Growth Hormone/IGF-1 Optimization, Brain Health, and Longevity
Why reversing somatopause should give you pause
The somatotropic axis presents one of the most fascinating contradictions in aging science: reducing GH/IGF-1 signaling extends lifespan by 40-70% in mouse models, yet the natural decline of these hormones with age (somatopause) is associated with cognitive decline, frailty, and neurodegeneration in humans. Understanding this “paradox” is essential before considering interventions like growth hormone supplementation or sermorelin (peptide that increase GH).
At first glance, the mouse and human data appear to point in opposite directions. In mice, genetic mutations that reduce GH/IGF-1 signaling from birth produce dramatic lifespan extension of 40-70%. In humans, the natural age-related decline in these hormones (somatopause) is associated with muscle loss, cognitive decline, and frailty. However, human genetic data from centenarians and Laron syndrome patients actually supports the mouse findings: reduced GH/IGF-1 signaling appears protective. The key question is why pharmacological supplementation fails to restore function if the decline is harmful, and whether the age-related decline is a cause of aging or merely a correlate.
Here I synthesize evidence from genetically modified mouse models, human clinical trials, GHRH analog research, and centenarian studies to provide a framework for understanding this complex biology.
The Somatopause: Age-Related Decline in GH and IGF-1
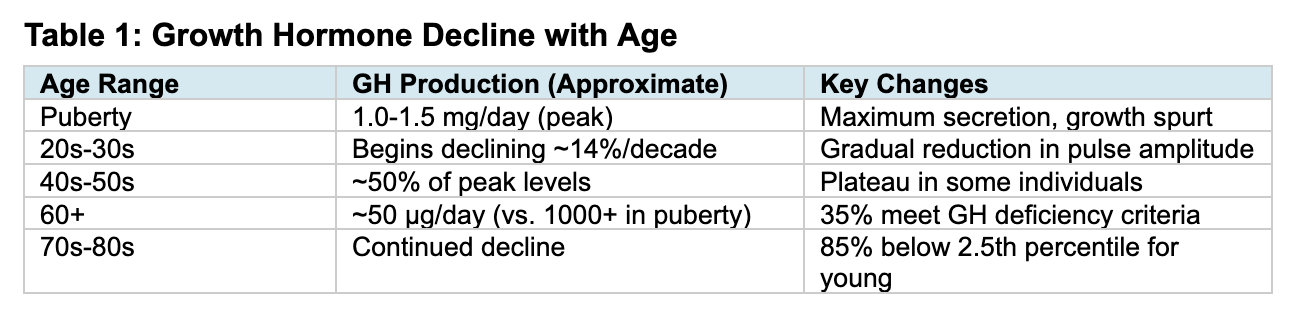
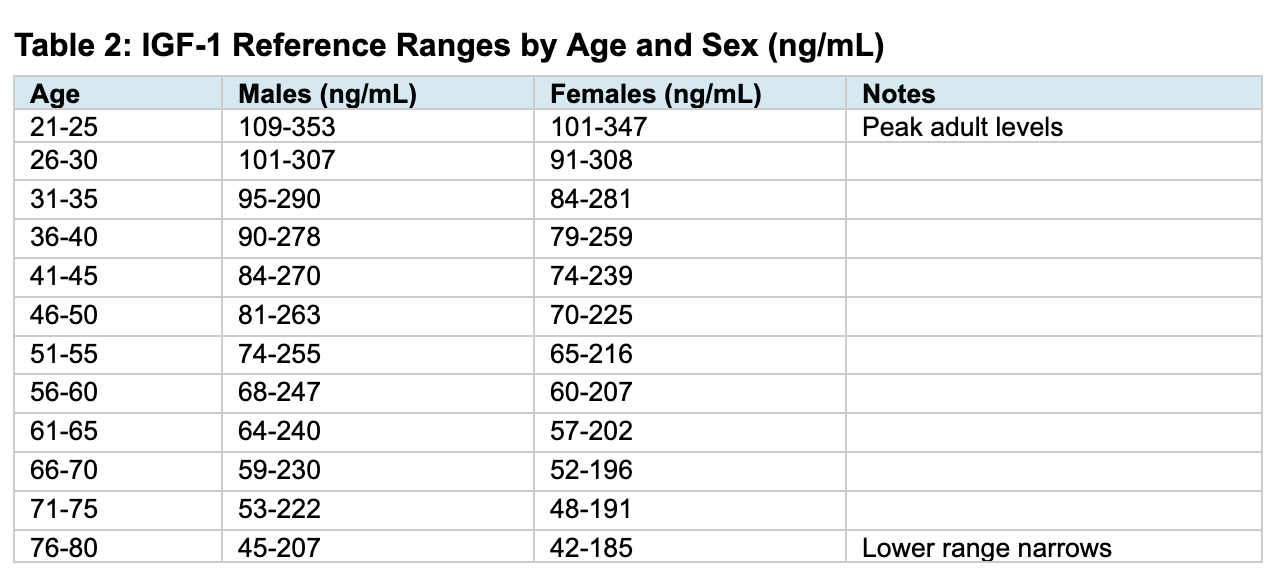
Beginning in early adulthood, growth hormone secretion progressively declines in a phenomenon termed “somatopause.” GH production decreases by approximately 14% per decade (J Clin Endocrinol Metab 1996), or roughly 1-2% per year (Mayo Clin Proc 2021). By age 60, approximately 35% of men meet criteria for GH deficiency, and 85% of healthy men aged 59-98 have IGF-1 levels below the 2.5th percentile for younger men (PMC3940699). This decline is associated with loss of lean body mass, increased adiposity, decreased bone density, and cognitive changes (Endotext 2019).
The reduction in GH and IGF-1 in healthy aging adults is primarily due to a reduction in the amplitude of GH secretory pulses rather than changes in pulse frequency or GH half-life. While the decline occurs in both sexes, some studies suggest the pattern differs: women may show a more uniform decline, while men exhibit a more prominent mid-life plateau before further reduction (PMC9206165).
Source: Warde Medical Laboratory reference ranges. Note that reference ranges may vary by assay and laboratory. The median IGF-1 level in healthy 18-year-olds is approximately 374 ng/mL, declining to 180 ng/mL by age 35-39 (48% of peak) and to 93 ng/mL by age 70+ (25% of peak) (PMC5627923).
Mouse Data: Reduced GH/IGF-1 Signaling Extends Lifespan
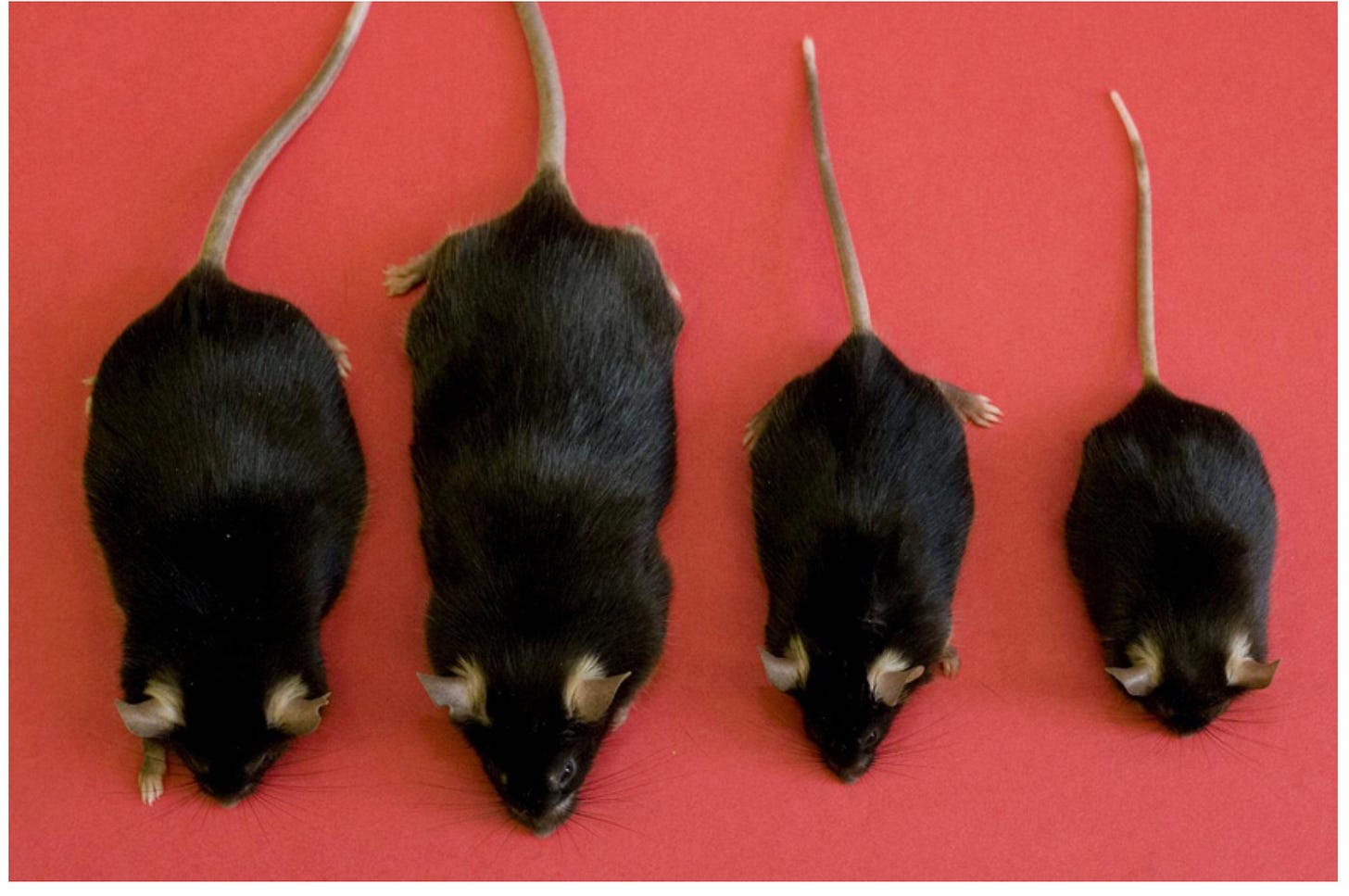
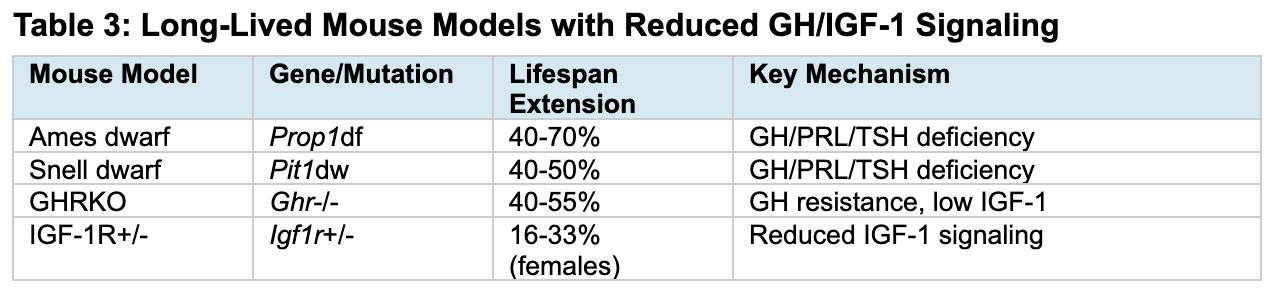
The most compelling evidence for GH/IGF-1 and longevity comes from dwarf mouse models. Ames dwarf mice (Prop1 mutation) live 50-70% longer than wild-type siblings, with enhanced insulin sensitivity, reduced oxidative damage, and preserved cognitive function into old age (Nature 1996; Horm Behav 2001). Snell dwarf mice (Pit1 mutation) show similar 42-70% lifespan extension (PNAS 2001). GH receptor knockout mice demonstrate 40-55% extension despite obesity, with one female reaching nearly 5 years (Endocrinology 2000). IGF-1 receptor heterozygotes, which develop normally without dwarfism, show 33% lifespan extension in females (Nature 2003).

Table 3: Long-Lived Mouse Models with Reduced GH/IGF-1 Signaling
Mouse Studies on IGF-1 and Alzheimer’s Pathology
The relationship between IGF-1 and Alzheimer’s in mice is context-dependent. Peripheral IGF-1 promotes amyloid-β clearance by enhancing transport of Aβ carrier proteins into the brain (Nat Med 2002; Neurobiol Aging 2006). However, adult-onset neuronal IGF-1R knockout in APP/PS1 mice with established pathology improved spatial memory and reduced plaques through enhanced autophagic clearance (J Neurosci 2015). This “paradox” resolves when considering timing: peripheral IGF-1 aids early clearance, while reducing neuronal signaling enhances proteostasis in established disease.
Human Clinical Trials and Studies with GH and IGF-1
In contrast to the mouse longevity data suggesting reduced GH/IGF-1 extends lifespan, human clinical trials have generally focused on GH supplementation to counteract the negative effects of somatopause. The results have been mixed, with consistent body composition changes but limited evidence for functional improvements.
The Rudman Study: A Landmark but Flawed Beginning
The modern interest in GH for anti-aging began with Daniel Rudman’s 1990 New England Journal of Medicine study, which treated 12 men aged 61-81 with recombinant HGH for 6 months (N Engl J Med 1990; 323:1-6). Results showed an 8.8% increase in lean body mass and 14.4% decrease in adipose tissue mass, leading to widespread media claims that GH could “reverse aging.” However, the study was unblinded, had no placebo control, and measured no functional outcomes such as strength, cognition, or quality of life.
Subsequent Randomized Controlled Trials
The Papadakis 1996 RCT provided more rigorous data. In this placebo-controlled trial of older men, GH treatment improved body composition (increased lean mass, decreased fat mass) but produced no improvements in muscle strength, physical function, or cognitive performance (Ann Intern Med 1996; 124:708-16). This dissociation between body composition and functional outcomes has been replicated in subsequent trials.
A 2007 meta-analysis of RCTs by Liu and colleagues analyzed 18 randomized controlled trials of GH in healthy elderly adults (Ann Intern Med 2007; 146:104-15). The pooled results showed a mean 2.1 kg increase in lean body mass and 2.1 kg decrease in fat mass. However, there was no improvement in bone density, cholesterol levels, maximal oxygen consumption, or muscle strength. Importantly, adverse events were significantly more common with GH treatment, including soft tissue edema, arthralgias, carpal tunnel syndrome, and gynecomastia. The authors concluded that GH cannot be recommended as an anti-aging therapy.
The TRIIM Trial: Epigenetic Age Reversal
The Thymus Regeneration, Immunorestoration, and Insulin Mitigation (TRIIM) trial, published by Fahy and colleagues, represents the first demonstration of epigenetic age reversal in humans (Aging Cell 2019; 18:e13028). This open-label pilot study treated 9 men (ages 51-65) for 12 months with a combination protocol including recombinant HGH (0.015 mg/kg individualized to IGF-1 response, 3-4x weekly), DHEA (50 mg daily), metformin (500 mg daily), and vitamin D3/zinc supplementation.
Primary outcomes showed mean epigenetic age decrease of approximately 2.5 years across four epigenetic clocks (Horvath, PhenoAge, Hannum, GrimAge), with statistical significance for each (p<0.01). The GrimAge decrease persisted 6 months after discontinuing treatment. MRI demonstrated significant thymic regeneration in 7 of 9 participants, with replacement of thymic fat by functional tissue.
Limitations are substantial: the study was open-label, included only 9 participants, had no placebo control, and measured no functional outcomes. The ongoing TRIIM-X trial (NCT04375657) aims to address some limitations with 80-100 participants including women, ages 40-80, with an active control group.
Human Epidemiological and Centenarian Data
A 2022 meta-analysis of 19 observational cohort studies with 30,876 participants established a clear U-shaped relationship between IGF-1 and all-cause mortality (Aging Cell 2022; 21:e13540). Low IGF-1 (<80 ng/mL) carried a hazard ratio of 1.33 (95% CI 1.14-1.57) for increased mortality. High IGF-1 (>200 ng/mL) carried a hazard ratio of 1.23 (95% CI 1.06-1.44). The optimal range of 120-160 ng/mL was associated with lowest mortality.
Studies of Ashkenazi Jewish centenarians by Barzilai’s group found that approximately 60% have genomic alterations in the GH/IGF-1 pathway, including IGF-1R mutations conferring partial receptor resistance (PNAS 2008). Importantly, these individuals may have normal or even elevated circulating IGF-1 but reduced cellular response, achieving signaling reduction without absolute deficiency.
The Ecuadorian Laron syndrome cohort (approximately 100 individuals with GH receptor mutations) demonstrates near-complete protection from cancer and diabetes despite severe obesity (Sci Transl Med 2011). Their lifespan is normal (not extended), but healthspan is dramatically improved with diseases appearing 20-30 years later than in relatives (MedlinePlus Genetics). Notably, unlike dwarf mice, Laron syndrome patients do not show extended lifespan, highlighting that the dramatic mouse findings do not translate directly to humans.
Mendelian Randomization Evidence: Testing Causality
Mendelian randomization (MR) studies use genetic variants as instrumental variables to infer causal relationships, avoiding the confounding and reverse causation that plague observational studies. MR evidence provides critical insight into whether the associations between IGF-1 levels and various outcomes are causal or merely correlational.
IGF-1 and Dementia/Cognitive Decline: No Causal Effect
A key 2018 MR study in Neurology examined whether genetically predicted IGF-1 levels affect Alzheimer’s disease risk using data from the International Genomics of Alzheimer’s Project (17,008 cases; 37,154 controls) and the Swedish Twin Registry (984 cases; 10,304 controls). The study found no evidence that genetic variation in IGF-1 or IGFBP-3 affects Alzheimer’s disease risk (Neurology 2018). This null finding is important: it suggests that the observational associations between low IGF-1 and cognitive decline may reflect reverse causation (dementia or frailty causing IGF-1 to decline) rather than low IGF-1 causing dementia.
A 2023 MR study in the European Journal of Epidemiology similarly examined genetically predicted IGF-1 in relation to Alzheimer’s disease using large GWAS datasets (21,982 cases; 41,944 controls) and UK Biobank proxy cases. The study investigated whether growth hormones are causally associated with AD risk, providing additional evidence on this question (Eur J Epidemiol 2023).
These MR findings have significant implications: if low IGF-1 does not causally increase dementia risk, then interventions to raise IGF-1 would not be expected to prevent cognitive decline. This aligns with the RCT data showing no cognitive benefit from GH supplementation despite body composition improvements.
IGF-1 and Cancer: Causal Risk Increase
In contrast to the null findings for dementia, MR studies consistently show that genetically elevated IGF-1 causally increases cancer risk. A 2020 MR study using 416 SNPs associated with IGF-1 levels found that each standard deviation increase in genetically predicted IGF-1 was associated with increased colorectal cancer risk (OR 1.11 in UK Biobank, OR 1.22 in consortium data, p=3.9x10-4). The study also found suggestive positive associations for prostate cancer (OR 1.03-1.10) and breast cancer (OR 1.08 in the Breast Cancer Association Consortium) (Cancer Med 2020).
A large 2022 collaborative analysis of 20 prospective studies with over 17,000 prostate cancer cases confirmed that IGF-1 is causally associated with prostate cancer risk, including aggressive disease. The MR analysis (using UK Biobank instruments and PRACTICAL consortium outcome data with 85,554 cases) found OR 1.07-1.10 per SD increase in genetically predicted IGF-1, with similar effect sizes for aggressive disease (OR 1.10) (Int J Epidemiol 2022).
MR studies of breast cancer similarly support a causal relationship. An analysis combining observational data from UK Biobank (206,263 women) with MR analysis using consortium data (122,977 cases; 105,974 controls) found consistent positive associations between IGF-1 and breast cancer risk (Ann Oncol 2020).
IGF-1 and Sarcopenia: Possible Causal Protection
Unlike the null findings for cognition, MR evidence suggests that higher IGF-1 may be causally protective against sarcopenia (age-related muscle loss). This finding is consistent with the biological role of IGF-1 in muscle protein synthesis and may explain why GH supplementation improves body composition (lean mass) even when it fails to improve functional outcomes like strength.
Summary: MR Evidence Challenges the Supplementation Rationale
The MR evidence creates a challenging picture for GH/IGF-1 supplementation as an anti-aging strategy:
No causal effect of IGF-1 on dementia risk, suggesting supplementation would not prevent cognitive decline
Causal increase in cancer risk (colorectal, prostate, breast) with elevated IGF-1, suggesting supplementation carries long-term harm
Possible causal protection against sarcopenia, which may explain body composition benefits but does not translate to functional improvement
The combination suggests a poor risk-benefit ratio for IGF-1-raising interventions in healthy aging adults
Sermorelin and GHRH Analogs
GHRH analogs (sermorelin, tesamorelin) stimulate endogenous GH release by binding pituitary GHRH receptors, preserving physiological pulsatile secretion and somatostatin feedback regulation. This contrasts with direct HGH, which produces a pharmacological profile that bypasses autoregulation and can cause supraphysiological IGF-1 levels (Clin Interv Aging 2006). Sermorelin was FDA-approved in 1997 for pediatric GH deficiency (discontinued 2008 due to manufacturing issues) and remains available through compounding pharmacies.
Tesamorelin, a 44-amino acid GHRH analog FDA-approved for HIV-associated lipodystrophy, provides the strongest cognitive outcome data for this drug class. In a randomized, double-blind, placebo-controlled trial, 152 adults (including 66 with MCI, ages 55-87) received tesamorelin 1 mg/day for 20 weeks (Arch Neurol 2012). Results showed significant improvement in executive function (p=0.005), with IGF-1 increasing 117% while remaining within physiological range. A neuroimaging substudy demonstrated increased GABA levels in posterior cingulate, dorsolateral prefrontal, and parietal regions (JAMA Neurol 2013).
Integrating the Evidence
The relationship between GH/IGF-1 and aging initially appears contradictory: mouse genetic data shows reduced signaling extends lifespan, while somatopause in humans is associated with decline. However, when examined carefully, the human genetic and MR data actually align with mouse findings, and the apparent contradiction dissolves.
Human Genetic Data Supports Reduced Signaling
The strongest human evidence comes from genetic studies, which consistently support the mouse findings that reduced GH/IGF-1 signaling is protective. Ashkenazi centenarians are enriched for IGF-1R variants conferring partial receptor resistance. Laron syndrome patients with complete GH receptor loss show near-complete protection from cancer and diabetes, with diseases appearing 20-30 years later than in relatives. These genetic findings suggest that, as in mice, reduced signaling confers protection in humans.
Association Is Not Causation: The Somatopause Question
The natural decline in GH/IGF-1 with age (somatopause) is associated with muscle loss, cognitive decline, and frailty. However, this association does not establish that somatopause causes these problems. The decline could be a consequence of aging rather than a cause, could be a protective adaptation, or could simply be correlated with aging through other factors.
Importantly, the RCT evidence for GH supplementation is weak. Despite decades of trials, direct HGH administration consistently fails to improve functional outcomes. The 2007 meta-analysis of 18 RCTs found no improvements in strength, physical function, bone density, or cognition despite clear body composition changes. If somatopause were truly causing age-related decline, restoring GH/IGF-1 levels should reverse that decline. The failure of supplementation trials to show functional benefits argues against a simple causal relationship between declining hormones and declining function.
As detailed in the Mendelian Randomization section above, MR studies provide strong evidence against a causal role for low IGF-1 in dementia. The 2018 Neurology study found no evidence that genetically predicted IGF-1 affects Alzheimer’s disease risk. This suggests that the observational associations between low IGF-1 and cognitive decline likely reflect reverse causation rather than low IGF-1 causing dementia. Meanwhile, MR studies consistently show that elevated IGF-1 causally increases cancer risk for colorectal, prostate, and breast cancers.
Taken together, the evidence tells a coherent story: genetic reduction in GH/IGF-1 signaling is protective (centenarian data, Laron syndrome, mouse models), elevated IGF-1 causally increases cancer risk (MR data), low IGF-1 does not cause dementia (MR data), and pharmacological restoration does not improve function (RCT data). The implication is clear: the age-related decline in GH/IGF-1 is a correlate of aging rather than a cause, supplementation is unlikely to reverse functional decline, and raising IGF-1 levels carries real cancer risk without functional benefit.
Several biological factors help explain why the somatopause is associated with decline without causing it, and why interventions have limited benefit.
A few caveats about translating mouse findings to humans are worth noting. Developmental reduction in GH/IGF-1 signaling (as in dwarf mice or Laron syndrome) triggers compensatory stress-resistance mechanisms that do not occur with age-related decline, which may explain why genetic reduction is protective while pharmacological restoration is not beneficial. Additionally, laboratory mice die primarily of cancer (70-90%), so the anti-cancer effects of reduced IGF-1 signaling may disproportionately benefit mice compared to humans, who die from a broader disease spectrum. The dramatic 40-70% lifespan extensions in mice also do not translate to humans: Laron syndrome patients show healthspan extension but normal lifespan. Finally, adult hippocampal neurogenesis (which IGF-1 supports in rodents) appears minimal in adult humans, so some cognitive mechanisms may not translate.
Interpreting the U-Shaped Mortality Curve
The U-shaped mortality curve in humans (with optimal IGF-1 at 120-160 ng/mL from observational data) suggests that both extremes are associated with increased mortality. However, the MR evidence helps interpret this curve: the association between low IGF-1 and mortality likely reflects reverse causation (frail, sick individuals have low IGF-1), while the association between high IGF-1 and mortality likely reflects true causal harm (elevated IGF-1 causes cancer). This asymmetry has important implications.
If low IGF-1 is a marker of poor health rather than a cause, then raising IGF-1 in frail individuals would not be expected to improve outcomes, which is consistent with the RCT data showing no functional benefit from GH supplementation. Conversely, if high IGF-1 causally increases cancer risk (as MR data confirms), then interventions that raise IGF-1 carry real long-term harm. The centenarian phenotype (reduced IGF-1 signaling but maintained tissue function) suggests that the goal should not be achieving a specific IGF-1 level, but rather maintaining adequate tissue function while avoiding supraphysiological signaling.
Reconciling the Tesamorelin Cognitive Benefit
The tesamorelin trial showing improved executive function appears to contradict the conclusion that GH/IGF-1 supplementation lacks functional benefit. Several considerations help reconcile this finding. First, it is a single trial requiring replication; the history of promising single trials in this field (including Rudman 1990 and TRIIM) counsels caution. Second, the cognitive effects may operate through mechanisms distinct from the body composition effects—the neuroimaging substudy showed GABA modulation, suggesting a neuromodulatory effect that may not require supraphysiological IGF-1 levels. Third, short-term improvement in executive function does not establish long-term dementia prevention, which is what the MR evidence specifically addresses. Fourth, GHRH analogs preserve physiological pulsatility and feedback regulation, potentially conferring different effects than direct HGH.
That said, the tesamorelin data represents the strongest evidence for cognitive benefit from any somatotropic intervention in humans. If replicated, it would suggest that GHRH analogs may have a role in cognitive enhancement distinct from direct HGH, possibly through preserved physiological signaling patterns or GABA-mediated mechanisms. This remains an open question requiring further investigation.
Limitations of Current Knowledge and Future Directions
Despite decades of research, significant gaps remain in our understanding of GH/IGF-1 and human aging. Acknowledging these limitations is essential for interpreting the evidence and identifying priorities for future research.
Key Limitations
Trial duration and endpoints: Most RCTs of GH supplementation have been short (weeks to months) with body composition rather than functional or clinical endpoints. No trial has been powered to detect effects on dementia incidence, cardiovascular events, or mortality. The longest follow-up data comes from observational studies with inherent confounding.
Mendelian randomization caveats: While MR provides causal inference stronger than observational association, it has limitations. The genetic variants used as instruments may have pleiotropic effects (influencing outcomes through pathways other than IGF-1), potentially biasing results. MR estimates lifelong genetically-determined IGF-1 levels, which may differ from effects of pharmacological manipulation in older adults. The null finding for IGF-1 and Alzheimer’s disease is informative but does not exclude effects on other cognitive domains or non-AD dementias.
Population heterogeneity: Responses to GH/IGF-1 may vary by baseline IGF-1 level, age, sex, genetic background, and health status. Most trials have studied relatively healthy elderly volunteers; effects in those with documented low IGF-1 or specific deficiency syndromes may differ. The optimal approach for a 50-year-old with low-normal IGF-1 may differ from that for an 80-year-old with frailty.
Limited data on GHRH analogs: While tesamorelin has cognitive trial data, long-term safety data for anti-aging applications is lacking. Whether the preserved physiological pulsatility of GHRH analogs confers meaningful safety or efficacy advantages over direct HGH remains unproven. Cancer risk data specific to GHRH analog use in healthy older adults does not exist.
Practical Implications
The evidence reviewed above leads to clear practical conclusions. Reduced GH/IGF-1 signaling is protective (genetic evidence), low IGF-1 does not cause dementia (MR evidence), elevated IGF-1 causally increases cancer risk (MR evidence), and GH supplementation does not improve functional outcomes (RCT evidence). These findings do not support pharmacological interventions aimed at raising IGF-1 levels in healthy older adults.
Interpreting IGF-1 Levels
The observational U-shaped mortality curve (lowest mortality at 120-160 ng/mL) should be interpreted cautiously. The association between low IGF-1 and mortality likely reflects reverse causation (frail or sick individuals have low IGF-1), while the association between high IGF-1 and mortality reflects true causal harm (cancer). This asymmetry means that raising low IGF-1 would not be expected to improve outcomes, while raising IGF-1 into the high range carries real risk. IGF-1 testing may be useful for diagnosing pathological GH deficiency from pituitary disease, but pursuing a specific IGF-1 target for anti-aging purposes is not supported by the evidence.
Lifestyle Versus Pharmacological Approaches
Lifestyle interventions should focus on supporting overall health rather than targeting IGF-1 levels: resistance training for muscle and bone health, adequate protein intake (1.0-1.2 g/kg), sleep optimization, and reducing chronic inflammation. These interventions have direct functional benefits independent of any effect on IGF-1. Notably, caloric restriction and fasting (which reduce IGF-1) have shown longevity benefits in animal models, consistent with the protective effect of reduced signaling.
Pharmacological GH or GHRH analog use for anti-aging purposes is not supported by the evidence. As detailed in the Human Clinical Trials section, RCTs consistently show body composition changes without functional benefit, and as detailed in the MR section, elevated IGF-1 causally increases cancer risk. The American Geriatrics Society Beers Criteria recommend against GH use in older adults except for documented pituitary disease. Common adverse effects from GH administration include edema, arthralgias, carpal tunnel syndrome, and impaired glucose tolerance. For individuals with documented GH deficiency due to pituitary disease, replacement therapy is appropriate; in this context, the goal is normalizing deficient hormone levels, not augmenting normal aging.
Summary
Do: Resistance training, adequate protein, sleep optimization, inflammation reduction
Consider: IGF-1 testing and endocrinology referral only if pituitary disease is suspected
Avoid: Pharmacological GH/secretagogue use for anti-aging; pursuing IGF-1 targets
Recognize: Low IGF-1 in healthy older adults is a marker, not a cause, of decline
Conclusion
The optimal strategy remains uncertain. The observational data suggests maintaining IGF-1 within a moderate range (approximately 120-160 ng/mL), but this must be weighed against the genetic evidence favoring reduced signaling, the weak RCT evidence for supplementation benefits, and the MR evidence for cancer risk at higher levels. The centenarian phenotype (with genetically reduced IGF-1 signaling but maintained tissue function) may provide a biological template, but replicating this pharmacologically has proven difficult. For most healthy older adults, lifestyle interventions (resistance training, adequate protein and sleep, inflammation reduction) represent a more favorable risk-benefit approach than pharmacological supplementation. Any pharmacological intervention should be reserved for documented deficiency states and undertaken with careful monitoring and awareness of the limited evidence base.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso