Dietary Fat and the Brain: What the Science Actually Says About Dementia Prevention
Type of fat, cheese guide, fish guide, facts about EVOO
Your brain is roughly 60% fat by dry weight. It is the fattiest organ in your body. So it should come as no surprise that the fat you eat plays a major role in whether your brain thrives or deteriorates as you age. What might surprise you is how much controversy still exists about which fats help, which fats harm, and how much fat is optimal for long term brain health.
I’ve been on a personal journey with this topic. As many of you know, I practice intermittent fasting and I was recently tipped into functional ketosis by extending my fasting windows. My biomarker results showed a worrying increase in ApoB (85→108), LDL, and CRP. That sent me down a rabbit hole into the dietary fat and dementia literature, which I wrote about in my article Keto or Ket-no for Brain Health?. This article is the companion deep dive focused specifically on dietary fat itself: how much, what type, and what the evidence actually supports.
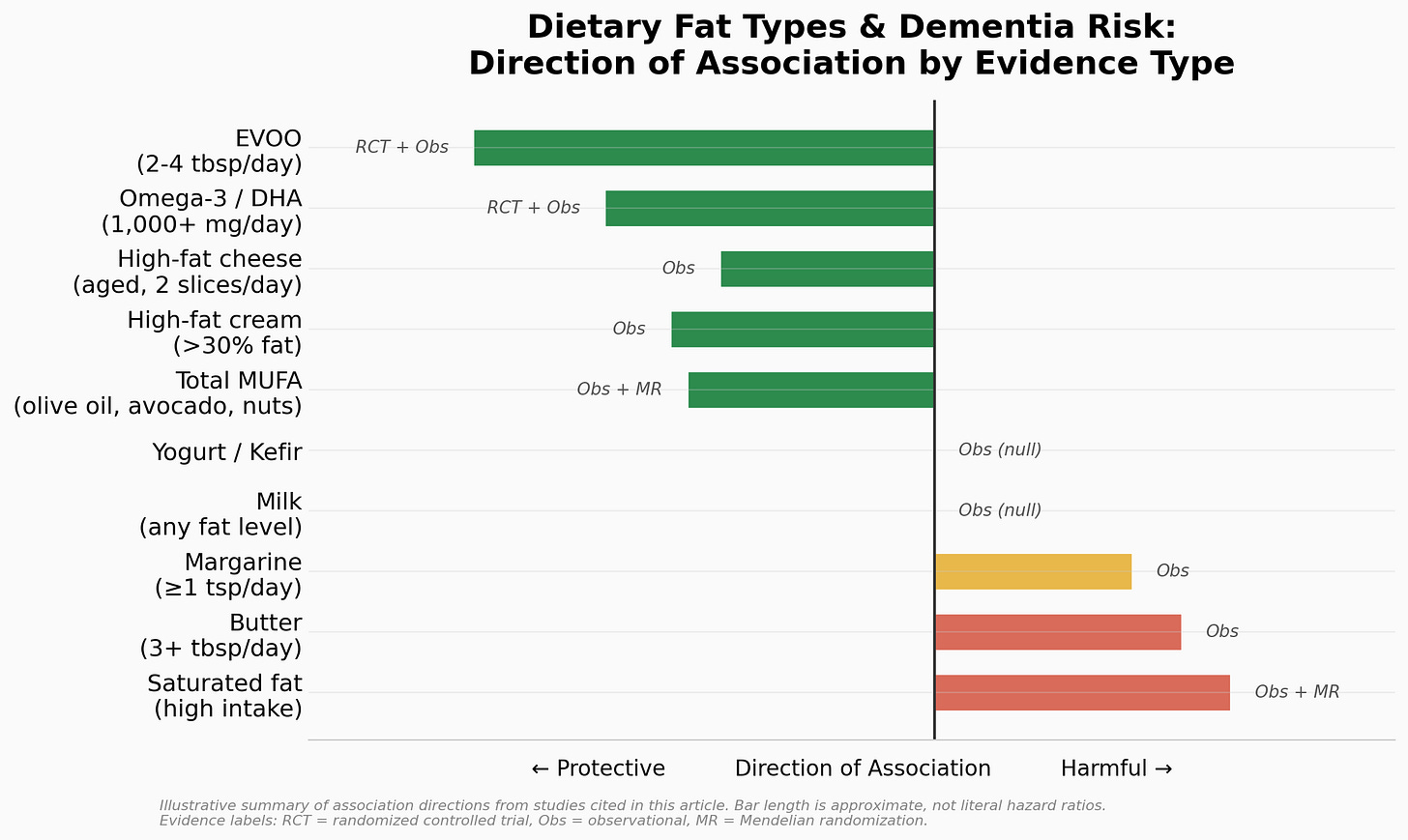
A caveat to be aware of is the evidence quality varies dramatically depending on which fat question you’re asking. Some claims are supported by large, well designed randomized controlled trials (RCTs). Others rest entirely on observational data, which can tell us about associations but not causation. I’ll walk through each category and be transparent about where the science is strong, where it’s suggestive, and where we’re still guessing.
Before we get into the evidence for each fat type, here is a practical framework for how to think about your daily fat intake for brain health. The standard USDA recommendation is 20 to 35% of calories from total fat. But the Mediterranean diet, which is the dietary pattern most consistently associated with lower dementia risk, runs closer to 35 to 40% of calories from fat. The difference is not the amount of fat but where it comes from. The AHA recommends up to 15% of calories from monounsaturated fat (MUFA), 8 to 10% from polyunsaturated fat (PUFA), and less than 10% from saturated fat (or less than 7% if you have elevated cardiovascular risk).
What does this look like in actual grams? On a 2,000 calorie diet with a brain-health-optimized Mediterranean fat profile of roughly 35 to 40% of calories from fat:
Total fat: roughly 78 to 89 grams per day. This is not a low fat diet. Your brain needs fat. The goal is to fill this budget with the right kinds.
Monounsaturated fat (MUFA): 33 to 44 grams per day (15 to 20% of calories). This is the largest single category and should be your dominant fat source. Three tablespoons of extra virgin olive oil alone provides about 30g of MUFA. Add half an avocado (7g) and a handful of almonds (9g) and you are well above the minimum. MUFA from olive oil is the fat type with the strongest brain health evidence across both observational and RCT data.
Polyunsaturated fat (PUFA): 18 to 22 grams per day (8 to 10% of calories). This includes both omega-6 (from nuts, seeds, and vegetable oils) and omega-3 (from fatty fish and fish oil). Within this category, aim for at least 1 to 2 grams of EPA+DHA from omega-3 sources daily. The omega-3 target is the most brain-specific number in this entire framework.
Saturated fat: no more than 13 to 22 grams per day (less than 7 to 10% of calories). If you have elevated ApoB, LDL, or cardiovascular risk factors, aim for the lower end. If you are an APOE4 carrier, err on the side of caution. Within this budget, prioritize saturated fat from cheese (which behaves differently metabolically than the same fat from butter or red meat, as I’ll discuss below) over saturated fat from processed foods, baked goods, and cooking with coconut oil or butter.
These are not rigid prescriptions. They are informed targets based on the convergence of cardiovascular guidelines, Mediterranean diet research, and the brain-specific evidence I’ll walk through in detail below. The single most important takeaway is the ratio: your fat intake should be dominated by MUFA (olive oil, avocado, nuts), supplemented with adequate PUFA (especially omega-3s from fish), with saturated fat kept as a relatively small fraction of total fat intake.
Total Fat Intake: The U Shaped Curve
Let’s start with the most basic question: how much total fat should you eat for brain health?
A 2025 prospective cohort study from the Health and Retirement Study followed nearly 6,000 participants for a median of 6 years and found that total fat intake had a nuanced relationship with dementia risk. Participants consuming the highest levels of vegetable fat (median intake of 23.5% of total energy) had a 31% lower risk of developing dementia compared with those consuming the lowest levels. But this protective effect was specific to the type of fat, not total fat per se.
A large Swedish study tracking nearly 28,000 people for 25 years reported something that adds to this complexity: a U shaped association between total fat intake and dementia risk. This means that both very low fat and very high fat diets may increase risk, with moderate intakes in the right forms being protective.
Evidence quality: Observational only. There are no RCTs that have randomized people to different total fat intake levels and followed them for dementia outcomes over years. The PREDIMED trial, which I’ll discuss below, came closest but was really testing a dietary pattern rather than a specific fat percentage. The U shaped curve is an interesting observation but needs to be interpreted cautiously because it comes from cohort data where total fat intake is confounded by food quality, socioeconomic status, and countless other lifestyle variables.
Saturated Fat: The Most Consistent Harmful Signal
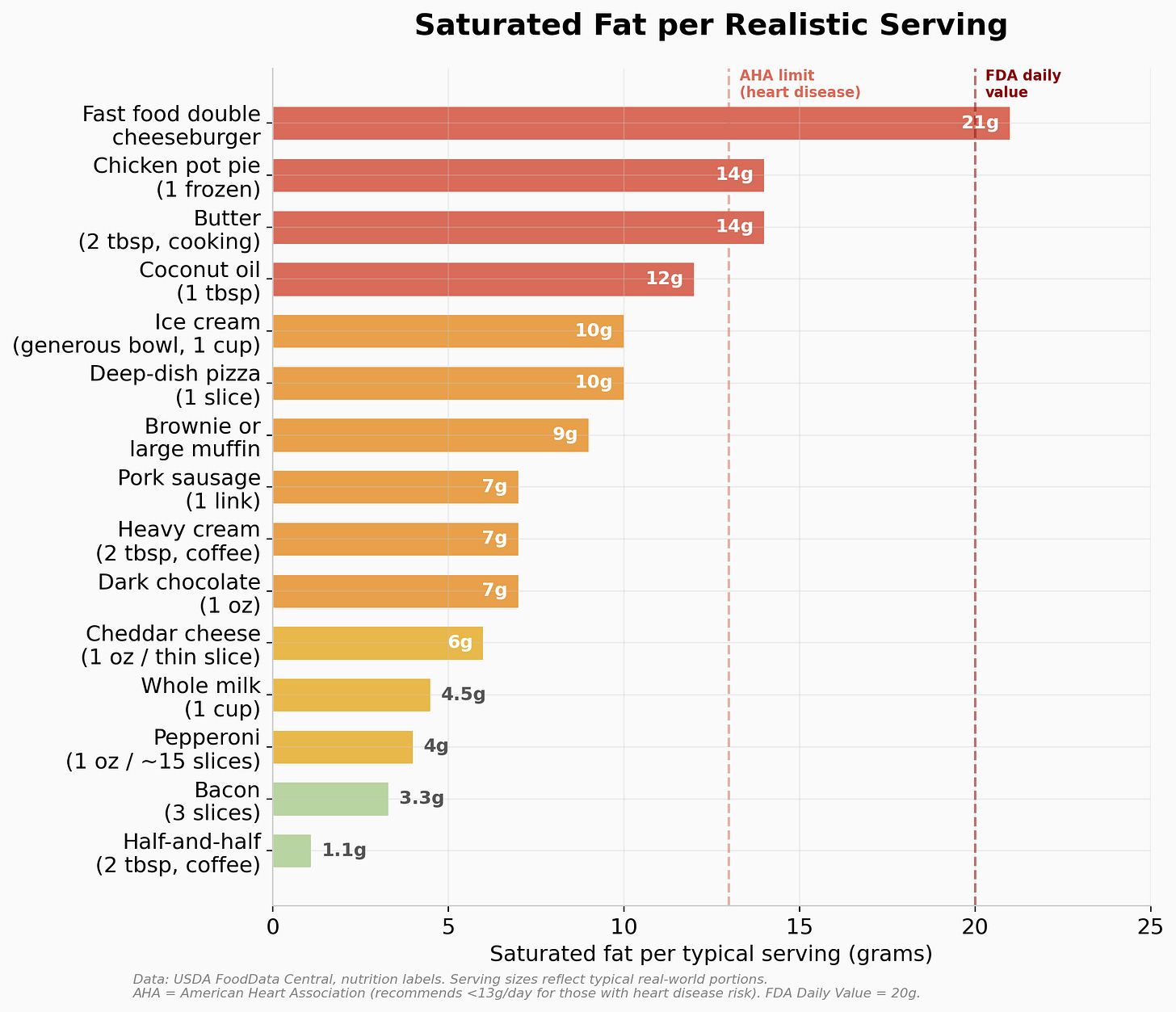
If there is one area where the observational evidence is fairly consistent, it’s that higher saturated fat intake is associated with increased dementia risk. But the source of saturated fat matters a lot, as I’ll discuss in the cheese section below. First, where is saturated fat actually hiding in realistic portions?
A few things jump out from this chart. A single fast food double cheeseburger can exceed the AHA’s entire daily limit for someone with heart disease risk. Coconut oil, which had a moment in the keto world, delivers 12g of saturated fat per tablespoon, nearly double what butter provides. And the “sneaky” sources (ice cream, baked goods, heavy cream in coffee) add up fast without people noticing. Meanwhile, three slices of bacon (3.3g) and a splash of half-and-half (1.1g) are relatively minor players despite their bad reputations. Context matters.
A meta-analysis of prospective cohort studies found that higher dietary saturated fat was associated with a 39% increased risk of Alzheimer’s disease and a 105% increased risk of all cause dementia. In dose response analysis, each 4 g/day increment of saturated fat (roughly a pat of butter or a single tablespoon of coconut oil) was linked to a 15% higher AD risk.
A 2025 Mendelian randomization study from Peking University added stronger causal evidence. Using genetic variants as instruments (which reduces the confounding that plagues observational studies), the researchers found that total dietary fat intake, especially saturated fat, contributed to AD risk independently of other nutrients. Mendelian randomization is not the same as an RCT, but it is a significant step up from standard observational data because it uses genetic variation as a natural experiment.
There’s also the metabolic context to consider. A 2025 study in middle aged adults found that saturated fat intake moderated the relationship between triglyceride levels and memory performance. For people with elevated triglycerides, cutting back on saturated fat appeared to be especially important for protecting cognitive function.
Evidence quality: Strong observational evidence plus Mendelian randomization support, but still no direct RCT evidence testing saturated fat restriction specifically for dementia prevention. The 2025 Dietary Guidelines Advisory Committee systematic review, which examined 83 articles including one RCT and 82 observational studies, concluded with moderate confidence that dietary patterns lower in saturated fat are associated with reduced cognitive decline. But note: the one RCT in that review (the MIND diet trial) did not show significant cognitive benefits compared to control. This is a case where the observational evidence is fairly strong and consistent but the limited RCT data has not confirmed it.
Monounsaturated Fats and Olive Oil: Strong and Possibly Independent Protection
The evidence for monounsaturated fats (MUFAs), particularly from extra virgin olive oil (EVOO), is among the most encouraging in the dietary fat and brain health literature.
A large prospective cohort study from the Nurses’ Health Study and the Health Professionals Follow Up Study (92,383 participants, 28 years of follow up) found that consuming more than half a tablespoon per day of olive oil was associated with a 28% lower risk of dementia related death. This held regardless of overall diet quality, which is notable because it suggests olive oil has benefits beyond simply being a marker of eating a Mediterranean diet. The substitution analysis was also compelling, replacing just 5 g/day of margarine or mayonnaise (about a teaspoon) with olive oil was associated with an 8 to 14% reduction in dementia mortality risk.
How Much Olive Oil?
The evidence converges on a range. The Nurses’ Health Study found that as little as half a tablespoon per day was enough to see a 28% reduction in dementia mortality. But the strongest evidence comes from the PREDIMED trial, the landmark RCT of 7,447 participants at high cardiovascular risk, where participants in the EVOO arm received 1 liter per week and were advised to consume at least 4 tablespoons per day. After 6.5 years, the EVOO group had significantly better cognitive performance across memory and fluency tasks compared to the low fat control diet. In a PREDIMED substudy of 522 participants, those allocated to Mediterranean diet plus EVOO had significantly higher Mini-Mental State Examination and Clock Drawing Test scores after 6.5 years of intervention. The European Food Safety Authority recommends a minimum of roughly 1.5 tablespoons per day for LDL protection and cholesterol optimization.
So the practical range is a minimum of 1 to 2 tablespoons daily for meaningful benefit, with 3 to 4 tablespoons being the dose tested in the best available RCT. Most people will land comfortably around 2 tablespoons as a realistic daily target. And quality matters enormously. It needs to be extra virgin, ideally a high polyphenol variety (look for “high phenolic” on the label, or ask your specialty store). That peppery throat burn when you swallow it straight? That is oleocanthal, and it is the sensory indicator that your oil actually contains the compounds that matter.
A quick note on cooking with olive oil, because I get this objection all the time. The claim that you “can’t cook with olive oil because of the smoke point” is one of the most persistent myths in nutrition, and it is flatly wrong. A quality EVOO has a smoke point of 350 to 410°F, which comfortably exceeds the 250 to 350°F range used for most home cooking including sautéing, roasting, and even pan frying. But more importantly, a landmark 2018 study comparing ten common cooking oils found that smoke point does not actually predict an oil’s safety or stability under heat. What matters is oxidative stability, and EVOO outperformed every other oil tested, including oils with far higher smoke points like canola and grapeseed, producing the lowest levels of harmful polar compounds and trans fats. The reason is straightforward. EVOO’s high monounsaturated fat content (primarily oleic acid) resists oxidation, and its polyphenols act as built in antioxidants that protect the oil during heating. UC Davis food scientists have confirmed that polyphenols and monounsaturated fats from EVOO actually transfer into cooked food, and that vegetables cooked in EVOO may contain more antioxidants than the same vegetables eaten raw. The entire Mediterranean basin has been cooking with olive oil for thousands of years. The populations with the lowest rates of dementia and cardiovascular disease on earth are not drizzling it on cold salads and calling it a day. They are frying eggs in it, roasting vegetables in it, and sautéing fish in it. Use it for everything.
Why Does Olive Oil Work? Five Distinct Mechanisms
This is where it gets really interesting, because EVOO is not just a “healthy fat.” It operates through at least five distinct neuroprotective mechanisms, and critically, some of these are unique to EVOO and have nothing to do with its MUFA (oleic acid) content.
Oleocanthal acts like ibuprofen in your brain. This phenolic compound inhibits COX-1 and COX-2 enzymes through the same mechanism as ibuprofen, reducing chronic neuroinflammation. But unlike ibuprofen, you can take it every day for decades without destroying your gut lining. Preclinical studies show it also directly increases clearance of amyloid beta plaques across the blood brain barrier and stimulates their degradation.
Polyphenols block both hallmarks of Alzheimer’s pathology. EVOO contains over 35 phenolic compounds, including oleuropein aglycone, hydroxytyrosol, and oleacein. Together, these disrupt amyloid beta aggregation AND inhibit tau hyperphosphorylation, meaning they interfere with the formation of both plaques and tangles. In mouse models, EVOO enriched diets improved behavioral performance, reduced amyloid accumulation, and enhanced autophagy (the cell’s garbage disposal system for damaged proteins).
Blood brain barrier protection. A small RCT at Auburn University (25 participants with MCI, 2 tablespoons of EVOO daily for 6 months) found that EVOO specifically enhanced BBB function and improved functional connectivity between brain regions. Refined olive oil improved memory activation but did NOT improve BBB integrity. This is a key distinction. The MUFA content helps, but the polyphenols do something additional and mechanistically different.
Hydroxytyrosol activates your cellular defense system. This compound upregulates Nrf2, a master transcription factor that switches on a cascade of antioxidant and detoxification enzymes, including HO-1. It also triggers mitochondrial biogenesis (making new, healthy mitochondria) and improves endoplasmic reticulum stress responses. Both tyrosol and oleuropein convert to hydroxytyrosol after ingestion, making it the central active metabolite of EVOO’s phenolic compounds.
Gut microbiome modulation. Emerging evidence shows EVOO at doses of 3 to 4 tablespoons per day beneficially alters gut microbiota composition, which is relevant because gut brain axis signaling via neuroinflammation is increasingly recognized as a contributor to neurodegeneration.
The critical takeaway here is that refined olive oil provides oleic acid (MUFA), which has modest benefits on its own. But EVOO provides oleic acid PLUS a pharmacological cocktail of polyphenols that cross the blood brain barrier and directly interfere with Alzheimer’s pathology. The polyphenol content of EVOO is roughly four times higher than that of refined olive oils. They are not the same product.
Evidence quality: Strong observational data supported by one major RCT (PREDIMED) and a small proof of concept trial. This is actually one of the better evidence profiles in nutrition research. The PREDIMED trial, despite its well documented randomization issues, provides genuine interventional evidence that a Mediterranean diet rich in EVOO improves cognitive outcomes. The observational data from multiple large cohorts is consistent and shows a dose response relationship. I’d rate this as probably the strongest evidence we have for any specific dietary fat intervention for brain health.
Omega 3 Fatty Acids: The Best Studied, Most Nuanced Story
Omega 3s, particularly DHA and EPA, are the most extensively studied dietary fats in relation to brain health. The brain preferentially accumulates DHA in neurons and synapses, and DHA makes up a substantial portion of neuronal membrane phospholipids. So there is strong biological plausibility for a protective effect.
The Observational Evidence
The observational evidence for omega 3s is robust. A 2023 meta-analysis in the American Journal of Clinical Nutrition incorporating 48 longitudinal studies and over 103,000 participants found that dietary intake of omega 3 fatty acids reduced the risk of all cause dementia or cognitive decline by approximately 20%. DHA intake showed the strongest association. Long term omega 3 supplement users in the ADNI (Alzheimer’s Disease Neuroimaging Initiative) cohort exhibited a 64% reduced risk of AD.
The dose response relationship is meaningful and graded. Each increment of just 100 mg/day of DHA or EPA (less than what you would get from a single standard fish oil capsule) was associated with an 8 to 10% lower risk of cognitive decline. Blood biomarker data supported this, with elevated plasma EPA and erythrocyte membrane DHA both associated with lower cognitive decline risk.
The mechanisms are multi-layered and well-characterized, and understanding them helps explain both why the observational data is so strong and why the RCTs have struggled. DHA and EPA operate through different but complementary pathways, and this is important because most people think of “omega 3s” as a single thing. They are not.
How DHA Protects the Brain: Four Mechanisms
DHA IS your brain’s membrane. DHA is the most abundant omega 3 in the brain, concentrated in neuronal membranes where it maintains membrane fluidity, modulates ion channel function, and supports synaptic signaling. Without adequate DHA, membranes become rigid and neurotransmission degrades. This is not an abstract biochemical concern: membrane fluidity directly affects how well your neurons communicate, and it deteriorates with aging.
Glymphatic clearance: helping your brain take out the trash. This is one of the most compelling mechanisms. DHA preserves AQP4 (aquaporin-4) polarity in astrocytes, which is essential for the glymphatic system to function. The glymphatic system is the brain’s waste disposal network, operating primarily during sleep to flush amyloid beta and other metabolic waste through cerebrospinal fluid channels. In mouse models, omega-3 supplementation markedly inhibited astrocyte activation and protected AQP4 polarization in affected brain regions. Critically, this effect was abolished in AQP4 knockout mice, confirming that DHA’s amyloid clearance benefits are specifically mediated through the glymphatic pathway. This connects directly to the sleep literature. Omega 3s may improve sleep architecture and reduce nocturnal neuroinflammation, indirectly supporting the very clearance mechanism they enhance.
Blood brain barrier transport. DHA upregulates LRP-1 (Lipoprotein Receptor-related Protein 1) in mice, a key efflux transporter on the BBB that actively pumps amyloid beta out of the brain and into the bloodstream. It also restores tight junction proteins (ZO-1) that maintain BBB structural integrity, which starts deteriorating early in AD pathology. About 75% of amyloid clearance from the brain occurs via BBB transport, making this a major clearance route that DHA helps keep functional.
Reduces amyloid production at the source. DHA increases LR11 (Lipoprotein Receptor 11) in brain cell membranes, which diverts amyloid precursor protein (APP) away from the secretases that chop it into toxic amyloid beta fragments. So DHA simultaneously reduces production AND increases clearance, working both sides of the amyloid equation.
DHA also supports synaptic plasticity by regulating BDNF (brain derived neurotrophic factor), which is essential for learning, memory, and the maintenance of existing neural circuits.
How EPA Protects the Brain: The Anti-Inflammatory Player
While DHA is the structural and clearance workhorse, EPA serves a distinct and complementary role.
Specialized pro-resolving mediators (SPMs). EPA serves as the substrate for producing resolvins and other bioactive lipid mediators that actively resolve inflammation rather than just suppressing it. This is a crucial distinction as EPA does not just block inflammatory signaling the way NSAIDs do. It generates molecules that actively clean up the aftermath of inflammation, restoring tissue to its pre-inflammatory state.
Suppresses pro-inflammatory cytokines. EPA directly reduces hippocampal IL-1β, interferon-γ, and protects against amyloid beta induced neuroinflammation, potentially mediated through PPARγ (peroxisome proliferator-activated receptor gamma) activation.
A Note on Omega-3 Form and Brain Uptake
One more thing worth knowing is that the form of omega 3 matters for brain uptake. LPC-bound (lysophosphatidylcholine) forms of DHA appear to have preferential access to the brain compared to standard triglyceride or ethyl ester forms found in most fish oil supplements. This may particularly matter for APOE4 carriers, who show altered DHA transport across the BBB. Krill oil naturally contains a mix of phospholipid bound omega 3s, and there are newer LPC enriched supplements entering the market (like LYSOVETA from Aker BioMarine), though clinical trials in dementia patients are still needed to confirm whether this translates to meaningful cognitive benefits.
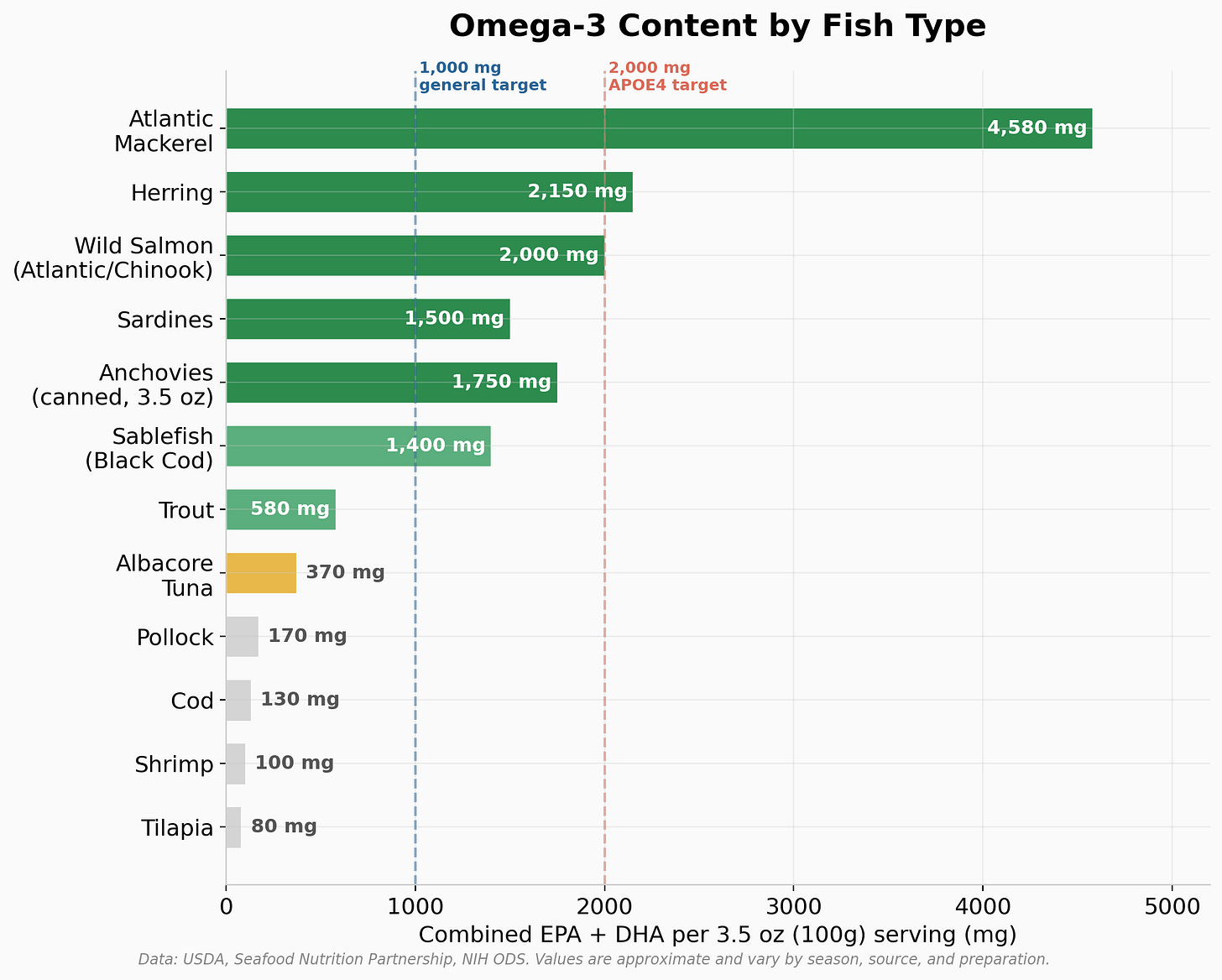
A Practical Fish Guide: How Much Do You Actually Need to Eat?
Not all fish are created equal when it comes to omega 3 content. The difference between the best and worst choices is enormous, and knowing this can help you plan realistically. Here is what a standard palm sized serving (about 3.5 ounces or 100 grams, cooked) delivers in combined EPA+DHA:
The omega 3 powerhouses (1,500+ mg per serving): Atlantic mackerel leads the pack at roughly 4,500 mg per serving, making it the single most concentrated commonly available source. Wild salmon (Atlantic, Chinook, or Coho) provides around 1,500 to 2,150 mg. Herring delivers about 2,150 mg. Sardines come in at roughly 1,500 mg. These are the fish that move the needle.
Solid contributors (500 to 1,500 mg per serving): Anchovies, sablefish (black cod), trout, and albacore tuna fall in this range. These are worthwhile but you would need to eat them more frequently to hit your targets.
Not worth relying on for omega 3s (under 500 mg per serving): Cod, tilapia, shrimp, catfish, mahi mahi, lobster, and scallops. These are perfectly healthy lean proteins, but if brain health omega 3s are your goal, they do not deliver meaningful amounts. You would need to eat an unrealistic quantity of cod or tilapia to match what one serving of salmon provides.
So what does this mean in practical terms?
For the general target of 1,000 mg EPA+DHA daily, eating one serving of wild salmon, mackerel, herring, or sardines three to four times per week gets you most of the way there. On non-fish days, a standard fish oil supplement (providing 500+ mg EPA+DHA per capsule) fills the gap.
For APOE4 carriers targeting 2,000 mg/day, this is harder to hit with food alone unless you eat fatty fish nearly every day. A realistic approach is eating salmon or mackerel four times per week (providing roughly 1,500 to 2,000 mg on those days) plus taking two to three high concentrate fish oil capsules daily (providing 600 to 1,000 mg each).
One practical note on mercury: the highest omega 3 fish also tend to be lower in mercury. Salmon, sardines, herring, anchovies, and trout are all low mercury choices that you can eat frequently without concern. The fish to limit for mercury reasons (swordfish, king mackerel, bigeye tuna, shark) are not even among the best omega 3 sources. Atlantic mackerel (as opposed to king mackerel) is both high in omega 3s and low in mercury. So the brain health optimal fish list and the mercury safe list overlap almost perfectly.
The RCT Evidence: Where Things Get Complicated
The RCT evidence for omega 3 supplementation and cognition is mixed.
A 2025 dose response meta-analysis of 58 RCTs found that each 2,000 mg/day of omega 3 supplementation (roughly the amount in a large serving of wild salmon, or 4 standard fish oil capsules) was associated with significant improvements in attention and perceptual speed, with moderate to low certainty evidence. That is encouraging but not overwhelming.
A Cochrane review of omega 3 supplementation for treatment of existing dementia found no clear benefit, though it noted the trials were generally short and the doses may have been inadequate.
A 2025 overview of systematic reviews found a small but positive overall effect of omega 3 supplementation on reducing cognitive decline. Interestingly, the analysis found no significant dose response relationship, suggesting a possible threshold effect rather than a linear dose response: benefits may occur once minimum effective levels are reached, with higher doses not necessarily producing superior outcomes.
Several factors likely explain the disconnect between the strong observational data and the mixed RCT results. First, most trials are short (3 to 6 months) for a disease that develops over decades. Second, many trials used doses below 1,000 mg/day (just two standard fish oil capsules), which may have been insufficient for brain effects, particularly in APOE4 carriers. Third, most trials enrolled people who already had significant cognitive impairment, at which point the window for prevention may have closed. Fourth, few trials measured baseline omega 3 status. If participants already had adequate levels, supplementation would show no added benefit.
This last point is worth dwelling on, because the mechanism data explains it precisely. DHA’s neuroprotective effects depend on intact membrane dynamics, functional AQP4 polarization, and responsive transcriptional machinery, all of which are severely compromised in late stage Alzheimer’s. Animal models confirm that DHA supplementation reverses cognitive deficits only when administered BEFORE significant amyloid deposition or synaptic loss. In humans, benefits appear limited to early stages or MCI, while trials in moderate to severe AD show minimal effects. This strongly supports a preventive rather than therapeutic role for omega 3s, and it explains exactly why so many RCTs “failed”: they were testing a prevention strategy in people who already had advanced disease. You cannot unclog the glymphatic system if the astrocytes that run it are already too damaged to respond.
Evidence quality: Very strong observational evidence with biological plausibility, supported by mixed RCT evidence that is getting stronger with better trial design. The totality of evidence supports omega 3 intake (particularly DHA) as protective, but the optimal supplementation strategy for prevention is not yet established by RCTs. The gap between epidemiological and interventional evidence likely reflects trial design limitations rather than a true absence of effect. More recent and better designed trials with longer duration, higher doses, and enrollment of people at genetic risk but without symptoms are needed.
The Cheese Paradox: Not All Saturated Fat Sources Are Equal
This is one of the more surprising findings in the recent literature, and it challenges a simple “saturated fat = bad for brain” narrative.
A 25 year prospective cohort study from Lund University, published in Neurology in December 2025, tracked nearly 28,000 Swedish adults and found that consumption of 50 g/day or more of high fat cheese (that is about two thin slices or a piece the size of a small matchbox; think cheddar, Brie, Gouda) was associated with a 13% lower risk of all cause dementia and a 29% lower risk of vascular dementia compared with those eating less than 15 g/day (barely half a slice). High fat cream (greater than 30% fat) at 20+ g/day (about a tablespoon and a half) was associated with a 16% lower risk of all cause dementia.
Here is what makes this particularly interesting: low fat cheese, low fat cream, milk of any fat level, butter, and fermented milk (yogurt, kefir) showed no association with dementia risk at all. Butter at high intake levels (40+ g/day, or about 3 tablespoons) was actually associated with increased AD risk. So this is not a generic “dairy is good” or “fat is good” story. Something specific about high fat cheese appears to be driving the association.
A separate Japanese cohort study (JAGES 2019 to 2022) in older adults found a similar pattern with habitual cheese consumption of one or more times per week associating with modestly lower dementia incidence over three years after propensity score matching.
So why would cheese be protective while butter from the same animal is not?
The lead author Emily Sonestedt from Lund University explained that cheese is a whole food with a complex food matrix where fat, protein, minerals, and bioactive compounds are packaged together in ways that likely affect how the body metabolizes the fat. Several mechanisms may be at play:
The fermentation factor. Cheese is a fermented food, and fermentation produces bioactive compounds not found in unfermented dairy. Lactic acid bacteria during cheese production synthesize vitamin K2 (menaquinone), B vitamins including folate and B12, and bioactive peptides with anti-inflammatory and antioxidant properties. The vitamin K2 content of cheese is partly determined by fat content and starter culture composition, which may help explain why high fat cheese shows associations that low fat cheese does not: fat soluble vitamins like K2 are concentrated in the fat fraction. MK-4 is the predominant form of vitamin K2 in the brain (more than 98%), and preclinical data suggests it has neuroprotective, antioxidant, and anti-inflammatory properties, though the direct evidence for K2 supplementation improving cognition in humans remains limited.
Bioactive peptides. Cheese fermentation and aging generate peptides that may reduce inflammatory cytokine release, modulate neurotransmitter activity, and in experimental models reduce amyloid plaque buildup. Soft ripened cheeses like Camembert and Brie produce oleamide (a fatty acid amide linked to memory performance in animal studies) through their white mold cultures. Aged cheeses like Parmesan contain gamma-glutamyl peptides. These are not present in butter or milk.
The dairy matrix effect. When fat is consumed within the cheese matrix (bound to protein and calcium in a complex structure), it appears to be metabolized differently than the same fat consumed as butter or cream. RCT evidence from Raziani’s 2018 study found that consuming regular fat cheese did not raise blood lipid levels the way equivalent amounts of butter did. Mendelian randomization studies have also linked cheese (but not butter) with lower risk of diabetes and hypertension, both of which are major dementia risk factors.
Source quality may matter too. What a cow eats determines what is in the milk, and that determines what is in the cheese. Dairy from grass fed cows tends to contain higher levels of omega 3 fatty acids and conjugated linoleic acid (CLA), both of which have independent anti-inflammatory properties.
Evidence quality: Observational only, no RCTs on cheese and dementia specifically. The Swedish study is impressive in its size and duration (28,000 people, 25 years), and the APOE4 interaction analysis adds biological plausibility. But as the authors noted, the observational design limits causal inference. The fact that the association strengthened when they excluded dementia cases in the first 10 years of follow up argues against reverse causation (sick people eating less cheese). But residual confounding is always possible. In Sweden, higher cheese intake correlated with higher education and income, and while the researchers adjusted for these factors, unmeasured lifestyle differences could still play a role.
My take: I would not start eating large quantities of cheese purely based on this data, but I also think the MIND diet’s recommendation to limit cheese to once per week may be too conservative, especially for non-APOE4 carriers. A moderate intake of high quality, fermented, full fat cheese (particularly aged varieties rich in vitamin K2 and bioactive peptides) appears to be at worst neutral and possibly protective. The key insight is that the food matrix matters enormously. The saturated fat in cheese does not behave in the body the same way the saturated fat in butter does, and our dietary recommendations need to catch up with that nuance.
A Practical Cheese Guide: Which Cheeses Deliver the Goods?
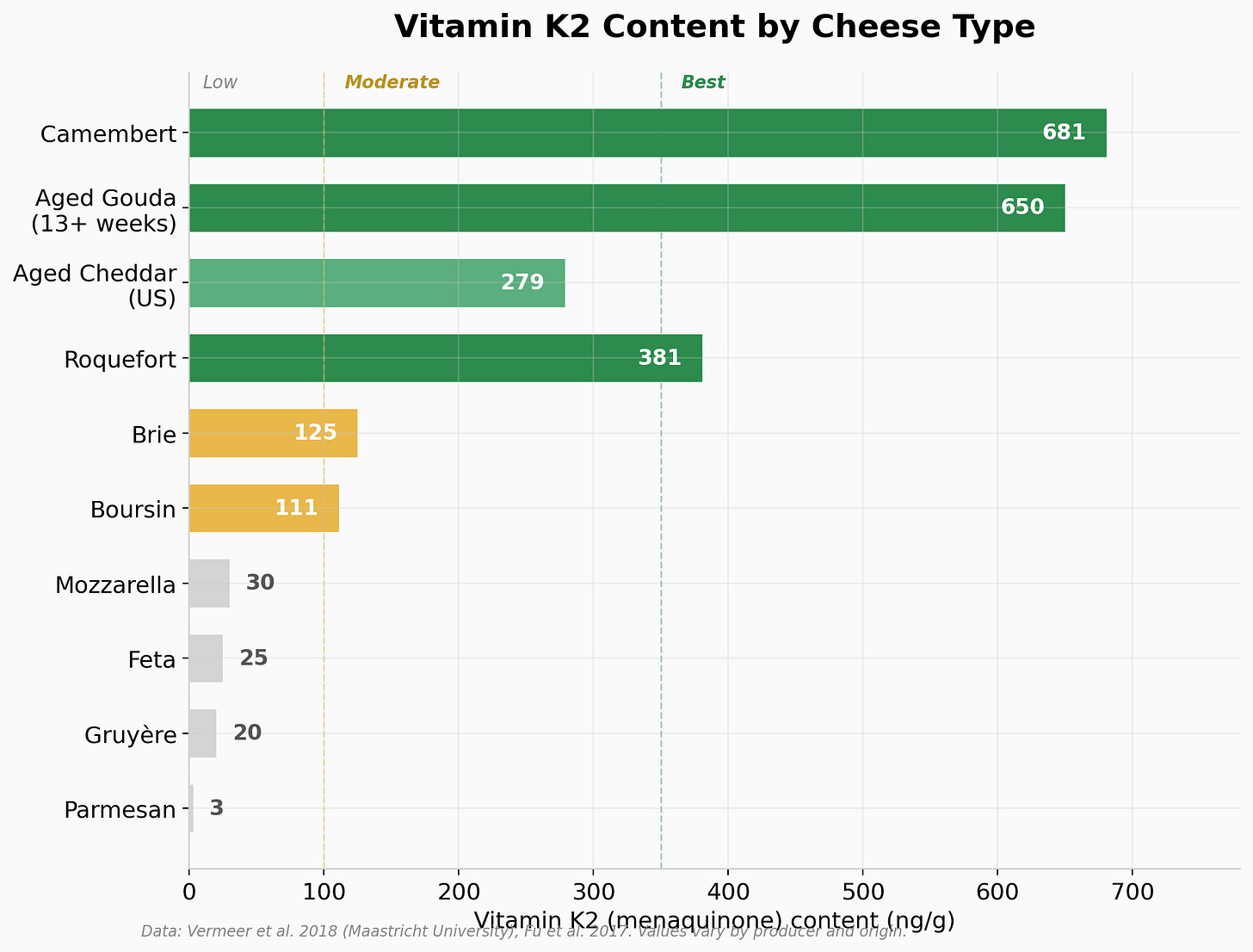
Not all cheese is created equal for brain health purposes. The key compounds driving the protective association (vitamin K2, bioactive peptides, the concentrated food matrix) vary dramatically by cheese type. The two biggest determinants are the bacterial cultures used (mesophilic cultures produce far more K2 than thermophilic ones) and the length of aging (longer fermentation generates more bioactive peptides and concentrates fat soluble nutrients).
The chart below ranks cheeses by vitamin K2 content because that is the most quantifiable differentiator we have good lab data for. But an important caveat is that K2 is not the whole story. The epidemiological studies that found cheese to be protective measured total cheese consumption, not K2 specifically. Several other bioactive compounds in cheese likely contribute to the association, and they do not perfectly track with K2 content. Bioactive peptides with anti-inflammatory and ACE-inhibitory (blood pressure lowering) properties accumulate during aging through any bacterial culture, not just K2 producing ones. Parmesan, for instance, has almost no K2 but is rich in gamma-glutamyl peptides. Spermidine, a polyamine that induces autophagy (cellular cleanup) and has been independently associated with reduced cognitive decline, is found broadly in aged cheeses. Oleamide, the fatty acid amide linked to memory in animal models, is specific to white mold cheeses like Camembert and Brie regardless of their K2 levels. And the food matrix effect (the way cheese’s protein-calcium structure changes how saturated fat is metabolized) applies to all real cheese, even fresh varieties. So think of K2 as the best available proxy for “how much fermentation chemistry happened in this cheese,” while recognizing that cheeses low in K2 may still contribute through other pathways.
The best choices: Aged Gouda (13+ weeks) is the standout, with the highest measured K2 content among commonly available cheeses, around 650 ng/g in full fat Dutch Gouda, and the K2 content increases with ripening time. Aged Edam is similar (same mesophilic bacterial cultures) and is notably high in MK-7, the long chain form with the longest half life in the body. Aged cheddar from traditional producers scores well, with US studies measuring 267 to 290 µg/100g total vitamin K. Jarlsberg has also been highlighted in research as high in K2. European Muenster is rich in MK-9. All of these share mesophilic bacterial cultures and extended aging.
Surprisingly good: Camembert scored high at 681 ng/g despite being a soft cheese, and Roquefort at 381 ng/g. The white mold and blue mold cultures used in these cheeses are strong K2 producers. Camembert also specifically generates oleamide, the fatty acid amide linked to memory performance in the animal studies mentioned above. Blue cheeses in general (Stilton, some Gorgonzola) can be high, but there is huge variability depending on the specific cultures, with one French blue cheese measuring over 700 ng/g while another measured only 50 ng/g.
Mediocre: Brie is often grouped with Camembert, but the data says otherwise. It measured only 125 ng/g, roughly five times lower than Camembert, likely due to differences in specific mold strains and aging conditions. Swiss cheese and Gruyère are relatively low in K2 because they use thermophilic (high temperature) bacterial cultures that produce little to no vitamin K2.
Low in K2 (but not necessarily useless): Fresh mozzarella, Parmesan, ricotta, cottage cheese, cream cheese, and fresh chèvre. The Maastricht University analysis specifically found that Mediterranean cheeses including mozzarella, feta, pecorino, and gorgonzola were low in menaquinone content, with Parmesan containing almost negligible amounts (3 ng/g) despite being one of the most heavily aged cheeses available. This is counterintuitive but makes sense: Parmesan uses thermophilic cultures that produce virtually no K2. Aging alone does not produce K2. You need the right bacteria. That said, Parmesan is rich in bioactive peptides and spermidine from its extensive aging, so it is not a nutritional blank. Fresh cheeses like mozzarella and chèvre have minimal fermentation time and fewer bioactive compounds overall, but they still deliver the food matrix effect that changes how their saturated fat is metabolized compared to butter. They are fine foods; they are just not the optimal choices if you are specifically trying to maximize the fermentation derived compounds that may be driving the protective association.
Goat cheese specifically depends entirely on the type. Fresh soft goat cheese is like mozzarella in this context: minimally aged, low K2, limited bioactive peptide development. But hard aged goat cheese is a different story. Swiss researchers found that K2 content was determined primarily by the bacterial cultures and aging time, not the milk source. So a well aged goat Gouda would have similar benefits to its cow milk counterpart.
One important caveat: where the cheese is made matters. Dutch Gouda tested much higher in K2 than US produced Gouda, likely due to differences in starter cultures and production traditions. If you are specifically trying to maximize K2 intake, European produced cheeses from traditional makers are likely a better bet than mass produced domestic versions.
The practical takeaway: if you want to maximize the fermentation derived compounds most likely driving the protective association, aged Gouda, Edam, aged cheddar, and Camembert are your best bets. Two thin slices per day puts you in the range the Swedish study associated with reduced dementia risk. But do not stress about occasionally eating mozzarella on a pizza or Parmesan on pasta. All real cheese delivers the food matrix effect, and many cheeses contribute bioactive compounds through mechanisms beyond K2. Variety is reasonable. Just make the high K2 aged cheeses your regular staples rather than the exception.
What About Full Fat Yogurt?
Given the cheese findings, you might assume yogurt would show similar benefits. It is fermented dairy, after all. But the data is surprisingly weak.
The Swedish Neurology study explicitly tested fermented milk (which includes yogurt, kefir, and buttermilk) at both high and low fat levels and found no association with dementia risk in either direction. A 2024 dose response meta-analysis in Advances in Nutrition found that one French study showed yogurt was associated with better verbal memory in some models, but a broader narrative review concluded that findings for yogurt “remain inconsistent” and that among fermented dairy products, cheese was “most likely responsible for the observed benefits.” One large Japanese prospective study did find a possible dose dependent protective effect for moderate yogurt intake, potentially mediated by probiotic content, but this has not been replicated consistently.
So why does cheese show a signal but yogurt does not? The most likely explanation comes down to the fermentation process itself. Cheese undergoes extended fermentation and maturation, often lasting months to years, which generates far higher concentrations of bioactive peptides, vitamin K2, and other complex compounds. Yogurt fermentation is comparatively simple and short. The bacterial cultures in yogurt (mostly Lactobacillus and Streptococcus thermophilus) produce less K2 and fewer of the complex peptides that emerge from the prolonged aging process in hard cheeses. The food matrix also differs: cheese concentrates fat soluble nutrients into a dense matrix, while yogurt retains a more dilute, liquid structure closer to milk.
This does not mean yogurt is bad for you. It has legitimate gut microbiome benefits through its probiotic content, and there are plausible gut-brain axis mechanisms by which those probiotics could support brain health over time. But the specific neuroprotective signal that appears for cheese simply does not show up for yogurt in the current literature.
And Half and Half in Your Coffee?
I get asked about this more than you might think. People who are otherwise very intentional about their diet worry about the splash of cream in their morning coffee.
Half and half is roughly 10 to 12% fat, well below the “high fat cream” threshold (greater than 30% fat) that showed a protective association in the Swedish study. The Swedish researchers did not specifically test light cream or half and half as a separate category. It would likely have been captured under the milk or low fat cream categories, neither of which showed any association with dementia in either direction.
A typical splash of half and half is around one to two tablespoons per cup. At 10 to 12% fat, that is roughly 1.5 to 3.5 grams of fat, most of it saturated. Even drinking two or three cups a day, you are looking at maybe a tablespoon or two of total fat, of which roughly half is saturated.
My honest assessment is that half and half in coffee is not something to worry about for brain health. The studies linking saturated fat to dementia risk are capturing people whose entire dietary pattern is saturated fat heavy, not people who put a tablespoon of cream in their morning coffee and then eat salmon and olive oil the rest of the day. If your olive oil intake, omega 3 intake, and overall dietary pattern are well constructed, a splash of half and half is noise, not signal. Your inflammatory markers and ApoB are being driven by much larger dietary levers than your coffee creamer.
If you wanted to optimize even this small detail, you could switch to full fat cream (which actually showed a protective association in the Swedish data, interestingly) or use it as an opportunity to add MCT oil to your coffee. But this falls squarely into diminishing returns territory.
The APOE4 Modifier: Why Genetics Matters
For my NeuroAge audience, this is critical. APOE genotype modifies the relationship between dietary fat and brain health in important ways.
An international expert consensus statement highlighted that high fat intake may exacerbate cognitive decline in APOE4 carriers, and that APOE4 carriers show altered DHA uptake and utilization patterns. Some studies suggest that APOE4 carriers may need higher omega 3 doses to achieve the same brain levels as non-carriers, potentially because of increased DHA utilization and metabolic demands. EPA may actually be lower before DHA in the pre-dementia stage in APOE4 carriers, suggesting it could serve as an early biomarker.
However, it is reassuring that the large omega 3 meta-analysis found that the protective effects remained significant even after adjusting for APOE4 status.
The Swedish dairy study also found that APOE4 status modified the association between high fat cheese and Alzheimer’s: among adults without genetic risk for Alzheimer’s, full fat cheese was associated with noticeably lower AD risk, while in APOE4 carriers the relationship was different.
The bottom line for APOE4 carriers is to be more deliberate about fat quality. Prioritize omega 3s (and possibly at higher doses), lean heavily into MUFA from olive oil and avocado, and be more conservative with saturated fat from dairy and red meat. Even the cheese data supports this distinction: the Swedish study found that high fat cheese’s inverse association with Alzheimer’s was present only among APOE4 non-carriers (p for interaction = 0.014), suggesting that APOE4 carriers may not get the same benefit and should be more cautious with saturated fat from any source, cheese included.
Bringing It Together: What I’m Doing and What I Recommend
After reviewing this literature, here is my practical synthesis, organized by evidence strength:
Strongest evidence (observational + RCT support):
Consume extra virgin olive oil daily. At minimum half a tablespoon, ideally 2 to 4 tablespoons. A good rule of thumb: use it as a generous drizzle on salads, vegetables, and finished dishes, and use it as your default cooking fat. The lower end of this range is supported by the Nurses’ Health Study (28% dementia mortality reduction), and the upper end is the dose tested in PREDIMED (the only large RCT with cognitive endpoints). Choose high polyphenol EVOO when possible. If it burns your throat a little when you taste it straight, that is oleocanthal, and that is a good sign. This is the closest thing we have to a dietary fat intervention with genuine interventional evidence for brain health, and the mechanisms (oleocanthal’s anti-inflammatory action, polyphenol mediated amyloid and tau interference, BBB protection, Nrf2 activation, and microbiome modulation) are among the best characterized of any dietary intervention.
Strong evidence (very robust observational data, emerging RCT support):
Prioritize omega 3 intake, particularly DHA. Aim for at least 1,000 mg combined EPA+DHA daily, with 750+ mg of that from DHA. To put that in food terms: a palm sized serving (about 4 ounces) of wild salmon provides roughly 1,500 to 2,000 mg of EPA+DHA, so three to four servings of fatty fish per week (salmon, mackerel, sardines, herring) gets most people close to the target, with a daily fish oil supplement (look for one providing at least 500 mg combined EPA+DHA per capsule) to fill the gap on non-fish days. If you are an APOE4 carrier, consider going higher, closer to 2,000 mg/day total omega 3. That is the equivalent of eating a serving of wild salmon nearly every day, or eating fatty fish three to four times per week plus taking two to three high concentrate fish oil capsules daily. APOE4 carriers should also consider phospholipid bound forms (like krill oil) which may have preferential brain uptake. The mechanistic case for omega 3s is compelling as DHA simultaneously maintains neuronal membrane integrity, supports glymphatic waste clearance during sleep, keeps the blood brain barrier functional, and reduces amyloid production at the source. EPA complements this by actively resolving neuroinflammation. But the key insight from the mechanism data is that these interventions work best as prevention: start early, before significant amyloid deposition occurs.
Consistent observational evidence (no direct RCT):
Minimize saturated fat. The epidemiological evidence is quite consistent, and the Mendelian randomization data provides additional causal support. Keep saturated fat well below the typical American diet level. This does not mean zero; it means choosing olive oil over butter and fish over red meat as your primary fat sources.
Suggestive evidence (observational only):
Total fat intake probably follows a U shape, with both very low fat and very high fat diets being suboptimal for brain health. A moderate fat intake (roughly 30 to 40% of calories) dominated by MUFA and omega 3s rather than saturated fat appears to be the sweet spot based on available data, but this has not been tested in RCTs.
The beautiful thing about this area of research is that the interventions are simple, affordable, and have essentially no downside. Drizzle olive oil on everything. Eat fish. Snack on walnuts. Your brain will thank you.
If you’ve had your genetics analyzed through NeuroAge, check your APOE status and lipid panel. Combined with your personal biomarkers, this information can help you and your healthcare provider develop a personalized fat optimization strategy. If brain health is your primary goal, as it is for me, getting the fat equation right may be one of the highest leverage dietary changes you can make.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso