NAD+ and Sirtuins for brain health and longevity
Reviewing the science, the controversial history, the trials, and the gaps
A paper published earlier this year in Science Advances invigorated enthusiasm for NAD+ and brain health. Researchers showed that NAD+ precursors corrected signs of Alzheimer’s disease in a mouse model. The study pointed to a novel mechanism beyond the energy metabolism story that has dominated NAD+ research, improving tau-related RNA splicing patterns, and it led to even more people asking me about NAD+ and Alzheimer’s prevention.
NAD+ supplements are widely available and heavily marketed, and many people are uncertain whether they should take them given the current scientific evidence. This article reviews what NAD+ is and what it does, how Sirtuins (longevity genes (arguably) that need NAD+ to function) fit into the picture, what human clinical trials have found, who is selling what and at what doses, the commercial disputes that have shaped the market, and where the field needs to go to answer the questions consumers are asking.
I am in a unique position to comment on this topic, having spent twenty years studying brain aging and Sirtuin biology before starting NeuroAge, including work linking a genetic variant in the Sirtuin SIRT5 to slower brain aging and work on SIRT6’s role in neurodegeneration. My postdoctoral work was in Professor Leonard Guarente's lab at MIT, which sits at the center of much of this story. That research background gives me a particular vantage point on this literature, and I will draw on those findings where relevant.
What NAD+ and sirtuins are, and why their connection to aging matters
NAD+ (nicotinamide adenine dinucleotide) is a molecule found in every cell of the body that assists enzymes in carrying out chemical reactions. Its most important roles are in converting nutrients from food into usable energy, repairing damaged DNA, and regulating gene expression. NAD+ is not just a passive coenzyme, it is actively consumed by a class of proteins called sirtuins, which are arguably longevity genes that regulate lifespan and healthspan. They use NAD+ as a required partner to remove chemical tags from histones and other proteins, controlling gene expression, DNA repair, mitochondrial function, and inflammatory signaling. Sirtuins cannot function properly when NAD+ levels are low.
This matters for aging because NAD+ levels decline with age in multiple human tissues, including blood, muscle, skin, and brain, through a combination of reduced synthesis and increased consumption by NAD+-degrading enzymes. The implication is that sirtuin activity may decline with age not because the sirtuins themselves are impaired, but because they are running low on fuel. If that is correct, then restoring NAD+ levels could in principle restore sirtuin function and slow aspects of the aging process that sirtuins regulate.
Because NAD+ itself cannot cross cell membranes efficiently, supplements use precursor molecules that enter cells and are converted to NAD+ from within. The two main ones are nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN), both forms of vitamin B3. Standard nicotinamide, the most common Vitamin B3 form, also raises NAD+ but at high doses inhibits the sirtuin proteins it is meant to support, which is why NR and NMN dominate the research and the market.
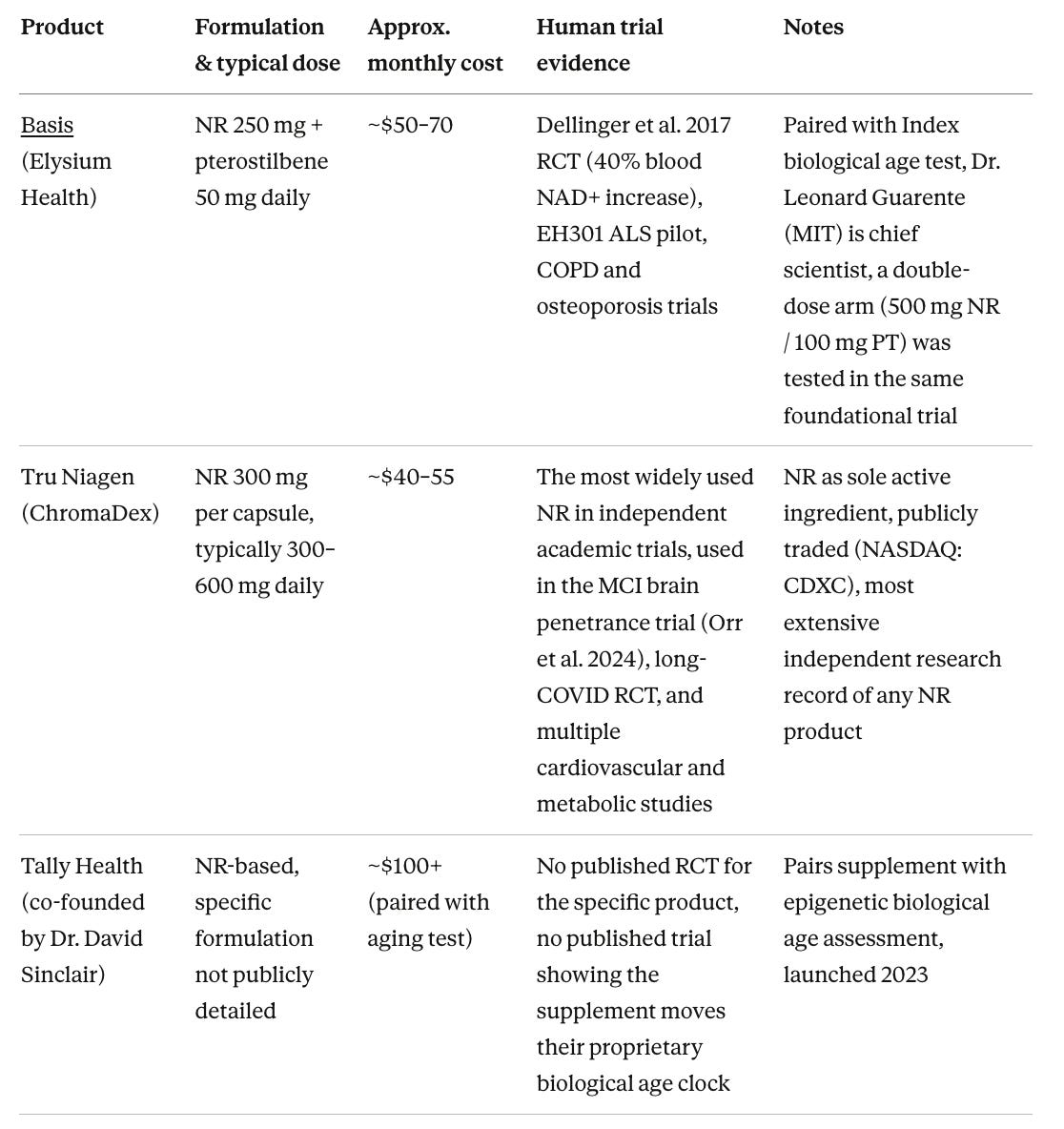
The Sirtuin aging hypothesis drove GlaxoSmithKline’s $720 million acquisition of Sirtris Pharmaceuticals in 2008, the largest single pharmaceutical bet on sirtuin biology, and eventually a consumer supplement market now worth several billion dollars annually. The main commercial players in the NAD+ space are Elysium Health, co-founded by Dr. Lenny Guarente, which sells Basis (NR combined with pterostilbene), ChromaDex, whose chief scientific officer is Dr. Charles Brenner, which makes Tru Niagen, and Tally Health and Metro International Biotech, both associated with Dr. David Sinclair, focused on NR-based consumer supplements and pharmaceutical-grade NMN respectively.
The history of how this connection was established, contested, commercialized, and partially validated is one of the more interesting stories in modern aging biology.
Much of it runs through a single laboratory, Dr. Leonard Guarente’s at MIT. Guarente spent decades studying how yeast cells age, building a research program that produced many of the pivotal findings in sirtuin biology and trained a generation of scientists who went on to shape the field. Those scientists include Dr. David Sinclair, a professor of genetics at Harvard Medical School who co-founded Sirtris Pharmaceuticals and later Tally Health, Dr. Shin-ichiro Imai, a professor at Washington University in St. Louis who identified the NAD-sirtuin connection and later co-founded Metro International Biotech to develop pharmaceutical-grade NMN, Dr. Brian Kennedy, formerly president and CEO of the Buck Institute for Research on Aging and now at the National University of Singapore, and Dr. Matt Kaeberlein, formerly a Director at the University of Washington and now Founder and CEO of Optispan, both of whom identified mTOR as an independent longevity pathway and eventually challenged key assumptions of the sirtuin story, and, later in the timeline, me.
How NAD+ and sirtuins became connected to aging
The early science (1906–1999)
1906 Arthur Harden and William Young discover that yeast fermentation requires a small cofactor, the first identification of what will eventually be named NAD+.
1930s Hans von Euler-Chelpin works out NAD+’s full chemical structure (both men later receive Nobel Prizes). Otto Warburg establishes it as the central electron carrier in cellular respiration.
1937 Conrad Elvehjem shows that pellagra (a disease causing dementia, skin lesions, and digestive failure) results from niacin (vitamin B3) deficiency, establishing niacin as an NAD+ precursor and connecting NAD+ to human health.
1970s–80s Yeast geneticists studying gene silencing identify Sir2 (Silent Information Regulator 2) as a regulator of chromosomal organization. Sir2 is the founding member of what will later be called the sirtuin family, a class of proteins that remove chemical tags from histones and other proteins to regulate gene expression, DNA repair, and energy metabolism. Not yet connected to aging or NAD+.
1995 Dr. Brian Kennedy, a PhD student in Guarente’s lab at MIT, publishes in Cell with Guarente and colleagues the first paper linking SIR genes to yeast aging, showing that a mutation in SIR4 that redistributes the SIR protein complex from telomeres to the nucleolus extends lifespan by more than 30 percent.
1999 Kaeberlein, now also in Guarente’s lab at MIT, publishes with Kennedy and Guarente in Genes and Development that overexpression of Sir2 itself extends yeast lifespan by 30 to 40 percent, establishing Sir2 as the key longevity factor in the SIR complex.
The NAD-sirtuin connection (2000–2007)
2000 Dr. Shin-ichiro Imai in Guarente’s lab publishes in Nature that Sir2 is an NAD-dependent deacetylase, meaning it requires NAD+ to function, directly linking the cell’s metabolic state to longevity through Sir2.
2000 Dr. Su-Ju Lin in Guarente’s lab publishes in Science that caloric restriction in yeast requires both Sir2 and NAD+ to extend lifespan.
2001 Dr. Heidi Tissenbaum, working with Guarente, publishes in Nature that worms with extra copies of the Sir2 equivalent gene live up to 50 percent longer.
2003 Dr. Konrad Howitz and Dr. David Sinclair of Harvard publish in Nature that resveratrol, a polyphenol compound found in the skin of red grapes and widely discussed in connection with the health benefits of red wine, appears to activate SIRT1 (the closest human equivalent of Sir2) and extends yeast lifespan by 70 percent.
2004 Dr. Charles Brenner at Dartmouth publishes in Cell the discovery of the NR kinase pathway, establishing nicotinamide riboside (NR) as a previously unrecognized vitamin B3 and a functional precursor to NAD+.
2004 Sinclair co-founds Sirtris Pharmaceuticals with entrepreneur Christoph Westphal to develop synthetic sirtuin-activating drugs.
2004 Dr. Blanka Rogina and Dr. Stephen Helfand at Yale publish in PNAS that Sir2 mediates caloric restriction-related longevity in fruit flies, completing a three-organism pattern in yeast, worms, and flies.
2004 Kaeberlein and Kennedy, now both at the University of Washington, publish in PLoS Biology that caloric restriction extends yeast lifespan through a pathway that operates independently of Sir2, directly contradicting the 2000 Lin and Guarente finding that CR requires Sir2 and NAD+. The two groups disagree about the interpretation, a dispute that would not be resolved for years.
2005 Kaeberlein and Kennedy publish in Science that the TOR and Sch9 kinases, rather than Sir2, define the primary pathway through which excess nutrients limit lifespan in yeast, and that CR extends lifespan independently of Sir2 through this pathway. The TOR kinase is the yeast equivalent of mTOR (mechanistic target of rapamycin), and rapamycin, a drug that extends lifespan in mice, inhibits it. This finding pointed toward what would become the most reproducible pharmacological aging intervention across species.
2007 As a PhD student in Dr. Etienne Sibille’s lab at the University of Pittsburgh (Dr. Sibille is now Founder of Damona Therapeutics), I contribute to a paper published in Molecular Psychiatry showing upregulated SIRT5 expression in the frontal cortex of serotonin 1b receptor knockout mice, a mouse model with anticipated brain aging, establishing a link between sirtuin gene expression and age-related brain changes in a mammalian model.
2007 Brenner’s lab publishes in Cell that NR extends yeast lifespan by activating Sir2, connecting NR to the sirtuin pathway. Brenner also joins the Sirtris scientific advisory board.
Peak, controversy, and collapse (2008–2013)
2008 GlaxoSmithKline acquires Sirtris for $720 million. Brenner leaves the Sirtris advisory board after the acquisition, growing skeptical of the sirtuin lifespan claims.
2009–2010 Independent research groups at Amgen and Pfizer publish findings suggesting resveratrol’s apparent SIRT1 activation in the original Howitz assay was an artifact of the fluorescent tag used in the experiment. GSK defends its program while acknowledging the technical dispute.
2010 GSK halts its Phase II trial of SRT501 (a resveratrol formulation) in relapsed or refractory multiple myeloma after five patients develop acute kidney failure and at least one patient in the trial dies. GSK concludes the kidney failures were most likely driven by the underlying disease, compounded by dehydration from SRT501’s gastrointestinal side effects (nausea, vomiting, diarrhea), rather than direct drug toxicity, but discontinues all development of SRT501.
2010 Dr. Gizem Donmez in Guarente’s lab publishes in Cell that SIRT1 overexpression in a mouse model of Alzheimer’s disease reduces amyloid burden by activating the α-secretase gene ADAM10, one of the most high-profile claims for a direct sirtuin neuroprotective role. The paper is retracted in 2014 for image doctoring, though Guarente maintains the underlying conclusion is supported by independent replication.
2011 Dr. David Gems, Dr. Linda Partridge, and a consortium from seven institutions publish in Nature (first author Camilla Burnett) that Sir2 overexpression effects in worms and flies disappear when genetic background is properly controlled. Guarente publishes a response in the same issue maintaining that a smaller effect of 10 to 14 percent persists in cleaned-up strains, down from the original 15 to 50 percent. The dispute is not fully resolved, and researchers assessed the competing evidence differently.
2011 As a PhD student at the University of Pittsburgh, I publish in Neurobiology of Disease that people carrying a high-expressing genetic variant in SIRT5, a mitochondrial sirtuin, have measurably younger brain ages across four brain regions and multiple cohorts, connecting sirtuin function directly to the rate of brain aging in humans.
2012 Donmez publishes in Journal of Neuroscience that SIRT1 overexpression extends lifespan and reduces alpha-synuclein aggregation in a Parkinson’s disease mouse model. This paper is retracted in 2016 for the same categories of figure manipulation. Donmez stepped down from her faculty position at Tufts by 2014.
2012 Dr. Yariv Kanfi and colleagues at Bar-Ilan University publish in Nature that male mice overexpressing SIRT6 live significantly longer, providing the first mammalian sirtuin lifespan result (females in that strain were unaffected).
2013 GSK shuts down Sirtris. No drug reaches approval from the $720 million investment.
2013 Dr. Ana Gomes and colleagues in Dr. David Sinclair’s lab at Harvard publish in Cell that declining NAD+ in aging muscle disrupts communication between the nucleus and mitochondria, creating a state resembling low oxygen even when oxygen is adequate, and that raising NAD+ levels restores mitochondrial function in old mice.
2013 Dr. Akiko Satoh in Imai’s lab publishes in Cell Metabolism that brain-specific SIRT1 overexpression extends lifespan in both male and female mice (the BRASTO mice), acting through enhanced neural activity in the hypothalamus, showing that where a sirtuin is expressed matters as much as whether it is expressed, since whole-body SIRT1 overexpression does not extend lifespan
NAD+ precursors take center stage (2014–present)
2014 Guarente co-founds Elysium Health to sell Basis (NR plus pterostilbene supplement), sourcing NR from ChromaDex, which has licensed Brenner’s NR patents from Dartmouth and signed Brenner as chief scientific officer.
2016 Brenner’s group publishes in Nature Communications that NR safely raises blood NAD+ in humans. The Elysium-ChromaDex supplier relationship collapses and litigation begins.
2016 Dr. Kathryn Mills and Dr. Shin-ichiro Imai publish in Cell Metabolism the first 12-month long-term NMN study in mice, showing suppression of age-associated weight gain, improved energy metabolism and physical activity, improved insulin sensitivity, and preserved eye function, without toxicity.
2017 Dellinger, Guarente, and colleagues publish the first completed human NR RCT in npj Aging, showing a 40 percent sustained increase in blood NAD+ with Basis in 120 older adults.
2018 As a postdoctoral researcher in Dr. Guarente’s lab at MIT, I contribute to a study led by Dr. Sergiy Libert (Cornell professor, formerly Guarente Lab postdoc, now at Calico) and published in Acta Neuropathologica Communications finding that SIRT6 levels are elevated in the brains of Parkinson’s disease patients, that brain-specific SIRT6 knockout mice are protected from Parkinson’s-like pathology, and that nicotine appears to provide partial neuroprotection by reducing SIRT6 levels. This is the opposite of what the whole-body lifespan data would have predicted, and a reminder that sirtuin activity in specific disease contexts can work differently than it does in healthy aging.
2019 As a postdoctoral researcher in Dr. Guarente’s lab at MIT, I publish in Life Science Alliance that sirtuin signaling is among the gene expression pathways most consistently downregulated with normal human brain aging, alongside mitochondrial and oxidative phosphorylation pathways, and that the rate of brain molecular aging and APOE4 status synergistically increase Alzheimer’s risk, work consistent with the hypothesis that declining NAD+ impairs sirtuin function in aging neurons.
2021 A federal court invalidates ChromaDex’s NR composition patents. A jury verdict results in Elysium paying approximately $3 million to ChromaDex while recovering approximately $1.9 million.
2021 A follow-on SIRT6 study by Kanfi’s group in Nature Communications finds lifespan extension and reduced frailty in both male and female SIRT6-overexpressing mice in a different genetic background.
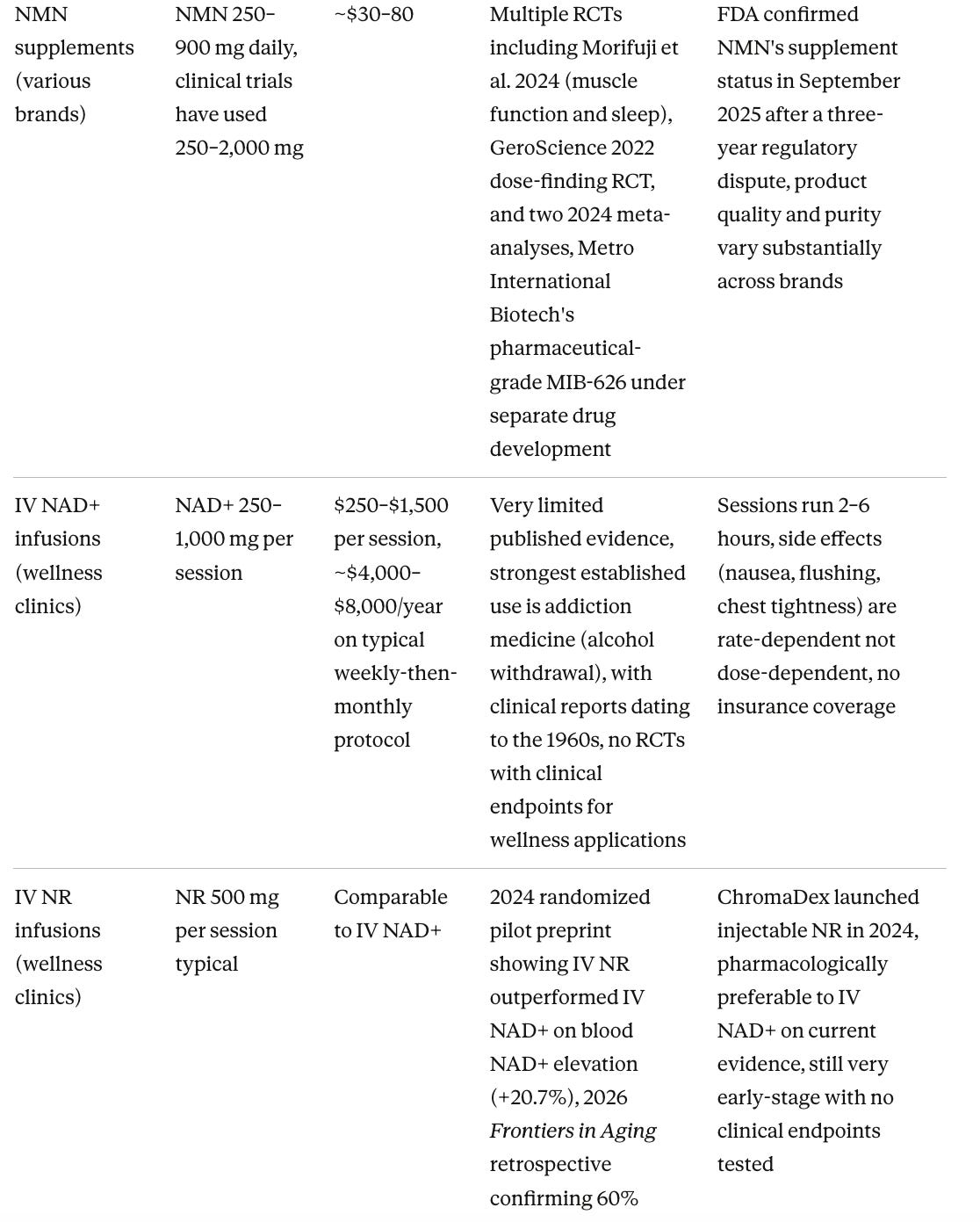
2022 Sinclair’s Metro International Biotech files an IND for pharmaceutical-grade NMN (MIB-626), triggering the FDA to exclude NMN from supplement sales under the drug preclusion rule. Brenner publishes a critique of Sinclair’s book Lifespan in Archives of Gerontology and Geriatrics.
2025 The FDA reverses course in September, acknowledging NMN was sold as a supplement as early as 2017. NMN returns to major retail channels.
The biochemical connection between NAD+ levels and sirtuin activity is direct and well-established, since sirtuins require NAD+ to function. Whether restoring NAD+ in aging humans meaningfully reactivates sirtuins in tissues to a degree that produces measurable health outcomes is what the current clinical trials are designed to test.
Clinical trials show reliable NAD+ elevation but more modest and inconsistent efficacy
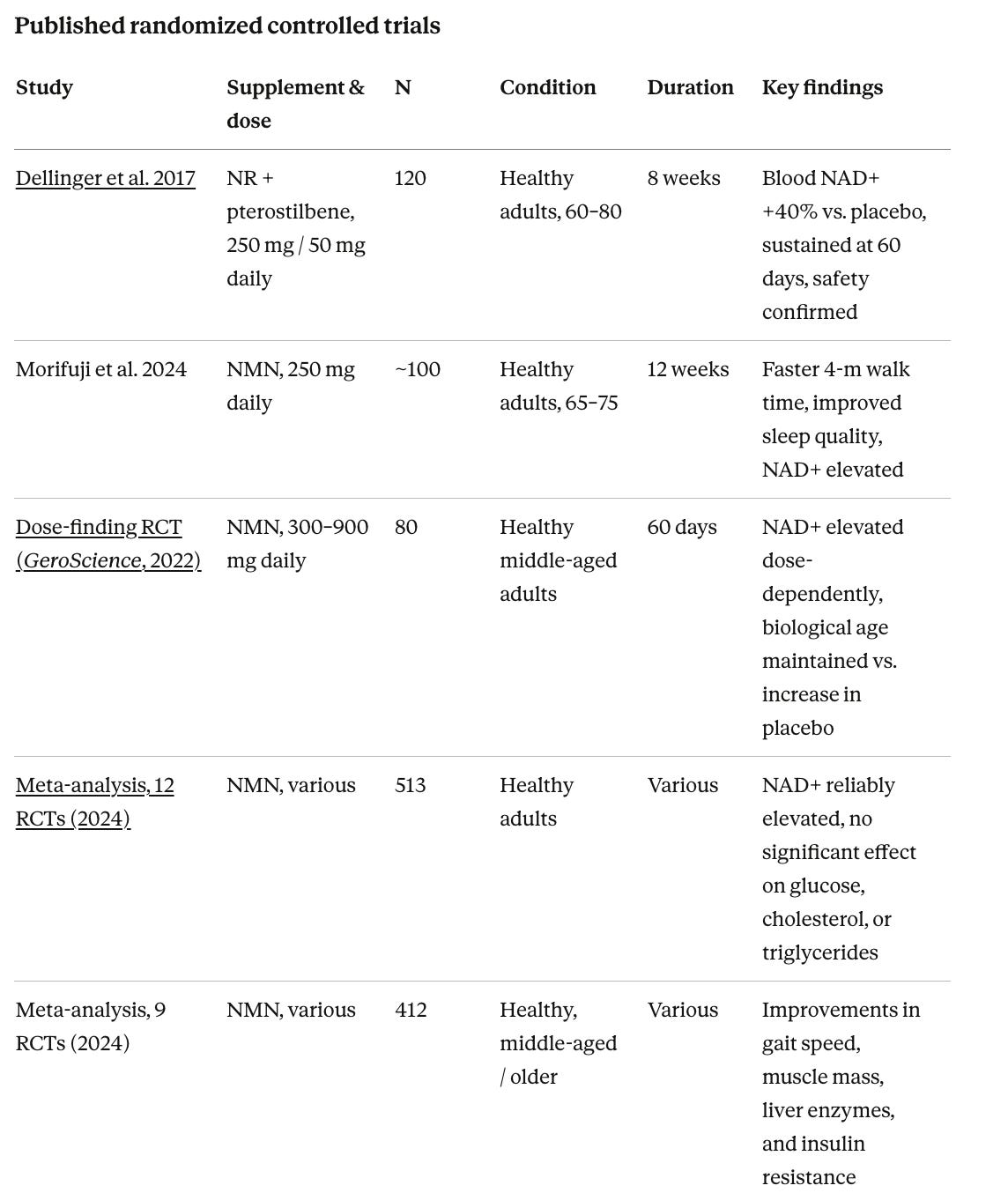
The human clinical evidence divides into studies measuring how much NAD+ the body produces in response to supplementation, and functional studies testing whether that NAD+ elevation translates into measurable health or cognitive benefits.
Clinical trials consistently show that NAD+ precursors are absorbed and raise NAD+. A foundational 2017 randomized, double-blind, placebo-controlled trial by Dr. Ryan Dellinger, Dr. Leonard Guarente, and colleagues tested Basis (a combination of 250 mg NR and 50 mg pterostilbene, a plant compound found in blueberries that activates sirtuins) in 120 healthy adults aged 60 to 80 over eight weeks. Whole blood NAD+ increased by approximately 40% at the recommended dose and was sustained through the study period. Subsequent comparisons have found that NR and NMN produce comparable chronic increases in blood NAD+ levels, while standard nicotinamide produces only a transient effect.
For efficacy data and functional outcomes, the data is mixed and emerging. A 2024 meta-analysis pooling nine NMN studies found improvements in gait speed, muscle mass, and insulin resistance in older adults, while a separate 2024 systematic review of 12 studies found reliable NAD+ elevation but no significant differences from placebo on metabolic endpoints including fasting glucose, total cholesterol, and LDL cholesterol. Risk-of-bias assessment rated most included studies as having at least some concerns.
ALS
The most developed disease signal. A 2019 pilot RCT (De la Rubia et al.) testing NR plus pterostilbene in 32 ALS patients at 1,200 mg daily over four months showed improvements in disease severity, pulmonary function, and muscle strength, but had baseline group imbalances that limit interpretation. A larger Phase 2 trial through Haukeland University Hospital is underway with results expected in 2026.
Mild cognitive impairment and early Alzheimer’s
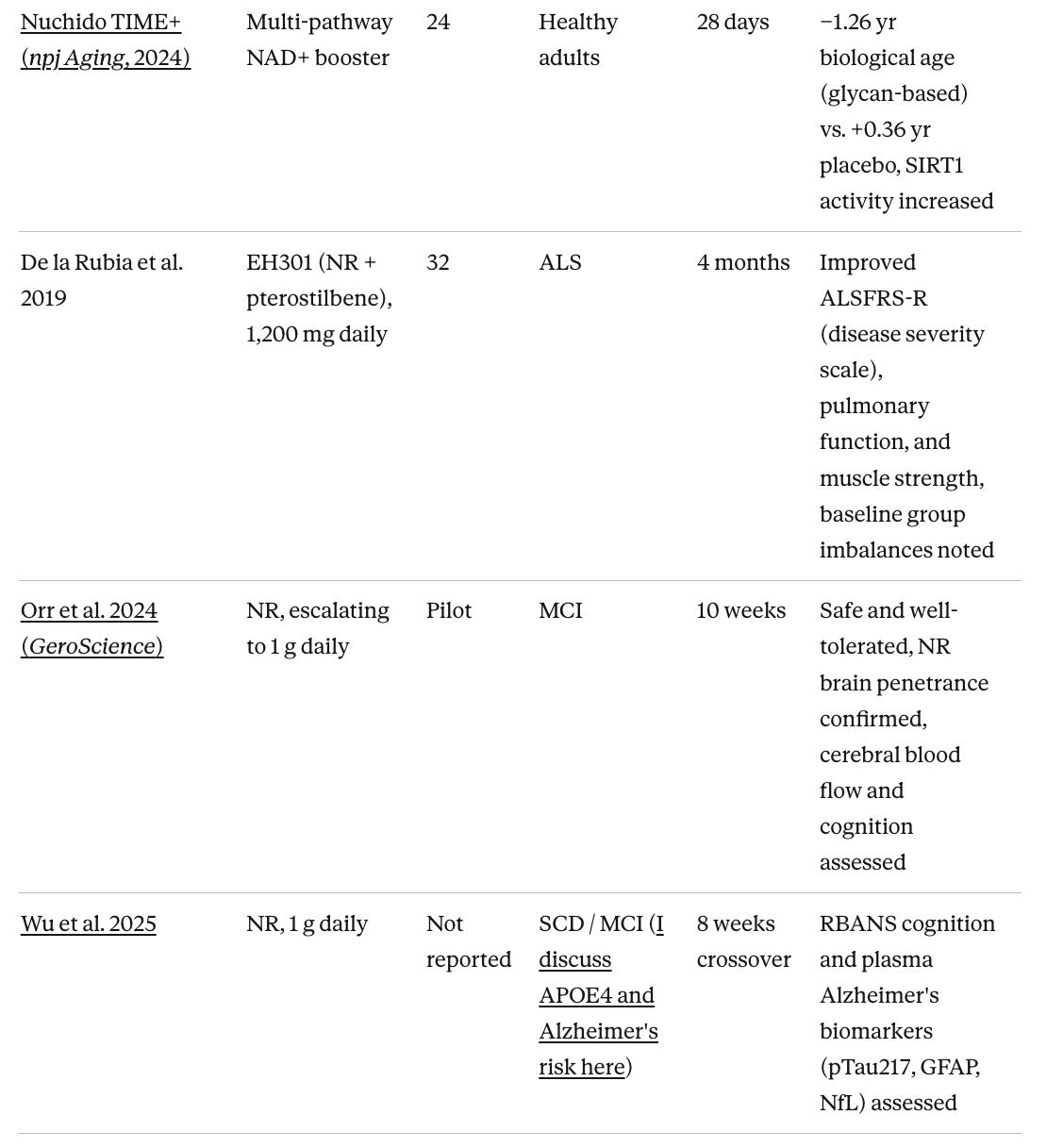
Orr et al. 2024 confirmed NR at up to 1 gram daily is safe in MCI patients and, notably, demonstrated that NR crosses the blood-brain barrier in humans. The brain penetrance finding is important as it had not been established in humans before. A 2025 crossover trial (Wu et al.) tested 1 gram NR daily in subjective cognitive decline and MCI patients and measured plasma Alzheimer’s biomarkers including pTau217, GFAP, and neurofilament light chain, but published results are not yet fully reported.
Long-COVID
A 2025 RCT published in eClinicalMedicine tested 2,000 mg NR daily in 58 long-COVID patients over 24 weeks, measuring cognitive symptoms and mitochondrial and neurological endpoints. Full findings are emerging.
Ongoing trials with no published results yet
The most anticipated are NADAPT at Haukeland, testing NR in approximately 295 patients with progressive supranuclear palsy and multiple system atrophy (both severe and currently untreatable), with results expected December 2028; NAD-HD testing NR at 2,000 mg daily in Huntington’s disease with results around 2028; and a progressive MS trial also at Haukeland completing December 2027.
The overall picture is that NR brain penetrance is confirmed, early ALS signals are encouraging but not definitive, and the most important trials are still running. No published neurological trial to date has used a validated biological aging clock as its primary endpoint.
Companies selling NAD+ precursors with paired biological aging tests have not yet published trials connecting their supplements to their own clock measures
One aspect of the clinical landscape that has not yet been addressed in the peer-reviewed literature deserves attention. Several companies that market NAD+ supplements also offer biological aging tests, most commonly DNA methylation-based clocks (which estimate biological age from chemical modifications to DNA) or blood-based calculators that detect changes in aging rate over relatively short time periods. These tools were specifically designed to evaluate whether interventions slow biological aging, and they are available at a resolution that would, in principle, allow a trial of six to twelve months to detect a meaningful signal if one exists.
As of the time of writing, no company with a financial interest in both an NAD+ supplement and a proprietary biological aging measure has published a peer-reviewed, placebo-controlled trial demonstrating that their supplement produces a statistically significant change on their own biological aging tool. This does not mean such studies have not been conducted, and there are legitimate reasons a trial might be underway but not yet complete, including cost, study duration, and statistical power requirements. It does mean that consumers purchasing these product combinations are currently asked to accept, on the basis of mechanistic plausibility and biomarker data, a chain of reasoning that has not been closed with the obvious endpoint.
Independent groups have produced relevant data. A 2022 multicenter, dose-dependent randomized controlled trial published in GeroScience found that NMN supplementation maintained blood biological age on the Aging.AI hematological clock (a tool that estimates biological age from 19 standard blood parameters) while the placebo group showed an increase over 60 days. A 2024 trial published in npj Aging tested a multi-pathway NAD+ supplement and found a reduction of approximately 1.26 biological years on IgG glycosylation profiling (a glycan-based measure of immune aging) compared to an increase of 0.36 years in placebo over 28 days, alongside increases in SIRT1 activity and reductions in inflammatory markers. Both studies used biological aging measures. Neither was conducted by the major companies that market combined supplement-and-clock packages.
Trials using well-validated second-generation epigenetic clocks, specifically GrimAge2, PhenoAge, and DunedinPACE, which have demonstrated responsiveness to interventions including caloric restriction and omega-3 supplementation in large, well-designed trials, have not appeared as primary endpoints in any published NAD+ supplementation trial. The field’s most rigorous tools for assessing whether an intervention slows biological aging have not yet been formally applied to NAD+ precursors in published, controlled research.
The NAD+ supplement market involves several companies, distinct formulations, and a range of dosing approaches
The major products currently on the market are summarized in the table below, covering formulation, typical dose, approximate monthly cost, and the state of human trial evidence for each.
Pterostilbene, the blueberry compound in Basis, belongs to the same chemical family as resveratrol but is considerably better absorbed when taken by mouth, and its inclusion was a deliberate scientific choice grounded in the sirtuin model Dr. Guarente has spent decades developing. The two things that are needed for Sirtuins to function are NAD+ as fuel and a signal to become active. The theory behind Basis is that NR provides the fuel while pterostilbene directly activates SIRT1 through a distinct mechanism, producing greater sirtuin activity than either ingredient alone, though whether that synergy holds at the doses used has not been tested in a head-to-head comparison of the combination versus NR alone.
One pharmacological caveat worth noting is that a substantial portion of orally administered NR and NMN is converted to nicotinamide by gut bacteria and liver enzymes before reaching the bloodstream, then recycled back to NAD+ via the salvage pathway, which means blood NAD+ reliably rises with supplementation but tissue-level increases, including in the brain, are less certain.
Intravenous NAD+ infusions are widely sold but their evidence base is thin
The main claim wellness clinics make for IV delivery, that it achieves superior bioavailability compared to oral supplements, is partly misleading. NAD+ is a large, charged molecule that cannot cross cell membranes intact, so when infused intravenously, most of it is broken down in the blood into NMN, then NR, then nicotinamide before entering cells, meaning it ultimately works through the same cellular recycling route as oral precursors with only the gut absorption step bypassed.
A 2019 pilot study in Frontiers in Aging Neuroscience found plasma NAD+ did not begin rising until two hours into a six-hour infusion, with elevated urinary metabolites suggesting rapid clearance rather than sustained cellular retention. IV NR appears pharmacologically preferable to IV NAD+ itself. A 2024 randomized pilot trial found IV NR raised whole blood NAD+ by 20.7% above baseline and significantly outperformed IV NAD+, and a 2026 Frontiers in Aging retrospective confirmed approximately 60% shorter infusion time with better tolerability, since IV NAD+ at faster rates produces nausea, flushing, and chest tightness that require slowing the drip. The published evidence base for IV delivery in general wellness applications remains a collection of small pilot studies, with the strongest established use in addiction medicine dating to the 1960s. For most people reading this piece, oral supplementation is the better-supported approach at a fraction of the cost.
Summary
The field has followed a recognizable trajectory. Kennedy and Kaeberlein’s work in the 1990s and early 2000s identified sirtuins and then mTOR as longevity regulators in yeast. Imai and Lin connected NAD+ to Sir2 activity, and the hypothesis that restoring NAD+ in aging tissues could restore sirtuin function attracted substantial scientific and commercial investment. GSK’s $720 million acquisition of Sirtris in 2008 was the peak. What followed was a difficult decade, the resveratrol activation mechanism turned out to be an assay artifact, the invertebrate lifespan claims weakened substantially when genetic background was properly controlled, two prominent sirtuin neuroprotection papers from Guarente’s lab were retracted for image manipulation, and GSK shut Sirtris in 2013 with no approved drug. The researchers who pushed back most consistently on the dominant narrative, Kaeberlein and Kennedy adding mTOR and rapamycin as a sirtuin-independent longevity pathway and Brenner questioning the sirtuin lifespan claims, added nuance that the field eventually absorbed, though Brenner’s position as chief scientific officer of ChromaDex complicates how to read his skepticism of competing sirtuin activation approaches.
The pivot to NAD+ precursors from the mid-2010s onward came from a different direction. Sinclair’s 2013 finding that NAD+ decline disrupts nucleus-mitochondria communication, Brenner’s earlier identification of NR as an orally bioavailable NAD+ precursor, and Imai’s NMN mouse data collectively shifted the focus from sirtuin activation to NAD+ replenishment as the proximal target. NR and NMN reliably raise blood NAD+ levels in humans, and this result has replicated consistently across trials. Some functional benefits in older adults are beginning to emerge, particularly in physical performance, and the disease-specific trials in ALS, atypical parkinsonism, Huntington’s, and progressive multiple sclerosis are the most serious human tests of the NAD+ replenishment hypothesis yet attempted.
Several questions remain unresolved. Whether the sirtuin lifespan effects in mice generalize to humans is unclear. SIRT6 overexpression extends lifespan in mice, but SIRT1 whole-body overexpression does not, and research I contributed to in Guarente’s lab found that SIRT6 is elevated in Parkinson’s disease brains and that reducing SIRT6 activity appears neuroprotective in that context, which is the opposite of what the lifespan data would predict. The mammalian sirtuin picture is tissue-specific and disease-context-dependent in ways that simple restoration narratives do not capture.
On the human trial side, no adequately powered, pre-registered randomized trial has yet used a validated biological aging clock as a primary endpoint for NR or NMN supplementation. That is the most important missing piece for consumers and clinicians trying to evaluate whether NAD+ precursors slow aging-related decline rather than simply raising a biomarker. Metabolic endpoints in healthy adults have been largely unaffected in the trials completed so far. And the commercial entanglement of the field, Guarente at Elysium, Sinclair at multiple ventures, Brenner at ChromaDex, means that some of the most prominent voices in the literature have financial stakes in the outcome and are in ongoing legal (and social media) disputes.
The mechanistic case connecting NAD+ decline, sirtuin impairment, and the hallmarks of aging is coherent and grounded in decades of careful work. NR and NMN have a good safety profile and an accessible price point. My own findings on SIRT5 genetic variants, brain molecular aging rates, and APOE4 synergy with aging are consistent with the broader hypothesis that sirtuin biology matters for brain aging in humans. The ongoing disease trials may produce results that reshape how the field thinks about brain-targeted NAD+ replenishment. Whether the gap between the preclinical evidence and the human functional evidence closes over the next several years is an open empirical question, and I am watching it closely.
Am I taking NAD+ supplements?
No, I’m not taking NAD+ supplements. 90% of drugs that work in mice fail in clinical trials and the clinical trial data is not there yet. The directionality and role of the various Sirtuins in human brain still needs to be worked out. My own work showed that Sirt5 might be protective in human brain but Sirt6 might be harmful. NAD+ supplements affect the action all of the sirtuins. For me to feel comfortable putting a supplement in my body, I need to know that it won’t harm me in some cases and help me in others.
New therapeutics that target specific sirtuins in specific tissues could resolve this issue. Basically this would be the continuation of what Sirtris attempted to achieve but was shelved by GSK.
Positive clinical trial data with aging clocks for dementia prevention and cognitive endpoints might also change the calculus for me. I am cautiously optimistic about the future of NAD+ and Sirtuin therapeutics and will continue to watch the field closely.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso