Get a CLU
Biomarker testing for Clusterin (CLU) and a therapy targeting it for cognitive decline (new study)
A new study published in Nature Drug Discovery from researchers at UCLA showed that a promising drug targeting Clusterin (CLU) protects against cognitive decline and Alzheimer’s in mice.
This is the first in a series of posts about the genes and proteins that we are testing for at NeuroAge and why they are important. We test for a one letter genetic typo in CLU as part of our Alzheimer’s genetic resilience score and also indirectly test for CLU levels in our blood biomarker test. This testing contributes to our assessment of brain aging and also is important for the personalized therapeutics that we will be able to recommend in the future.
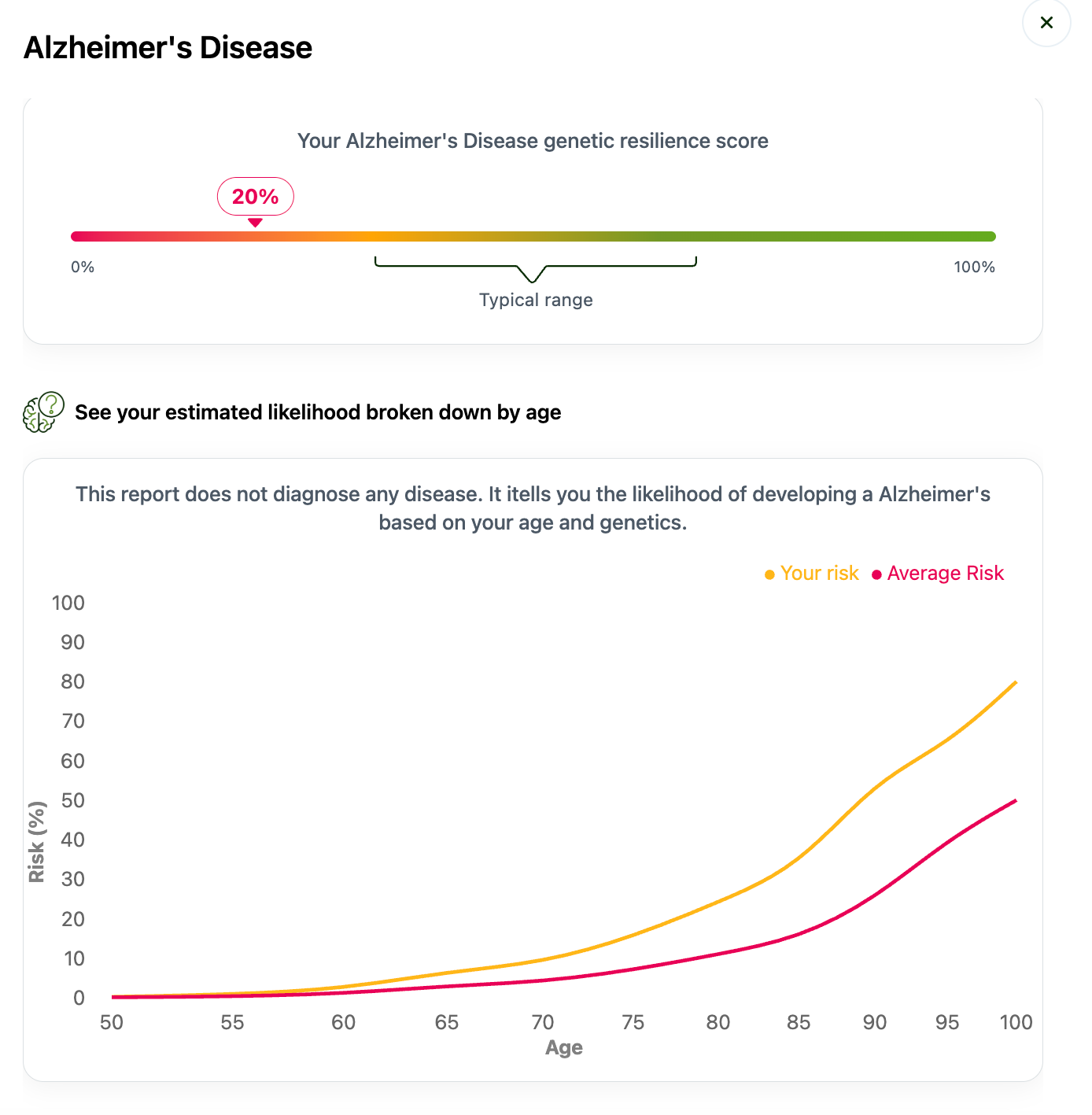
At NeuroAge we test for 41 different typos in genes that together make up people’s risk score for Alzheimer’s Disease. By contrast, 23andMe only tested for one typo in one gene, APOE4. You can think of our testing like a weighted average of all your risk genes that combine into a single score, known as a polygenic risk score (PRS). It’s a bit more complicated than that but this is the general idea. You can see demo data for the genes we test for here.
If you have had your NeuroAge genetics resilience score assessed, you can also see whether you have the CLU typo or not.
What is Clusterin (CLU)?
Clusterin, also known as APOJ, is one of the genes with typos that contributes to the Alzheimer’s score. If you recognize APOJ as being similarly named to APOE, you have made a correct leap. APOE is the single biggest risk gene for most forms of Alzheimer’s disease, accounting for about 30% of the risk. People with an APOE4 typo have 2-8x the risk of Alzheimer’s as people without it. The other 70% of Alzheimer’s genetic risk is accounted for by 40 other typos in genes.
Both APOE and APOJ are apolipoproteins (APO), which are components of spherical “vehicles” (chylomicrons) that transport lipids and cholesterol in the blood stream and throughout the body.
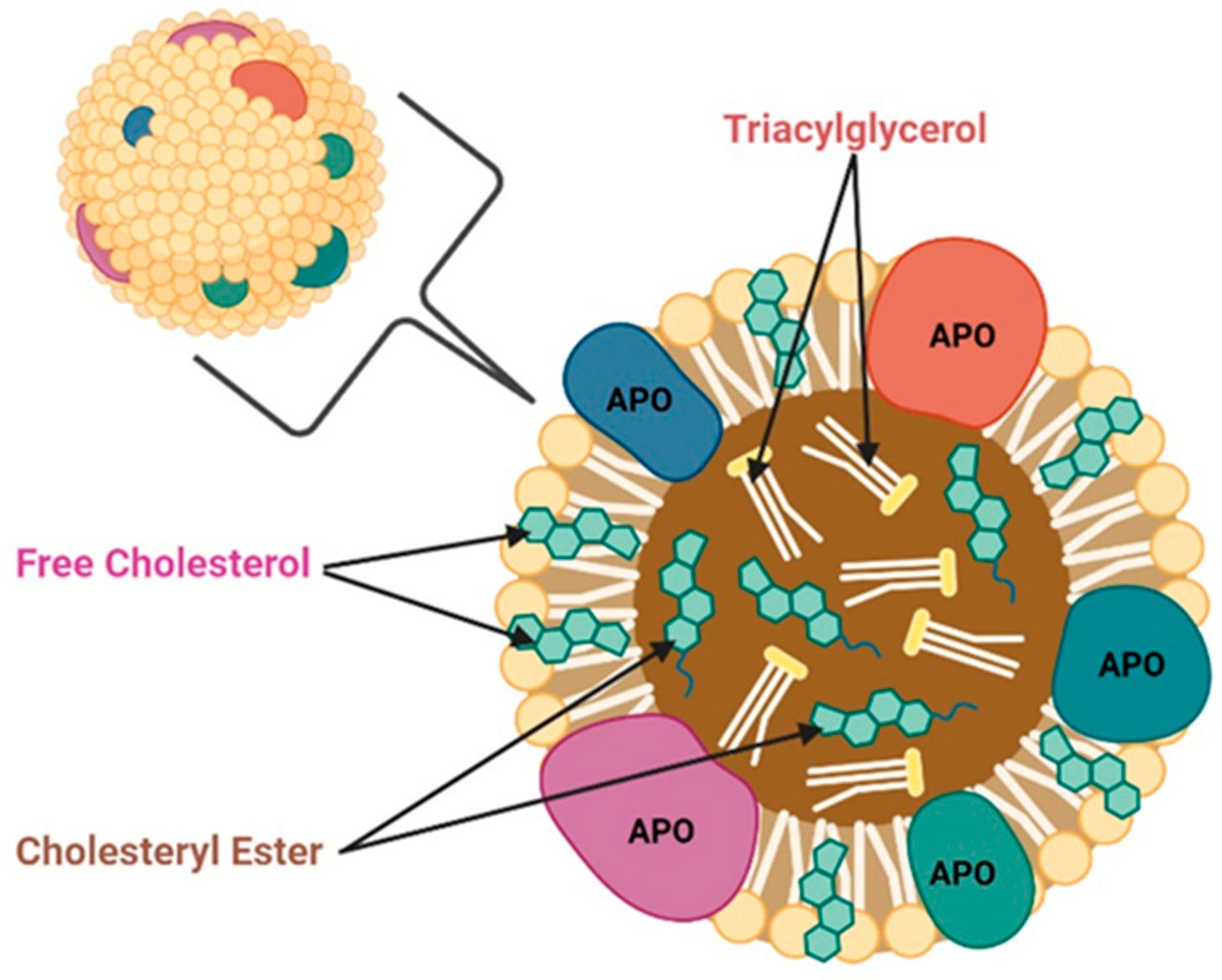
Some people may also be familiar with other components of this transport system, APOB and Lp(a), as these are commonly tested for in yearly doctor’s visits to track cardiovascular health. This is yet another connection between heart health and brain health. What’s good for the heart is good for the brain, as the saying goes.
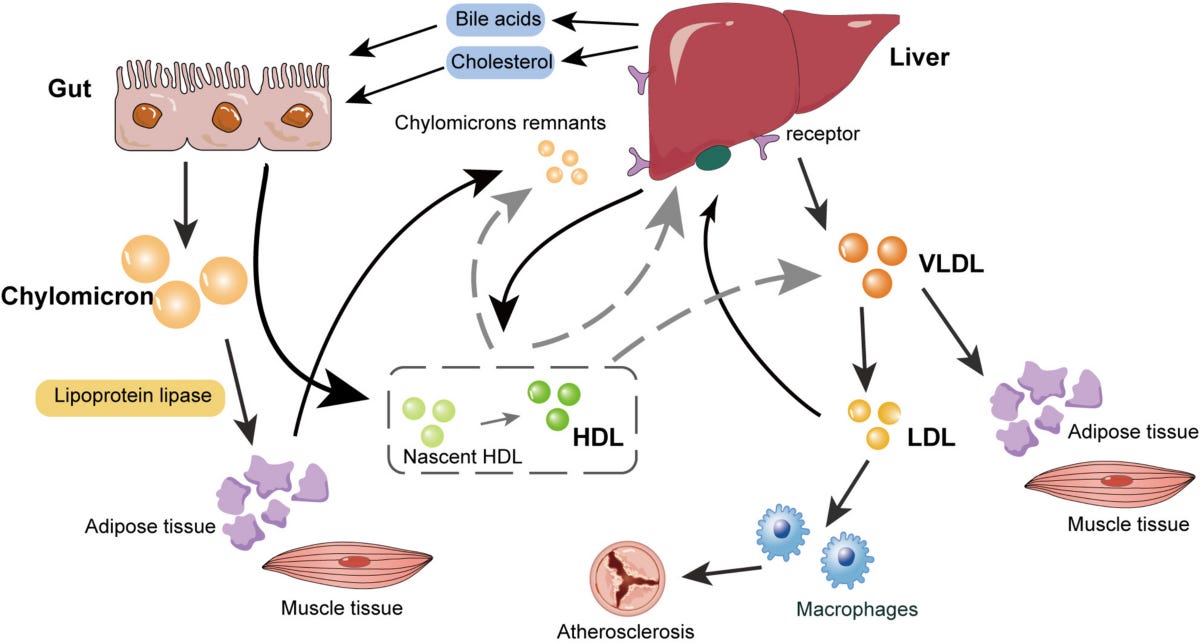
APOJ (Clusterin) is a lesser known cousin of APOE and APOB and has particular functions in the brain. It is produced by brain immune cells, astrocytes and microglia, and is found in the same toxic protein aggregates (amyloid plaques), that are known risk factors for Alzheimer’s disease.
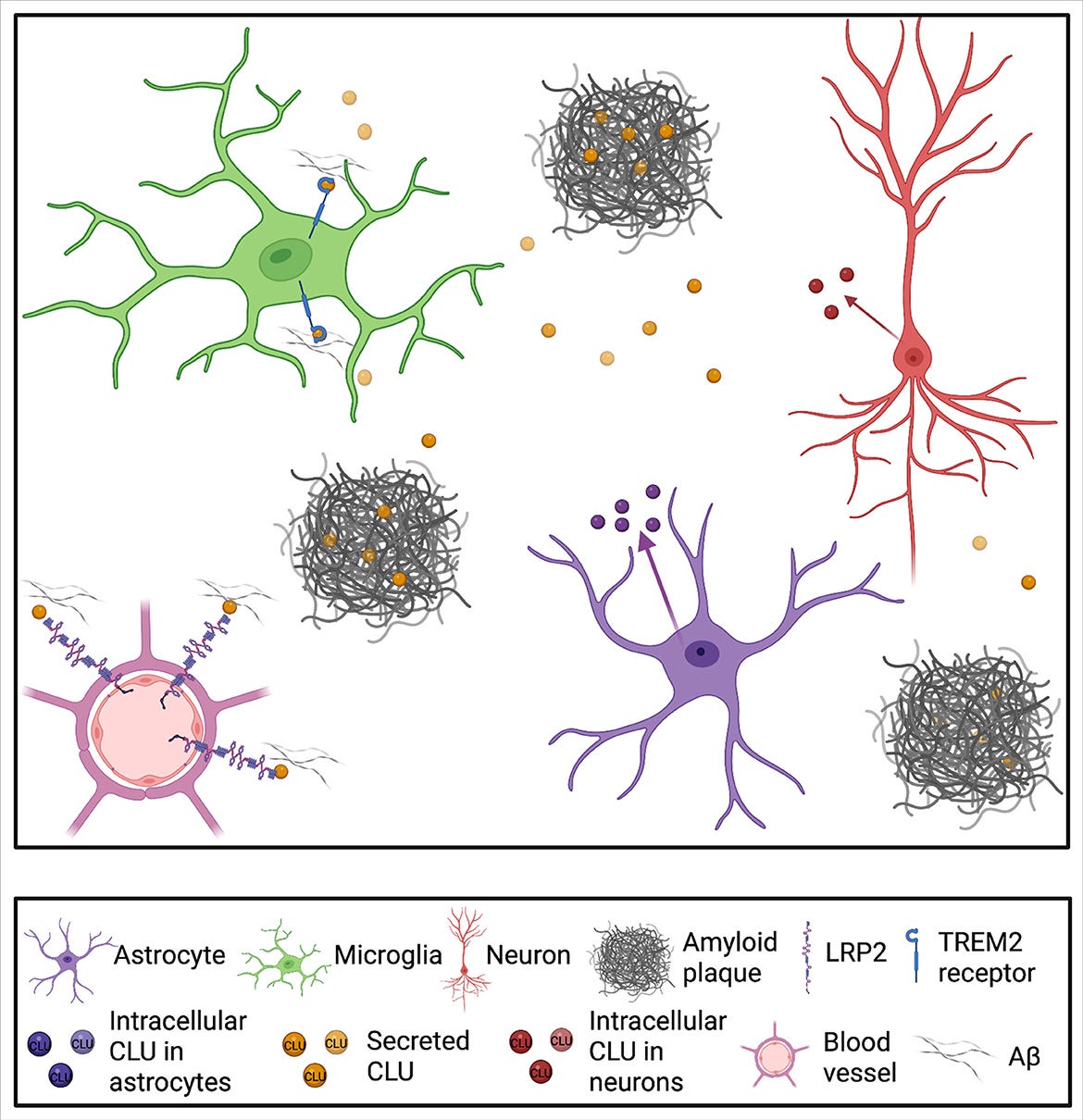
Increased levels of CLU are found in the brains of people with Alzheimer’s Disease, but it is not totally clear whether this is protective or problematic. CLU also has other functions throughout the body and there are multiple types of CLU protein, which may contribute in different ways to Alzheimer’s.
People with typos in their CLU protein have less of one type of CLU, secreted CLU (sCLU), than people without the typo. sCLU is thought to protect against amyloid plaque build up.
How does the new CLU targeting drug work?
In a new study, researchers at UCLA identified a small molecule (pill form) that modifies epigenetics and leads to increased sCLU expression. When they administered this drug to mice, they showed improvements in their memory and less p-Tau (toxic protein that builds up in Alzheimer’s) in their brains (see figure below).
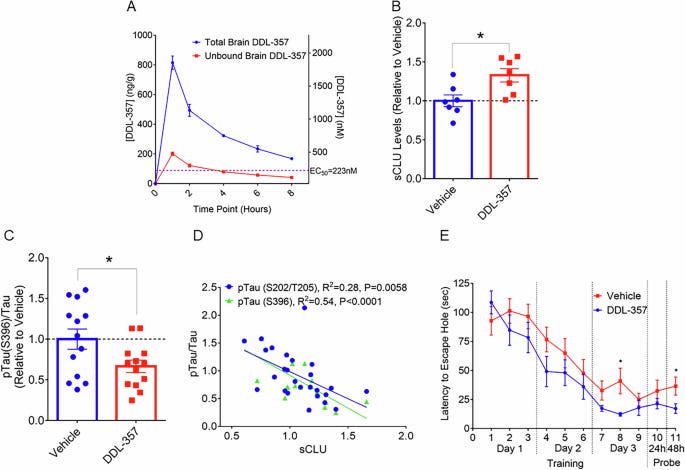
There are two main caveats to this study. The most important one is that mice are poor models for Alzheimer’s and aging of the mouse brain and human brain are very different. In particular, protein levels of many genes increase or decrease with age in opposite directions in mice and humans. This has been large hurdle for therapies tested in mice succeeding in clinical trials. I would like to see human brain organoid studies or studies in dogs or primates to confirm the direction and significance of the findings in mice.
The other caveat is that while this drug was identified by screening for sCLU levels, it does not exclusively target sCLU. It has off-target effects as well, potentially leading to less or more of other proteins. The authors point to this as a positive in the discussion, as it may target other helpful pathways. I don’t disagree with this assessment but it does muddy the waters in terms of understanding the drug’s mechanism.
Are we creating drugs to target CLU at NeuroAge?
In our own research CLU is what we are calling an “expression-regulator”, which means that RNA levels of CLU predict people’s brain age in our AI-based platform. CLU is about #500 on our list of most significant expression regulators (p<0.00001), with less CLU being associated with younger brain ages. Because it’s not in the top 50 and because the biological mechanism and directionality are a little unclear, we are not designing drugs to target CLU ourselves.
I hope that this drug succeeds and would be excited to partner with its creators to offer it to people who may benefit once it has passed testing in clinical trials.
Testing for CLU levels and genetic typos is important for Alzheimer’s risk assessment and possibly for other diseases as well. It also shines a light on who may benefit the most from future CLU therapeutics. We may in the future see CLU testing become as common as APOB testing.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso