things from a wearable reading is not currently possible, which is one reason the causal question between HRV and dementia risk remains unresolved.
What the prospective data show
The evidence that HRV predicts disease outcomes is among the most replicated findings in cardiovascular medicine. A meta-analysis of cohort studies found that lower HRV was associated with roughly double the risk of all-cause mortality and roughly 40% higher risk of cardiovascular events. A UK Biobank analysis of over 51,000 participants followed for a median of 11.5 years found that reduced HRV was independently associated with increased long-term risk of atrial fibrillation, stroke, and all-cause mortality, with effect sizes comparable to diabetes, hypertension, and smoking.
The relationship extends into metabolic disease. Research from the Maastricht Study found a graded decline in HRV across the spectrum from normal glucose metabolism to prediabetes to type 2 diabetes, with autonomic dysfunction detectable before the clinical diagnosis. The Rotterdam Study following over 7,600 participants found that declining HRV predicted developing type 2 diabetes, particularly in younger individuals, suggesting autonomic dysfunction precedes metabolic deterioration. HRV is also inversely associated with circulating inflammatory markers. In the MIDUS II study, higher HRV was independently associated with lower levels of markers of inflammation including CRP, fibrinogen, and IL-6, consistent with the vagal anti-inflammatory mechanism above.
The evidence linking HRV to brain aging and dementia risk is accumulating. A longitudinal analysis of 2,702 middle-aged adults from the Whitehall II cohort found that low RMSSD at midlife was associated with cognitive decline progressing roughly 3 years faster per decade, with participants in the lowest quintile having 37% higher odds of low cognitive function at follow-up. These associations held after adjusting for cardiovascular risk factors, lifestyle, and medication, and the sample was free of coronary disease and stroke at baseline, which strengthens the case that the HRV signal is not simply secondary to established vascular disease. A meta-analysis of 24 studies found that dementia patients had significantly lower parasympathetic HRV than healthy controls (Hedges’ g = −0.36), with the largest reduction in Lewy body dementia, suggesting different dementia subtypes may leave distinct autonomic fingerprints. The Framingham Offspring Cohort found that lower RMSSD predicted developing dementia over ten years of follow-up in adults over 60, consistent with the idea that declining vagal tone is detectable before clinical diagnosis. The central interpretive challenge across this literature is that neurodegeneration affects brainstem autonomic centers early in the disease course, meaning reduced HRV could partly reflect early subclinical pathology rather than being an independent upstream risk factor. No Mendelian randomization studies of the HRV-dementia relationship have yet been published, so the causal direction has not been formally tested with genetic instruments.
Researchers have directly tested whether low HRV causes disease or reflects it
Observational associations cannot rule out confounding, where a third factor such as physical inactivity independently causes both low HRV and disease, or reverse causation, where disease suppresses vagal tone rather than the other way around. These distinctions matter for whether HRV is worth targeting therapeutically.
The most rigorous approach is Mendelian randomization, which uses inherited genetic variants as instruments to approximate a randomized experiment. A 2024 Mendelian randomization study found that peak-valley respiratory sinus arrhythmia, a vagally mediated HRV index, showed a genetically supported causal relationship with cardiac arrest. For metabolic disease the picture is more complicated, as the Rotterdam Study found declining HRV predicted incident type 2 diabetes longitudinally, but bidirectional Mendelian randomization found no significant causal relationship in either direction, suggesting the association reflects shared underlying pathology rather than a causal chain.
The clearest causal evidence comes from animal experiments. When the vagus nerve is severed in rodents, the protective suppression of cytokine release during sepsis is abolished and mortality rises. When VNS is administered after lethal endotoxin exposure, cytokine levels fall and survival improves. These controlled interventional experiments establish that vagal nerve signaling is an active regulator of inflammatory outcomes. The field’s current consensus, per a 2025 comprehensive review, is that most HRV-disease evidence remains associative and that formal causal inference through larger genetic studies and adequately dosed interventional trials is the critical next step.
How to interpret your own number
HRV is measured in milliseconds using a metric called RMSSD (root mean square of successive differences), which is what most consumer wearables including Oura, Apple Watch, Garmin, and WHOOP report. Wearable optical sensors typically read 6 to 11% lower than ECG-derived values, so your true value is modestly higher than what the device shows.
Population percentiles provide context but a single reading is nearly meaningless, since what matters is your own stable baseline and whether it is trending over time. There are also legitimate reasons a healthy person runs below average, including genetic constitution, higher resting heart rate, sleep quality, psychological stress, and current training load.
One important limitation of consumer HRV tracking is that it captures a snapshot, often a single overnight reading, under conditions that are not standardized. Visit-to-visit blood pressure variability, which measures how much your blood pressure fluctuates across multiple clinical readings over months, may actually be a more consistent predictor of dementia risk in real-world settings. A retrospective cohort study of 48,204 adults found that clinically derived blood pressure variability predicted incident dementia independently, while HRV alone did not show a consistent association in the same dataset. When both were high-risk (high blood pressure variability and low HRV combined), dementia risk was more than double. The likely reason is that HRV measured opportunistically in clinical data is noisy, sensitive to posture, anxiety, recent activity, and the circumstances of the recording, whereas blood pressure variability aggregated across multiple visits captures something different and more stable, reflecting the brain’s ability to maintain consistent perfusion over time and the cumulative stress placed on cerebrovascular autoregulation. The practical implication is that HRV and blood pressure variability are measuring partly different things and are additive. If you track HRV, it is also worth asking your doctor about your blood pressure variability across visits, particularly if you have a family history of dementia or are an APOE4 carrier.
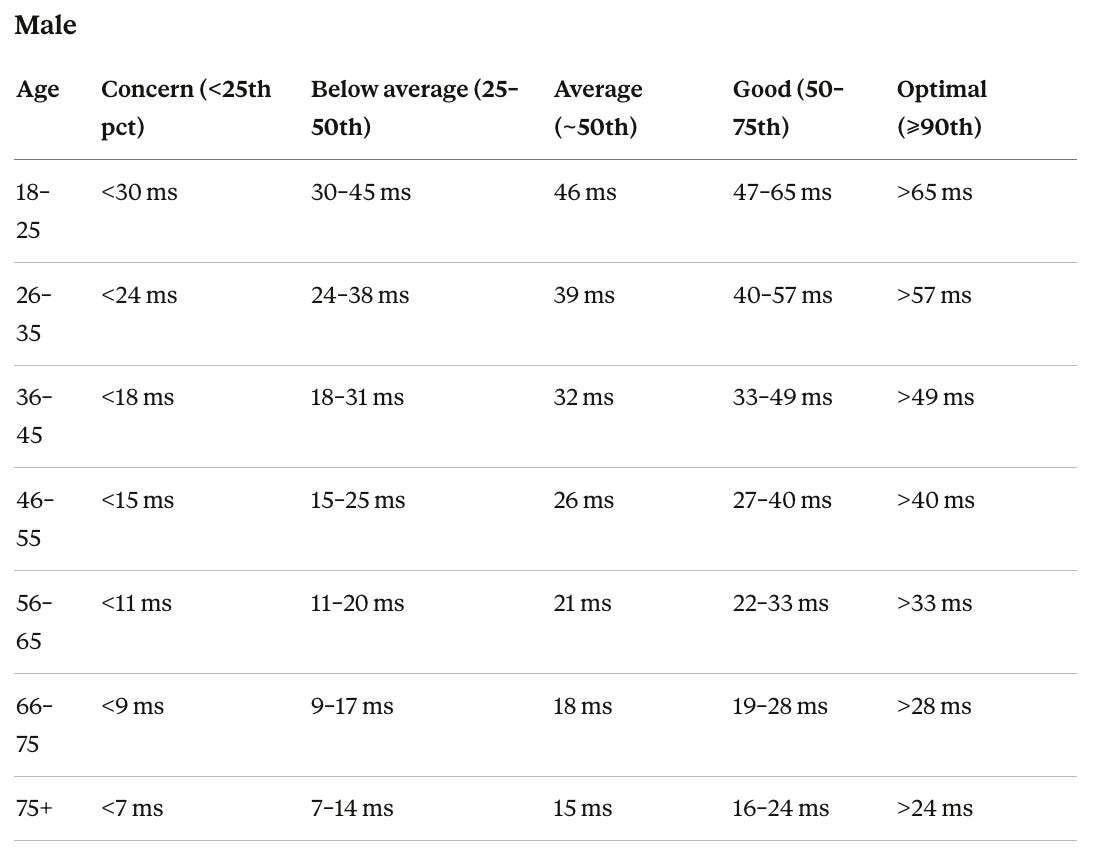
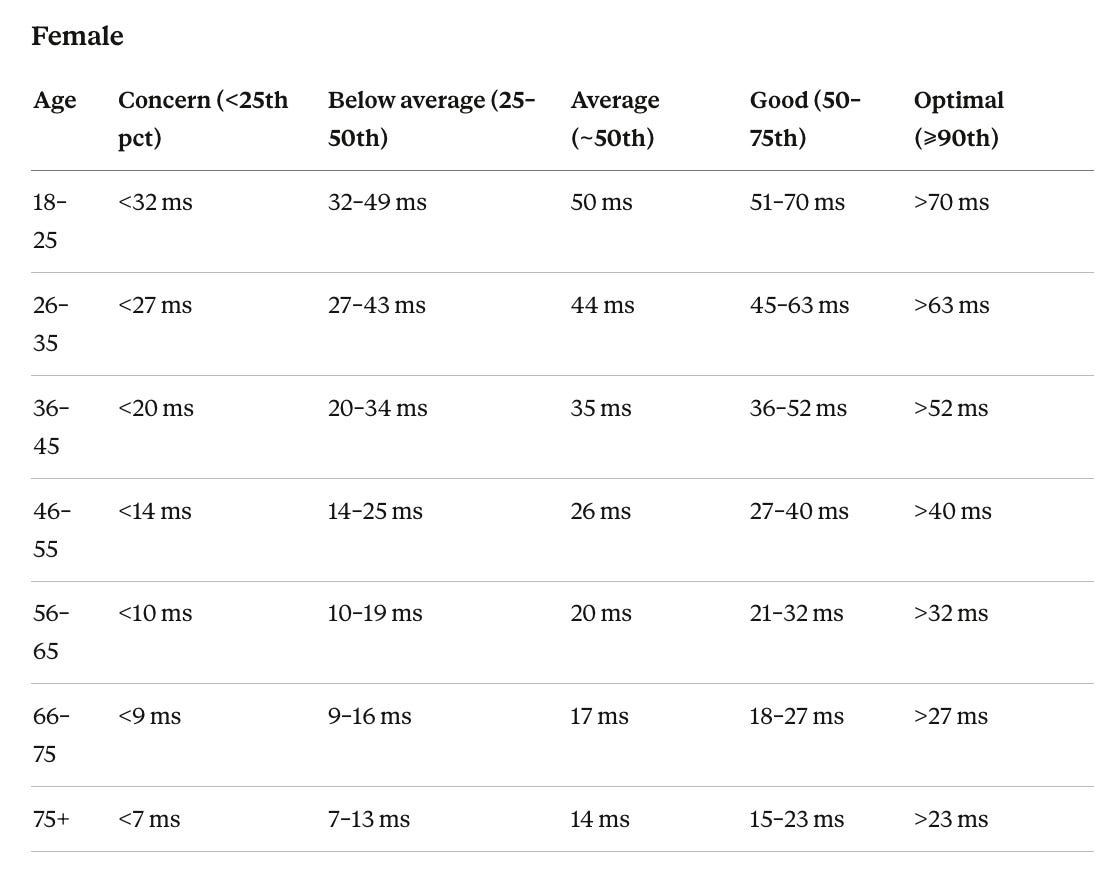
The benchmarks below are drawn from published population studies including the Lifelines Cohort Study and the Baependi Heart Study. Women in reproductive years run slightly higher RMSSD than men of the same age, driven by estrogen’s enhancing effect on vagal tone. This advantage narrows after menopause and largely converges above age 60.
HRV Reference Ranges by Age and Sex
The concern threshold is the 25th percentile for that sex and age group. Persistently below the 10th percentile, or a sustained drop of 20% or more from your own 30-day rolling average, is the pattern most clinical literature uses to flag something worth investigating.
What the randomized trial evidence shows
Aerobic exercise training has the deepest RCT evidence of any intervention. A meta-analysis of 16 RCTs in healthy adults found exercise improved RMSSD with a standardized mean difference of 0.84 (p=0.0005). A larger meta-analysis of 34 RCTs found effects were more pronounced in people with existing health conditions and in interventions lasting at least eight weeks. The mechanism goes beyond fitness, as endurance training enhances baroreflex sensitivity, the responsiveness of the system that adjusts heart rate to match blood pressure changes, and produces structural adaptations in the brainstem circuits that generate vagal outflow. Zone 2 training, a conversational pace for 30 to 60 minutes four or more times per week, produces the most consistent chronic gains. Effects are larger in people with low baseline vagal tone. For a deeper look at exercise thresholds for brain health, see my pieces on VO2 max and dementia prevention, exercise for cognitive sharpness, and how much muscle you need for longevity.
Slow-paced breathing and HRV biofeedback has the strongest acute evidence of any intervention. A meta-analysis of 223 studies found significant increases in vagally mediated HRV during sessions, immediately after, and after multi-session interventions. Breathing at 5 to 7 breaths per minute drives physiological resonance between respiration and the body’s blood pressure regulation system, producing the largest acute RMSSD elevation of any short intervention. A four-week RCT confirmed HRV improvements after 20 minutes of daily resonance breathing. A three-month RCT in 43 pre-hypertensive adults found HRV biofeedback reduced blood pressure significantly while deep breathing without feedback and no treatment did not.
Sleep quality and architecture has strong indirect RCT evidence. A meta-analysis of 11 randomized trials found experimentally induced sleep deprivation significantly reduced RMSSD. In a 12-week exercise RCT in poor sleepers, HRV improvements were partially mediated by sleep quality gains, suggesting exercise partly raises HRV by restoring sleep architecture. The parasympathetic system dominates during slow-wave sleep (the deepest, most physically restorative stage) and early REM sleep, the period when vagal tone is actively restored each night, and sleep apnea fragments precisely these stages. For a broader discussion of sleep and brain health see my articles on sleep and the brain and the glymphatic system.
Alcohol reduction is among the highest-yield changes based on observational and mechanistic evidence. Even moderate habitual consumption suppresses RMSSD through the following night, scaling with the amount consumed, through sympathetic nervous system activation and disrupted deep sleep. People who begin tracking HRV often notice this before anything else. No well-powered RCT specifically tests alcohol reduction as an HRV intervention, and the gap reflects practical challenges in trial design rather than ambiguity about the effect. For a comprehensive look at alcohol’s effects on the brain see my piece on alcohol, brain health, and longevity.
Chronic psychological stress reduction matters through several converging mechanisms. Sustained cortisol elevation directly suppresses the brainstem nuclei that generate vagal outflow. Chronic stress also amplifies activity in the amygdala while degrading the prefrontal cortex’s inhibitory control over it, and people with stronger prefrontal-amygdala connectivity consistently show higher resting HRV, making this top-down regulation a primary determinant of vagal tone. A third loop runs through inflammation. Chronic stress elevates IL-6 and TNF-alpha, which act back on the brain to further suppress vagal output. Longitudinal data from the Whitehall II cohort found job strain was associated with lower HRV independent of cardiovascular risk factors. Post-traumatic stress disorder produces some of the most severe HRV suppression outside of frank cardiac disease. A UK Biobank analysis found loneliness was among the strongest psychosocial predictors of low HRV, with effects comparable in size to moderate physical inactivity. Cognitive behavioral therapy for anxiety and PTSD consistently improves HRV alongside symptom reduction in randomized trials, suggesting the autonomic improvement follows restored prefrontal-vagal regulation.
Cold water immersion has solid acute evidence. A meta-analysis of 24 studies found significant RMSSD increases (SMD 0.61, p<0.001) persisting up to 15 minutes post-immersion, and a systematic review of 12 RCTs in post-exercise recovery found all 12 reported parasympathetic reactivation. Cold contact with the face activates facial nerve endings that trigger the diving reflex; hydrostatic pressure activates pressure-sensitive receptors in blood vessel walls that increase vagal outflow. Most evidence concerns the acute post-exercise recovery window rather than chronic resting HRV improvement.
Omega-3 supplementation has sufficient trial data to be credibly evidence-based above 1 gram per day. An ancillary VITAL trial analysis in 911 participants found 1g per day of marine omega-3 significantly increased RMSSD versus placebo over two years (p=0.040). A crossover RCT found 3.4g per day increased RMSSD by 9.9% versus placebo over eight weeks. A fish oil meta-analysis found significant increases in high-frequency HRV power, a frequency-based measure of vagal activity (SMD 0.30, p=0.005). Effects are dose-dependent and more pronounced in people with higher cardiovascular risk.
Sauna use has observational associations with improved HRV and plausible mechanisms involving improved blood pressure regulation and heat shock proteins, but no meta-analysis of RCTs powered for HRV outcomes yet exists.
Electrical stimulation of the vagus nerve is the most direct test of the autonomic hypothesis. Stimulation activates nerve fibers that carry signals toward the brain, traveling to the nucleus tractus solitarius (a brainstem region that regulates autonomic outflow), which suppresses sympathetic activity, increases parasympathetic activity, and engages the cholinergic anti-inflammatory pathway. Implanted cervical VNS has been FDA-approved for treatment-resistant epilepsy since 1997, with four RCTs showing 22 to 43% seizure reductions. In heart failure, INOVATE-HF, the largest trial at 707 patients, found no reduction in mortality, but post-hoc analysis revealed adequate vagal engagement was achieved in only 30% of patients; the comparative analysis concluded insufficient stimulation dose, not a failure of the concept, likely explains the null result. The most persuasive recent positive trial is RESET-RA, which randomized 242 rheumatoid arthritis patients to active or sham VNS and found a significantly higher clinical response rate at three months (35.2% versus 24.2%, p=0.0209), providing prospective human evidence that directly stimulating the vagus nerve suppresses systemic inflammation. Non-invasive options include transcutaneous cervical devices such as gammaCore (FDA cleared for migraine and cluster headache) and transcutaneous auricular devices, which remain investigational in the United States. Consumer ear-clip devices marketed for wellness are unregulated and untested in rigorous trials. A Bayesian meta-analysis of 17 sham-controlled taVNS studies found strong evidence that acute taVNS does not reliably change HRV at the group level.
Where popular belief outruns the trial evidence
High-intensity interval training raises HRV chronically, but acute suppression in the 24 to 48 hours after hard sessions creates apparent volatility in daily readings that can obscure the long-term gain.
Popular supplements marketed for HRV have a thin evidence base. Magnesium may have a modest effect in deficient individuals, but adaptogens and similar compounds have not produced meaningful HRV improvements in rigorous trials.
Acute meditation sessions raise HRV during the session. A 2023 analysis found experienced meditators breathe roughly 1.5 times more slowly than non-meditators, which is likely the primary driver. The chronic resting-state effect is not established. A meta-analysis of 19 RCTs found mindfulness and meditation-based interventions were not efficacious in increasing resting vagal HRV versus control conditions (Hedges g = 0.38, 95% CI −0.014 to 0.77, nonsignificant), with high variability across trials unexplained by intervention duration, setting, or control type.
Technologies on the horizon
Consumer wearables are moving toward more accurate sensors. Current optical wrist sensors read 6 to 11% below ECG values and are vulnerable to motion and temperature. The next generation includes radar-based contactless systems that can measure heartbeat during sleep without any worn device, and long-wear adhesive ECG patches that capture continuous R-wave-based readings rather than optical estimates. Several companies are also working on closed-loop devices that detect HRV dropping in real time and trigger a slow-breathing prompt or a brief vagal stimulus in response, essentially acting as an autonomic pacemaker.
On the stimulation side, miniaturized implantable VNS devices are shrinking toward the size of a cardiac monitor and may eventually be placed through a needle rather than surgery. If RESET-RA data support an FDA approval for rheumatoid arthritis, the commercial pathway would accelerate development across other inflammatory conditions including inflammatory bowel disease, where open-label VNS data are already positive. The broader goal in bioelectronic medicine is to treat inflammatory and metabolic disease by dosing an electrical signal to a specific nerve at a specific frequency, adjusting the dose automatically based on real-time physiological feedback, rather than relying on fixed stimulation protocols.
The most useful near-term clinical application may be in recovery monitoring. HRV drops substantially during and after viral illness, surgery, and physiological stress, and recovery of the HRV baseline tracks biological recovery closely. Several groups are testing whether continuous HRV monitoring in the weeks after hospitalization predicts readmission better than clinical assessment alone.
For personal use, the missing capability is contextual interpretation. The same reading means something different depending on training load, illness, recent stress, and sleep quality. Machine learning models trained on large wearable datasets are beginning to build personalized baselines that compare your reading to your own history under similar conditions rather than to a population average. The longer-term research direction is combining HRV with other passive digital signals, including sleep architecture, gait, and voice, to build composite measures of autonomic and cognitive health that track change over years. For dementia prevention specifically, that kind of longitudinal multimodal tracking is probably where the clinical utility will be largest.
Putting it together
HRV reflects how well your nervous system is modulating your heart, managing inflammation, and adapting to stress. Lower values predict higher rates of cardiovascular events, metabolic disease, and all-cause mortality across large prospective cohorts, and genetic evidence supports at least one causal pathway. The interventions with the clearest RCT support are aerobic exercise, daily slow-paced breathing, and omega-3 supplementation above 1 gram per day. Sleep quality, alcohol reduction, and psychological stress management have strong mechanistic and observational support even where direct HRV-specific RCTs are sparse. Your device reading on any given morning reflects dozens of short-term variables. The trend in your baseline over months and years, and whether the habits that support vagal tone are consistently in place, is what matters.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso