Can Flickering Lights and Clicking Sounds Protect Your Brain from Alzheimer’s?
Reviewing the evidence for 40 Hz devices
A growing number of consumer devices now claim to boost brain health by delivering light and sound at a specific frequency, 40 Hz, or 40 cycles per second. The science behind them traces back to a single MIT lab and a series of remarkable findings in mice. The question is whether those findings hold up in humans, and whether the devices already on the market are ahead of the evidence or riding a genuine wave of progress.
Here is what we know so far.
It started with a graduate student and a hunch
Sometime around 2014, a doctoral student named Dr. Hunter Iaccarino walked into Dr. Li-Huei Tsai’s office at MIT’s Picower Institute for Learning and Memory with an idea. Dr. Tsai’s lab had been studying gamma oscillations (fast brain waves associated with attention and memory) and had figured out how to generate them in mice using a technique called optogenetics, where brain cells are genetically modified so they can be switched on by laser light delivered through a tiny fiber optic cable implanted in the brain. Nobody had tried to connect those artificially generated gamma waves to any molecular changes in the brain. Dr. Iaccarino wanted to boost gamma oscillations in Alzheimer’s model mice and measure what happened to amyloid-beta levels.
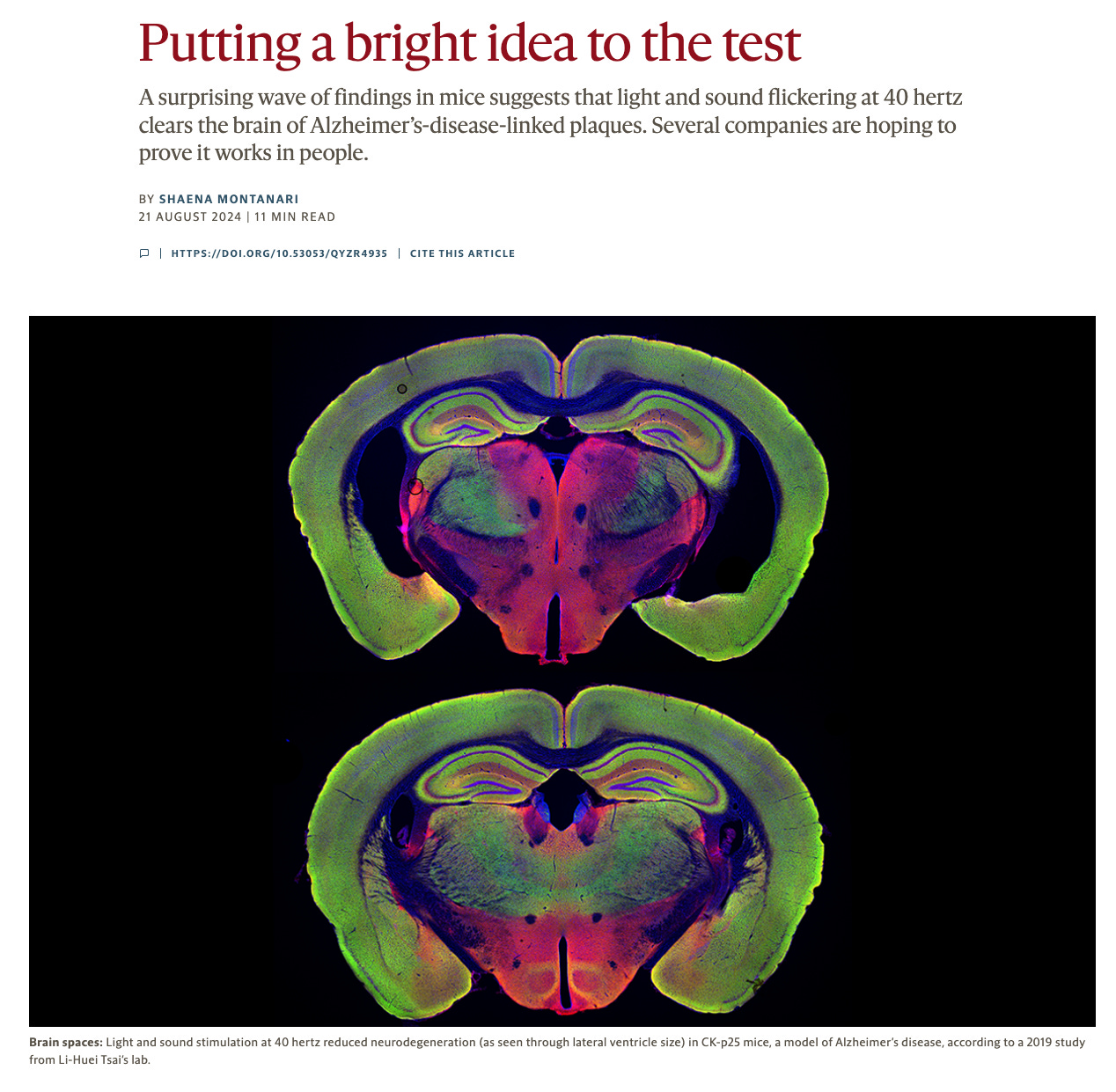
It took a single experiment. After one hour of driving gamma oscillations at 40 Hz in the hippocampus (the brain’s memory center), amyloid-beta levels dropped. According to a profile in The Transmitter, the result was so unexpected that Dr. Iaccarino ran down the hallway to show Dr. Tsai the readout.
But optogenetics requires brain surgery, which isn’t ideal for a consumer product. So the team asked whether they could get the same gamma entrainment using something external, something that did not require cutting open the skull. They tried flickering a light at 40 Hz and found that mice exposed to the strobe showed the same amyloid reduction in the visual cortex. That 2016 Nature paper launched an entirely new field of research, and within months, Dr. Tsai and MIT neuroscientist Dr. Ed Boyden co-founded Cognito Therapeutics to see if the approach could work in humans.
Your brain has a rhythm, and Alzheimer’s disrupts it
Brain cells communicate in coordinated waves of electrical activity. One type, called gamma oscillations, occurs in the 30 to 100 Hz range and is associated with attention, memory formation, and the brain’s ability to integrate information across regions. Think of it like an orchestra playing in sync. When the musicians are coordinated, the music sounds right. When they fall out of rhythm, things break down.
In people with Alzheimer’s disease and in mouse models of the disease, gamma oscillations are disrupted early and progressively. This observation raised a straightforward question. What happens if you restore the rhythm?
The mouse data
That 2016 Nature paper showed more than just an amyloid drop. The 40 Hz light stimulation also triggered microglia, the brain’s resident immune cells, to cluster around and engulf amyloid plaques. The frequency mattered, 20 Hz did not work, 80 Hz did not work, only 40 Hz produced the effect.
A 2019 follow-up in Cell added sound to the picture. Clicking tones at 40 Hz drove gamma activity in the brain’s hearing center and the hippocampus. Seven days of auditory stimulation improved spatial and recognition memory in Alzheimer’s model mice and reduced amyloid in those regions. Combining light and sound together reached brain areas that neither could reach alone, including parts of the prefrontal cortex (involved in planning and decision-making), and reduced amyloid plaques across the entire outer layer of the brain.
Since then, the MIT group and others have extended these findings across multiple Alzheimer’s mouse models, showing that 40 Hz stimulation preserves neurons, protects synapses (the connections between brain cells), reduces a modified form of tau called phosphorylated tau (the other major toxic protein in Alzheimer’s), and sustains learning and memory. Tactile stimulation, meaning vibration at 40 Hz applied through the body, has also shown similar neuroprotective effects in mice, making this a third sensory route to the same outcome.
The proposed mechanism involves the brain’s waste clearance system
For years after the initial discovery, the “how” remained unclear. A 2024 study in Nature from the same MIT group proposed an answer.
The brain has a waste disposal network called the glymphatic system, a series of channels running alongside blood vessels that flush out metabolic debris, including amyloid. (I have written about this system before in the context of sleep and sleep deprivation, since glymphatic clearance is most active during deep sleep.)
The 2024 study showed that 40 Hz light and sound stimulation increased the flow of cerebrospinal fluid (the clear fluid that surrounds and cushions the brain) into brain tissue and accelerated the drainage of interstitial fluid (the fluid between brain cells) out of the brain. When the researchers blocked this waste-clearing system, the amyloid-lowering effect of stimulation disappeared entirely. The mechanism involved a specific type of brain cell called a VIP interneuron (named for the signaling molecule it releases, vasoactive intestinal peptide). In response to 40 Hz stimulation, these cells released more of that signaling molecule, which caused nearby blood vessels to pulse more strongly. That increased pumping action drove fluid through the brain’s waste-clearing channels more efficiently.
In other words, the stimulation appears to turn up the brain’s own plumbing system to flush out toxic proteins more efficiently.
You do not need photoreceptors in the brain for this to work
A common objection online is that “light cannot penetrate the skull, so this must be nonsense.” This reflects a misunderstanding of how the therapy works.
40 Hz light therapy does not require light to reach the brain directly. The light hits your retina, just like any other visual stimulus. Light-detecting cells in the retina sense the flickering and convert it into electrical signals. Those signals travel through the optic nerve to a relay station in the center of the brain called the lateral geniculate nucleus, and then to the visual cortex (the area at the back of the brain that processes what you see). The flickering light is not “penetrating the skull” any more than watching television is.
But the visual cortex is not where Alzheimer’s does most of its damage. The hippocampus (the brain’s memory center) is. So how does a rhythm that enters through the eyes end up affecting the hippocampus? The answer is that the brain is a network. The visual cortex sends signals forward to higher processing areas in the temporal lobe, which in turn connect to the entorhinal cortex, the main input gateway to the hippocampus. The 40 Hz rhythm gets carried along those connections like a beat propagating through a chain of musicians. The auditory pathway actually has a more direct route, because the hearing center sits in the temporal lobe, anatomically close to and well-connected with the hippocampus. That is likely why the 2019 Cell paper found that sound alone could drive gamma activity in the hippocampus, while light alone mainly affected the visual cortex. Combining light and sound works better than either alone because each sensory route carries the 40 Hz rhythm into the brain through a different network of connections, and they converge on shared structures from multiple directions.
We know this propagation actually happens in humans, not just in mice. A 2025 study published in Communications Biology used brain wave recordings from electrodes implanted directly inside the brains of eleven epilepsy patients and found that 40 Hz visual stimulation successfully synchronized neural activity in the hippocampus, temporal lobe, and frontal cortex. An earlier intracranial study similarly found that adding a cognitive task during 40 Hz light stimulation increased gamma synchronization in the hippocampus and insula (a deep brain region involved in awareness and emotion), both of which are affected early in Alzheimer’s.
The legitimate scientific debate is not about whether flickering light can influence deep brain activity. It clearly can, and this has been measured with electrodes placed directly inside the human brain. The real questions are whether this externally driven synchronization engages the same brain circuits as the brain’s own naturally generated gamma oscillations, and whether the downstream benefits on amyloid clearance and cognition hold up in large clinical trials.
Human clinical data are encouraging but early
The MIT pilot trials. In a 2022 study published in PLOS ONE, Dr. Tsai’s team enrolled 15 people with mild Alzheimer’s in a randomized, controlled trial. Participants used an at-home device (a light panel and speaker) delivering 40 Hz stimulation for one hour daily. After three months, those receiving active stimulation (compared to a non-40 Hz control group) showed less brain shrinkage in the ventricles (fluid-filled spaces inside the brain that enlarge as brain tissue is lost) and hippocampus, increased communication between brain regions in a network called the default mode network (which is disrupted early in Alzheimer’s), better performance on a face-name memory test, and improved daily activity rhythms.
The two-year follow-up. Five participants continued using the devices after the trial ended. A 2025 study in Alzheimer’s and Dementia reported outcomes at the 30-month mark. The three female participants with late-onset Alzheimer’s showed slower cognitive decline compared to thousands of matched patients in national databases. In two of them who provided blood samples, a blood marker of Alzheimer’s brain pathology called pTau217 was significantly reduced. However, the two male participants with early-onset disease did not show significant cognitive benefits, and the brain volume preservation seen at three months was no longer significant at 30 months.
The Cognito Therapeutics OVERTURE trial. Cognito’s headset, called Spectris, delivers 40 Hz light and sound. Their OVERTURE trial enrolled 76 participants with mild-to-moderate Alzheimer’s in a six-month randomized, double-blind study where some participants received the real treatment and others received a fake version. An important detail that often gets buried in Cognito’s press materials is that the trial’s primary endpoint, a cognitive-functional composite score called MADCOMS, did not reach statistical significance. Neither did two key secondary endpoints, CDR-SB and ADAS-Cog14, which are standard measures used in Alzheimer’s trials. By the most conventional reading, OVERTURE was a negative trial. The measures that did reach significance were other secondary endpoints, including daily functioning (ADCS-ADL), a brief cognitive screening test (MMSE), and brain volume on MRI.
Data presented at AD/PD 2025 showed that Spectris preserved the structure of the corpus callosum, the thick bundle of nerve fibers connecting the two halves of the brain, compared to matched controls from the Alzheimer’s Disease Neuroimaging Initiative. It is worth noting that the corpus callosum is a white matter tract, not the hippocampus. Hippocampal shrinkage is the brain area of Alzheimer’s that most clinicians and researchers focus on. If the trial had shown hippocampal preservation, you would expect them to lead with that. The fact that they highlighted the corpus callosum instead suggests that hippocampal effects were either not measured, not significant, or not as strong.
Spectris has received FDA Breakthrough Device Designation, which sounds more impressive than it is. The designation is a process benefit, not a stamp of approval. It means the FDA considers the device novel enough and the disease serious enough to warrant faster review and more frequent communication with the company during development. It does not lower the bar for actual approval. Cognito received the designation in early 2021, based on preliminary data, before the full OVERTURE results were published. HOPE’s primary endpoints are functional (ADCS-ADL) and cognitive (MMSE) measures, not brain volume. Brain volume preservation alone would not be sufficient for FDA approval in mild-to-moderate Alzheimer’s.
The HOPE pivotal trial. Cognito recently completed enrollment of 670 participants in HOPE, a Phase 3 trial across 70 U.S. sites, with a 12-month treatment phase followed by a 12-month period where all participants (including those previously on the fake treatment) receive the real device. Topline results are expected in 2026. This trial will be the definitive test of whether 40 Hz sensory stimulation works as a disease-modifying treatment in humans.
Two main devices exist, at very different stages
Cognito Spectris is a medical-grade headset with opaque goggles and headphones, available only through clinical trials. Cognito Therapeutics has raised approximately $423 million in venture funding since 2016, including a $73 million Series B in 2023 led by FoundersX Ventures (with participation from the Alzheimer’s Drug Discovery Foundation, Starbloom Capital, IAG Capital, and WS Investment Company) and a $196 million Series C in early 2025. Dr. Rick Kuntz, former Chief Medical and Scientific Officer at Medtronic, sits on the board. A key feature is that every participant’s brain wave response is verified by EEG before they begin using the device, confirming that 40 Hz stimulation is actually producing the intended effect in that individual. Users sit still with eyes closed during the one-hour sessions. If the Phase 3 trial succeeds, Spectris would likely become available by prescription.
OptoCeutics EVY is a consumer device classified as an FDA general wellness product (not a medical device). It was co-founded in 2018 by Dr. Mai Nguyen and Dr. Jes Broeng, a DTU (Technical University of Denmark) professor, after a collaboration between DTU light researchers and brain scientists at UC Berkeley. The company is much earlier stage than Cognito, having raised approximately $3 million in angel and seed funding from investors including Kaare Danielsen and Sebastian Scripps, along with EU grants. The company is headquartered in Copenhagen and has about nine employees.
EVY uses a patented technology called Invisible Spectral Flicker, which alternates between two shades of white light that are close enough in tone that the flickering is barely perceptible. This is a meaningful comfort improvement over a raw 40 Hz strobe, which most people find unpleasant and which can trigger seizures in susceptible individuals. OptoCeutics has a randomized controlled trial ongoing in Denmark, and smaller safety and feasibility studies have shown that the device successfully induces gamma oscillations and is well tolerated.
In practice, EVY is a compact, portable light panel that you plug into a wall outlet and place on a flat surface 1.5 to 3 feet away. The light needs to be in your field of vision, but you do not need to stare directly at it. You go about your normal activity for at least one hour, eat breakfast, drink coffee, read, watch TV, or work at a computer. The light is dim and gentle enough that most people describe it as unremarkable. A companion smartphone app (EVY Insight, available for iOS and Android) lets you adjust brightness, play a 40 Hz audio track called “Breath 40” through your own headphones for combined light-and-sound stimulation, and track your daily usage. OptoCeutics recommends morning or early afternoon sessions and daily consistency for the best results. The onboarding process includes a call with a dedicated team member who walks you through setup.
The EVY costs $1,999 with free shipping and comes with a 90-day money-back guarantee and a 10-year warranty.
There is also an iPad-based app called AlzLife that combines 40 Hz screen flicker and sound with cognitive games. A feasibility study enrolled 27 participants with cognitive complaints, mild cognitive impairment, or Alzheimer’s, with 11 completing six months of use. The study demonstrated feasibility and safety but was not designed to test clinical effectiveness.
Important caveats
Sample sizes remain small. The longest human follow-up involves five people. The OVERTURE trial had 76. Even HOPE at 670 participants is modest by pharmaceutical trial standards.
Independent replication is limited, and Cognito’s own trial missed its primary endpoint. Nearly all the positive evidence comes from Dr. Tsai and Dr. Boyden’s lab or from Cognito’s own trials. Both are co-founders of Cognito Therapeutics. This is standard in academic-to-commercial translation, and the conflicts are disclosed, but it means most of the positive evidence traces back to a financially interested group. And even that evidence requires careful reading as OVERTURE, Cognito’s largest completed trial, failed to show a statistically significant benefit on its primary cognitive-functional endpoint or on standard secondary measures like CDR-SB and ADAS-Cog14. The positive results that Cognito highlights came from other secondary endpoints. One notable attempt at independent replication, published in Nature Neuroscience in 2023 by Dr. Mattia Soula and colleagues, found that 40 Hz light flickering did not synchronize gamma oscillations in the hippocampus of Alzheimer’s model mice and produced no reliable changes in amyloid plaque burden. The Tsai lab published a rebuttal arguing the study had technical limitations, but the disagreement has not been resolved.
Effects in healthy people are unclear. A study testing 40 Hz audiovisual stimulation in cognitively healthy adults found no improvement in visual perception or spatial memory tasks, despite successfully inducing brain wave synchronization. The cognitive benefits may be specific to brains already affected by Alzheimer’s pathology, which would have important implications for anyone using these devices preventively.
The longest follow-up involved five people. The two-year MIT follow-up is often cited as evidence that 40 Hz stimulation works over the long term, but it included only five participants, three with late-onset Alzheimer’s and two with early-onset disease. The late-onset group appeared to do better, but with a sample this small, there is no way to know whether those differences reflect the biology of the disease, the stimulation, or random chance. This study generates hypotheses, not conclusions.
Consumer devices are not verified at the individual level. Unlike Cognito’s clinical protocol, consumer devices like EVY and AlzLife do not confirm with a brain wave recording that 40 Hz synchronization is actually happening in a given user’s brain. You are assuming it works as intended.
Where things stand
The preclinical science behind 40 Hz gamma stimulation is interesting. The proposed explanation for how it works, that the stimulation activates a specific type of brain cell that increases blood vessel pulsing, which drives the brain’s waste-clearing plumbing system to flush out amyloid, is technically plausible. But that entire chain was demonstrated in a single 2024 paper from a lab whose founders have equity in Cognito Therapeutics, and it has not been independently confirmed. The one lab that tried to reproduce the results, couldn’t. The safety profile across all studies appears excellent, with no serious adverse events reported.
The clinical evidence in humans is early and mixed. The largest completed controlled trial missed its primary endpoint. The positive signals come from secondary measures and post hoc analyses, which are useful for generating hypotheses but not for drawing firm conclusions. The real inflection point will be the HOPE trial results, expected in 2026. If a properly powered Phase 3 trial with a control group shows that 40 Hz stimulation slows cognitive decline and preserves brain structure in Alzheimer’s patients, that would be a meaningful advance. If it fails, the entire premise of consumer 40 Hz devices will need to be re-evaluated.
For now, the devices already available (EVY, AlzLife) appear safe. But “safe” and “effective” are different things, and anyone considering a consumer 40 Hz device should think carefully about what they are committing to. The EVY costs $1,999 upfront. The recommended protocol is one hour every day, ideally in the morning or early afternoon, indefinitely. Over a year, that is 365 hours of your life, or roughly nine full work weeks, spent sitting in front of a light panel for a benefit that has not been demonstrated in a large controlled trial. For a person with Alzheimer’s or their caregiver, that daily hour also competes with time that could be spent exercising, socializing, sleeping well, or doing other things with far stronger evidence for slowing cognitive decline. None of this means EVY is a bad bet. It may turn out to work. But buying a device and committing to daily use carries real costs in money and time, and those costs deserve to be weighed against the current state of the evidence, which is promising but unproven.
Am I going to buy a 40 Hz device to try and slow down my brain aging and prevent dementia? Nope. It doesn’t sound like what I want to do with my time and there is no evidence that this would be helpful for people without Alzheimer’s. Would I buy it if I already had cognitive impairment or Alzheimer’s? Probably not, based on what I know of the literature so far.
The strongest evidence-based strategies for reducing dementia risk remain the ones we already know well, including regular exercise, quality sleep, cardiovascular risk factor management (controlling blood pressure, blood sugar, and cholesterol), and staying cognitively and socially engaged. For a deeper look at the full set of modifiable risk factors, see my earlier post on the Nine Pillars of Healthy Brain Aging. If someone has the resources and time to add 40 Hz stimulation on top of those foundations, the risk appears low. But it should not replace them.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso