BMI and Your Brain. What the Science Actually Says.
Observational studies, randomized trials, and genetic evidence tell a complicated story about BMI and dementia. Here’s how to make sense of it.
If you’ve spent any time in the longevity or brain health space, you’ve encountered two conflicting claims. One says that being overweight or obese in midlife raises your risk of dementia. The other says that carrying extra weight in later life is somehow protective. Both are backed by published studies. And both can’t be entirely right at the same time.
Before we dig in, a brief cultural note. A research team that tracked the BMIs of Bond girls as a proxy for the cultural ideal of feminine beauty found that until the mid-1980s, the ideal BMI hovered around 20-21. Then it dropped through the 1990s into the underweight range of 17-18.5, where it has largely remained. The current GLP-1 craze in Hollywood is pushing even more people toward lower body weights, often without resistance training to preserve muscle mass.
Meanwhile, the average American woman has a BMI of about 27-28. The average American man, roughly 29-30. So the public is being pulled between two extremes. The celebrity standard that says thinner is always better. And a population norm that has drifted well into the overweight and obese range.
Neither extreme is optimal for your brain. The answer sits right around BMI 22 to 25, depending on your age, sex, and body composition. An important caveat that will come up repeatedly is that BMI cannot distinguish muscle from fat. A lean, muscular person with a BMI of 27 is in a completely different metabolic situation than a sedentary person at the same BMI carrying visceral fat. Every BMI target below should be interpreted with that limitation in mind.
The relationship between body mass index and brain health changes across the lifespan. It depends on when you measure BMI, how long you follow people, what type of study you run, and whether you account for the fact that Alzheimer’s disease itself causes weight loss years before diagnosis. I’ll walk through three types of evidence: observational studies, randomized controlled trials, and Mendelian randomization studies.
The Observational Evidence. Why Midlife Matters.
The 2024 Lancet Commission on dementia prevention now lists 14 modifiable risk factors that together account for approximately 45% of global dementia cases. Midlife obesity is one of them. This didn’t happen by accident. It reflects decades of large cohort studies showing the same general pattern.
A meta-analysis of 19 studies involving nearly 590,000 participants found that midlife obesity (BMI ≥ 30) was associated with a 33% increased risk of dementia. But what about the overweight range? This is where it gets interesting. That same meta-analysis found only a modest, non-significant trend for midlife overweight (BMI 25-30). However, an earlier meta-analysis by Loef and Walach found that midlife overweight did significantly increase dementia risk, with a relative risk of 1.34 (95% CI 1.08-1.66), compared to 1.91 for obesity. And a dose-response meta-analysis pinpointed the thresholds more precisely. All-cause dementia risk in midlife became significantly elevated when BMI surpassed 29, which is still in the overweight range. For Alzheimer’s specifically, the inflection point was around BMI 30. For vascular dementia, it was around BMI 32.
That same dose-response meta-analysis also found that midlife underweight (BMI < 18.5) carried a 39% increased risk of cognitive impairment and dementia. The Swedish Adoption/Twin Study of Aging followed people for 40 years after midlife BMI measurement and found that higher midlife BMI scores preceded lower cognitive ability and steeper cognitive decline, even after excluding people who developed dementia. This suggests that the cognitive effects of excess weight extend beyond clinical dementia into everyday thinking ability, and importantly, that even the overweight range matters for cognition even if it doesn’t always reach significance for dementia diagnosis.
The bottom line from observational data is that midlife cognitive risk follows a rough U-shape. Both underweight and obesity clearly increase risk. The overweight range likely carries modest but real risk, particularly above BMI 29, even if it’s harder to detect in every study. The safe zone appears to be normal weight, BMI 18.5-25, though as I’ll explain later, someone in the “overweight” range with high muscle mass and low visceral fat may be at lower risk than someone at BMI 23 who is sedentary and metabolically unhealthy.
But then late-life studies come along and flip the script.
The “Obesity Paradox” in Late Life
When researchers measure BMI in people already 65 or older, higher BMI often appears protective. A massive UK study of nearly 2 million people found that underweight individuals (BMI < 20) had 34% higher dementia risk, and every increasing BMI category was associated with lower risk. A dose-response meta-analysis confirmed this: late-life overweight and obesity conferred 21% and 25% reduced risk, while late-life underweight conferred 64% increased risk. The Cardiovascular Health Study found late-life underweight (BMI < 20) associated with 62% increased dementia risk.
This is the “obesity paradox,” and before you take it as license to gain weight after 65, you need to understand what’s driving it.
Reverse Causation. The Ghost in the Data.
Alzheimer’s pathology, particularly amyloid-beta accumulation, likely begins 20 to 30 years before the first clinical symptoms, and possibly earlier. One of its earliest manifestations is weight loss.
The Framingham Heart Study followed over 2,000 people for nearly four decades and found that those who eventually developed dementia showed a characteristic pattern: BMI rose through early midlife, then began declining decades before diagnosis. Those with this rising-then-falling trajectory had nearly four-fold increased dementia risk. An individual-participant meta-analysis of 1.3 million people confirmed this. BMI measured more than 20 years before diagnosis showed a 16% increase in risk per 5 kg/m² increment. BMI measured within 10 years of diagnosis showed a 29% apparent decrease. The reversal was entirely explained by follow-up duration.
This is critical for interpreting the entire late-life BMI literature. When you measure BMI in older adults and follow them for a short time, you’re comparing people losing weight because their brains are already deteriorating to people who aren’t.
What Mendelian Randomization Tells Us
Observational studies can never fully untangle cause and effect. Mendelian randomization (MR) uses genetic variants as instruments. Because genes are fixed at conception, they can’t be affected by reverse causation. If genetic variants that raise BMI also raise dementia risk, that’s strong evidence for a causal effect. The MR evidence differs depending on the type of dementia.
MR and Alzheimer’s Disease
Multiple MR studies have found that genetically predicted higher BMI does not significantly increase the risk of Alzheimer’s disease. A Copenhagen study of 399,536 individuals, a three-sample MR study from the AD Genetics Consortium, and the 2023 EADB analysis in JAMA Network Open (the largest MR analysis of its kind) all converged on the same finding: genetically higher BMI does not appear to cause Alzheimer’s disease specifically. The EADB study actually found a slight protective association, likely reflecting the complex biology of BMI-related genetic variants and their relationship with early Alzheimer’s pathology.
MR and Vascular Dementia. A Different Story.
The picture changes completely for vascular dementia. A recent MR study in The Journal of Clinical Endocrinology & Metabolism found that each standard deviation increase in genetically predicted BMI was associated with a 54-63% increased risk of vascular dementia, while confirming no causal effect on Alzheimer’s specifically. A 2024 UK Biobank MR study of over 400,000 participants confirmed this: genetically predicted higher BMI increased all-cause dementia risk, with the signal driven largely by vascular and mixed dementias rather than pure Alzheimer’s disease.
The Reverse MR. Alzheimer’s Genes Cause Weight Loss.
Perhaps the most elegant MR finding comes from “reverse” MR. A landmark UK Biobank study and its replication in the All of Us cohort showed that Alzheimer’s genetic risk scores were associated with lower BMI, with the association emerging around age 47 to 54 and strengthening with age. This is some of the strongest evidence that the “protective” effect of higher BMI in late-life studies is driven by the disease causing weight loss decades before diagnosis.
The RCT Evidence. What We Know and What We Don’t.
We do not have a single large RCT showing that intentional weight loss in midlife reduces dementia incidence. These trials would require randomizing thousands of people in their 40s and following them for decades. What we do have are trials of intermediate steps.
The US POINTER trial, a $50 million study funded by the Alzheimer’s Association, reported its two-year results in 2025. It found that a structured multi-domain lifestyle intervention (including diet, exercise, cognitive training, and cardiovascular risk management) appeared to delay cognitive aging by one to nearly two years compared to a self-guided control group. Weight management was part of the intervention, though not the sole focus.
A meta-analysis of weight loss intervention studies identified 13 longitudinal studies and 7 RCTs showing that weight loss in overweight and obese people was associated with improvements in attention, memory, and executive function. The effects were modest but consistent.
And then there’s the GLP-1 receptor agonist story, which is adding a fascinating new chapter.
GLP-1 Agonists. A Natural Experiment in Weight Loss and Brain Health.
The data on GLP-1 receptor agonists (drugs like semaglutide and liraglutide) and dementia risk is generating enormous excitement in the field. (I wrote a deep dive on whether GLP-1s may be the first true longevity drugs on my Substack.) Real-world data from over 147,000 propensity-matched pairs showed GLP-1 agonist use was associated with a 70% reduction in incident dementia. A Cleveland Clinic pharmacoepidemiologic study published in 2025 confirmed these findings, with GLP-1 agonist initiation associated with an AD hazard ratio of 0.69 or lower.
The ELAD Phase 2b trial of liraglutide in 204 people with mild to moderate Alzheimer’s showed that liraglutide reduced brain atrophy in memory-critical areas by nearly 50% compared to placebo and slowed cognitive decline by about 18% over one year, though it did not meet its primary endpoint. The EVOKE trials of semaglutide in established early AD also failed to show cognitive benefit despite reducing CRP by about 30%. A critical question is whether EVOKE started too late. These participants already had clinically diagnosed Alzheimer’s, meaning the pathological cascade had been running for two decades or more. GLP-1 agonists may be able to protect neurons that are still functional but cannot resurrect those already gone.
GLP-1 agonists do much more than cause weight loss. They reduce neuroinflammation, improve insulin signaling, and may have direct neuroprotective effects through brain GLP-1 receptors. The field is still waiting for trials that test these drugs earlier in the disease trajectory, in people with mild cognitive impairment or cognitively normal people at high risk. There is genuine reason to be hopeful given the strength of the observational signal and the biological plausibility.
The Mechanisms. How Excess Weight Damages the Brain.
Understanding mechanism helps us judge biological plausibility. The pathways are well-established.
Brain insulin resistance. The brain uses about 20% of the body’s glucose. Obesity promotes brain insulin resistance, impairing the PI3K-Akt pathway critical for neuronal survival and synaptic plasticity, and accelerating both amyloid-beta accumulation and tau hyperphosphorylation.
Neuroinflammation. Visceral fat releases pro-inflammatory cytokines (IL-6, TNF-alpha, CRP) that cross the blood-brain barrier and activate microglia. Chronic microglial activation damages neurons, impairs hippocampal neurogenesis, and reduces BDNF, creating a self-amplifying cycle.
Blood-brain barrier disruption. Obesity-induced inflammation decreases tight junction protein expression, allowing peripheral inflammatory molecules and toxic metabolites to enter the brain.
Ceramide-mediated neurotoxicity. Obesity drives hepatic ceramide production, a sphingolipid that crosses the blood-brain barrier and directly induces oxidative stress and neurodegeneration.
Adipokine dysregulation. Obesity increases leptin resistance and decreases adiponectin, which is associated with worse cognitive function and higher AD risk.
Vascular damage. Obesity promotes hypertension, dyslipidemia, and atherosclerosis, reducing cerebral blood flow and promoting white matter lesions. This is the primary pathway by which BMI causally increases vascular dementia risk.
Putting It All Together. What’s the Optimal BMI for Brain Health?
Here’s how I synthesize the evidence across all three study types.
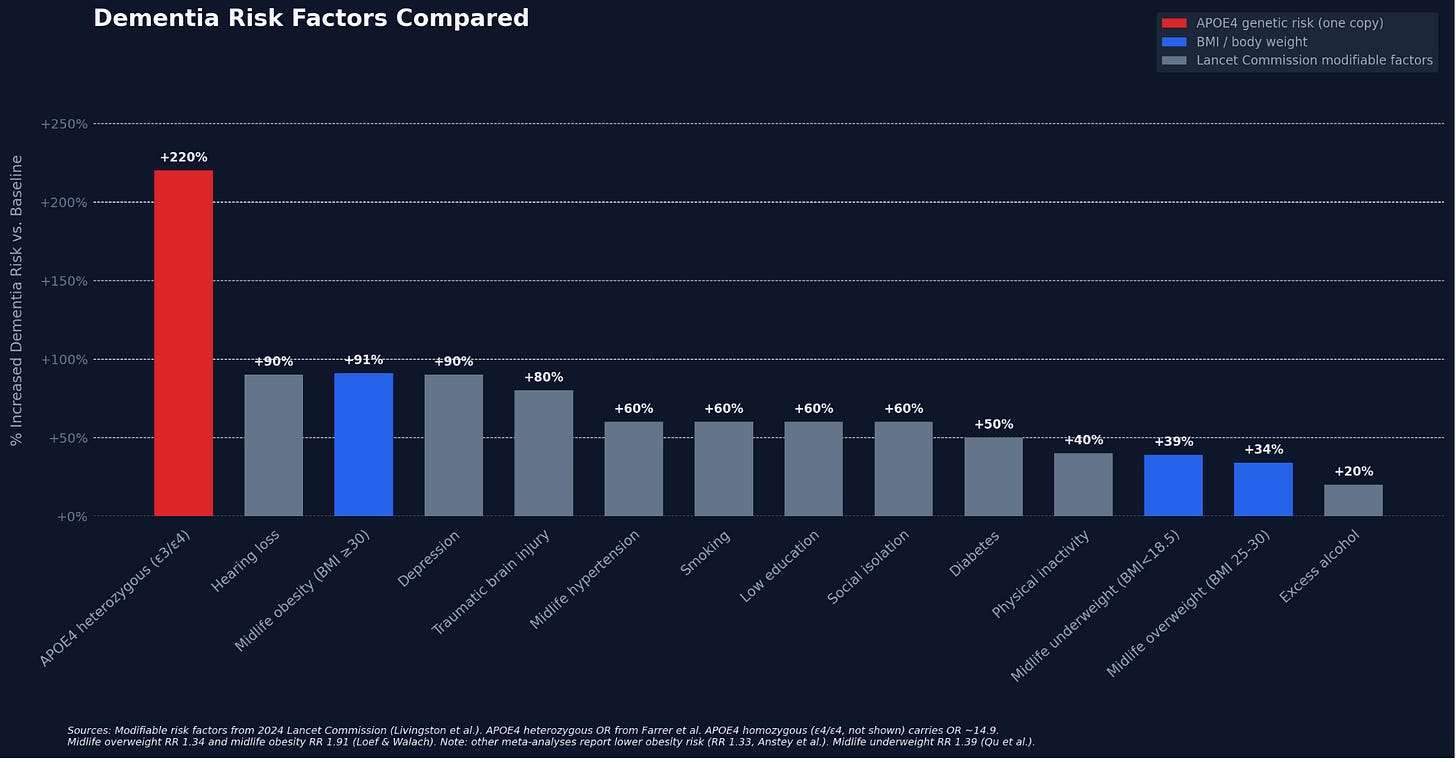
Before diving in, it helps to see where BMI falls relative to other known dementia risk factors. The chart below compares the percentage increase in dementia risk for BMI categories against other modifiable risk factors from the 2024 Lancet Commission and APOE4 genetic risk. Carrying one APOE4 allele (approximately 25% of the population) dwarfs every modifiable factor, with an odds ratio of 3.2. Midlife obesity at +91% (from Loef and Walach; other meta-analyses report +33%) sits among the most impactful modifiable risk factors. Midlife overweight and underweight carry more modest risk (+34% and +39%). The key takeaway: BMI risk is modifiable, and when you stack multiple modifiable factors together, the effects compound.
A midlife BMI somewhere around 22 to 24.9 is likely the sweet spot for both brain health and longevity, assuming average body composition. That “18.5 to 25” range I see quoted everywhere is technically correct but misleadingly broad. The actual nadir of risk is not the midpoint of that range. It’s the upper end. And again, these numbers assume a typical ratio of fat to lean mass. If you strength train seriously and carry significant muscle, your “optimal” BMI will be higher than these population-level estimates, and that’s fine, because BMI is really a proxy for metabolic health, and muscle is metabolically protective.
The best data comes from the all-cause mortality literature. The Global BMI Mortality Collaboration (239 studies, nearly 4 million never-smokers without pre-existing disease) found the mortality nadir is age-dependent: BMI 22 for ages 35-49, BMI 23 for ages 50-69, and BMI 24 for ages 70-89. A meta-analysis of 230 cohort studies found lowest mortality at BMI 20-22 in studies with 20+ years of follow-up. A UK study of 3.6 million adults found lowest risk in the BMI 21-25 range for most causes of death. The dementia-specific data is less granular, but risk starts climbing meaningfully above BMI 29, and the Framingham Offspring Study found a continuous relationship where each 1-unit BMI increase at ages 40-49 was associated with higher dementia risk.
For a midlife adult, the convergence of evidence points to a BMI around 22 to 25 (assuming average body composition). The lower end (22-23) may be optimal for younger midlife adults, the higher end (23-25) for older adults where maintaining muscle mass becomes critical. Below BMI 20, mortality and dementia risk climb. Above 25, most-cause mortality rises. Above 29, dementia risk rises meaningfully.
These are population-level estimates. A BMI of 26 in someone with high muscle mass, low visceral fat, and excellent insulin sensitivity is a fundamentally different biological state than a BMI of 24 in someone who is sedentary and metabolically unhealthy. Body composition matters far more than BMI.
The overweight range (BMI 25-29) is not a free pass. Meta-analyses vary, with some finding a significant 34% increased dementia risk and others finding a non-significant trend. Dose-response data show risk climbing around BMI 29. But the Swedish twin study showed that even midlife overweight precedes lower cognitive ability and steeper cognitive decline, independent of dementia diagnosis. You don’t have to develop clinical dementia for excess weight to affect your brain. BMI 25-29 in midlife likely carries modest but real cognitive risk, particularly at the upper end and especially if it reflects poor metabolic health rather than high muscle mass.
Being underweight (BMI < 18.5) in midlife is harmful, but interpretation requires nuance. The dose-response meta-analysis found a 39% increased midlife risk. However, midlife underweight is strongly confounded by chronic illness, smoking, depression, and malnutrition. Some MR studies suggest genetically predicted lower BMI may actually reduce dementia risk, conflicting with the observational findings. That said, severe caloric restriction, inadequate protein, and low lean body mass all have plausible pathways to brain harm through reduced growth factor signaling and loss of neuroprotective myokine release from muscle.
In late life, low and declining weight reflects reverse causation. Late-life underweight carries 62-64% increased dementia risk in virtually every study, but Alzheimer’s pathology drives weight loss beginning decades before diagnosis. The All of Us replication study confirmed that AD genetic risk scores start pulling BMI downward around age 47-54. A meta-analysis of 23 cohort studies found that losing more than 0.5% of body weight per year was associated with 28% higher dementia risk. If you or a loved one is losing weight without trying after age 60, take it seriously and consider a cognitive assessment. But interventions should focus on preventing muscle loss and malnutrition, not simply adding body weight.
Metabolic health matters more than a number on the scale. BMI doesn’t capture visceral fat, insulin sensitivity, inflammation, or fitness, all of which independently affect brain health. This raises the “metabolically healthy obesity” (MHO) question. The Whitehall II cohort found that MHO before age 60 still carried a 69% increased dementia risk compared to metabolically healthy normal-weight individuals. Being metabolically normal did not erase midlife obesity risk. Late-life MHO shows different results. A Korean study of 12 million adults found late-life MHO associated with lower AD risk (HR 0.85), and a 2024 meta-analysis confirmed this (HR 0.79), but these findings are likely confounded by the same reverse causation.
There is also a deeper problem. MHO is frequently transient. The MESA study found nearly half of MHO participants developed metabolic syndrome during follow-up. The English Longitudinal Study of Ageing put it at 44.5% within 8 years; the Framingham Heart Study at 71% over 13 years. Being metabolically healthy while obese is better than being metabolically unhealthy at the same weight, but it is not equivalent to being metabolically healthy at a normal weight, and the protection may erode over time.
And that brings us to an important point.
Why Body Composition Matters More Than BMI
Two people at BMI 26 can have radically different risk profiles. Someone who strength trains and carries significant lean mass may be “overweight” by BMI while having low visceral fat, excellent insulin sensitivity, and no elevated brain risk. Body composition, not body weight, is what actually matters.
Visceral Fat Is the Real Culprit
A 2025 Neurology study found that visceral adiposity was associated with increased dementia risk even after accounting for BMI. A UK Biobank study of 412,000 participants found high belly fat linked to 13% higher risk of neurodegenerative disease. A study of middle-aged adults at high AD risk found that hepatic fat was associated with reduced gray matter volume, and in men, pancreatic fat predicted lower cognition and hippocampal volume.
The disconnect between BMI and visceral fat is striking. A study of older Latino adults found that higher BMI appeared protective against dementia (the classic paradox), but higher waist circumference in the same cohort predicted 80-90% increased cognitive impairment risk. A systematic review comparing 20 obesity indicators found that central obesity measures showed the strongest association with memory decline, outperforming BMI.
What Body Fat Percentage Should You Aim For?
No RCT has randomized people to different body fat levels and measured dementia outcomes. General health guidelines suggest 10-20% for men and 18-28% for women, shifting somewhat with age. But visceral fat distribution matters more than total percentage. Track your waist-to-height ratio rather than fixating on BMI. Absolute waist cutoffs (40”/35”) are crude because they don’t account for height. A systematic review of 78 studies across 14 countries found that waist-to-height ratio outperforms both absolute waist circumference and BMI, with a universal threshold of 0.5 across sexes and ethnicities. The simple rule: keep your waist to less than half your height. If your WHtR is under 0.5, fasting insulin is low, and CRP is under 1.0, your body fat is probably in a range that supports brain health, even if your BMI says “overweight.”
Muscle Mass and Grip Strength. The Other Side of Body Composition.
A 2024 meta-analysis found that sarcopenia was significantly associated with mild cognitive impairment (OR = 1.58), Alzheimer’s disease (OR = 2.97), and non-AD dementia (OR = 1.68). A 2025 meta-analysis of 27 studies confirmed these findings, with even “possible sarcopenia” (low strength alone) carrying 96% increased cognitive impairment risk.
The UK Biobank body composition study found high grip strength associated with 26% reduced neurodegenerative risk, a bigger protective effect than the harm from excess belly fat. A 2025 Swedish study using DEXA and brain MRI found that higher lean muscle mass was associated with greater cortical thickness and larger hippocampal volumes, while higher visceral fat was associated with greater cerebral small vessel disease and older brain age.
The biology supports this. Skeletal muscle releases myokines during contraction (including irisin and BDNF) that cross the blood-brain barrier and support neurogenesis and synaptic plasticity. More muscle also means better whole-body insulin sensitivity and better brain insulin signaling. Sarcopenia and dementia share overlapping pathophysiology, and are in many ways two manifestations of the same aging process.
Sarcopenic Obesity. The Worst of Both Worlds.
The combination of low muscle mass and high body fat, called sarcopenic obesity, appears to be particularly damaging to the brain. It’s increasingly common in older adults, who naturally lose muscle with age while gaining fat. A 2025 meta-analysis found that sarcopenia prevalence was approximately 31% in patients with mild Alzheimer’s disease and increased with disease severity.
This has important clinical implications. Weight loss interventions in older adults must preserve muscle mass. Caloric restriction without adequate protein intake and resistance training can worsen sarcopenic obesity, potentially doing more harm than good for the brain. This is why the approach to weight management needs to be fundamentally about body composition optimization, not just making a number go down on the scale.
The emerging GLP-1 data is promising for prevention but the key trials haven’t been done yet. The large reductions in dementia risk seen in real-world GLP-1 studies are exciting, but these are observational. The RCT data so far (ELAD, EVOKE) tested these drugs in people who already had Alzheimer’s disease, and the disappointing cognitive results may simply reflect that intervention came too late in the disease course. We are still waiting for prevention trials in people with MCI or high-risk profiles but no dementia, and those results could change the landscape entirely. In the meantime, one real concern with GLP-1 agonists is muscle mass loss. Up to 40% of the weight lost on semaglutide can come from lean tissue, which is precisely the wrong direction for long-term brain health if resistance training isn’t part of the program. With adequate protein intake and resistance training, people on GLP-1 agonists lose minimal muscle with weight loss.
Optimal Body Composition Targets for Brain Health
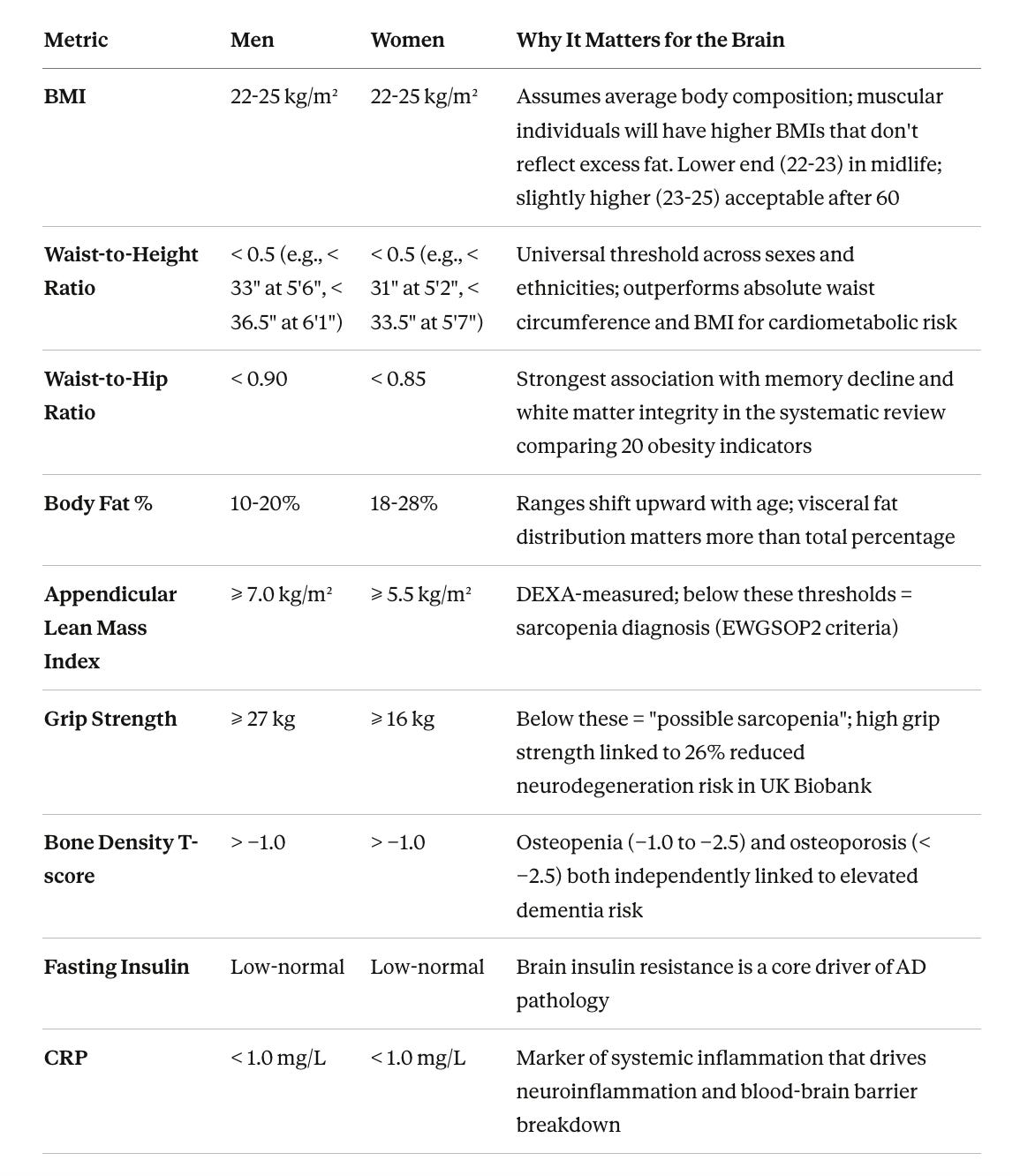
Pulling together the evidence reviewed throughout this article, here are the body composition targets that the current literature suggests are most protective for long-term cognitive health.
These are not definitive cutoffs derived from a single dementia-prevention RCT, because no such trial exists. They are synthesized from the mechanistic, observational, and Mendelian randomization evidence reviewed above. What you should take from this table is that body composition matters far more than body weight, and that the combination of low visceral fat, high muscle mass, intact bone density, and good metabolic markers defines the phenotype most consistently associated with cognitive protection.
Why Everyone Should Get a DEXA Scan
BMI is a crude tool that conflates muscle with fat and ignores visceral fat distribution. A DEXA scan (dual-energy X-ray absorptiometry) solves most of these problems in a single 10-minute, low-radiation appointment. Modern DEXA provides a three-compartment breakdown of bone mineral content, lean tissue, and fat mass, giving you precise data on total body fat percentage, regional fat distribution, estimated visceral adipose tissue, appendicular lean mass (for sarcopenia diagnosis), and bone mineral density.
This matters practically because two people at the same BMI can have radically different risk profiles. A person at BMI 26 with high muscle mass and low visceral fat is in a fundamentally different metabolic state than someone at BMI 24 with low muscle and high visceral fat. Only a DEXA scan reveals the difference. For anyone on a GLP-1 agonist or any weight loss program, serial DEXA scans are especially valuable because they tell you what you’re losing. If 40% of the weight lost is lean tissue, you need to adjust protein intake and resistance training before the muscle loss becomes a problem.
DEXA for Bone Density
Osteoporosis and dementia share overlapping biology driven by chronic inflammation, hormonal decline, vitamin D deficiency, and physical inactivity. A meta-analysis of 8 studies found osteoporosis patients had double the cognitive impairment risk (OR 2.01). A large prospective cohort confirmed elevated dementia risk with osteoporosis, especially in those with fractures (HR 2.43). A Rotterdam cohort study in Neurology found lower femoral neck bone density associated with increased AD risk.
The mechanisms are increasingly understood. Estrogen regulates both bone and brain metabolism. Osteocalcin, a bone-derived hormone, crosses the blood-brain barrier and supports hippocampal function. And the consequences of osteoporosis, particularly hip fractures, are devastating for cognitive outcomes. Every adult over 50 should have a baseline DEXA scan. If your T-score is below -1.0, address bone health and brain health simultaneously, because the biology is shared.
What to Ask For
Ask for a whole-body DEXA with body composition analysis, not just the standard bone density screen. This gives you fat mass, lean mass, visceral fat estimate, and regional breakdown in addition to bone data. The radiation dose is minimal (equivalent to a day or two of background radiation). I recommend a baseline in your 40s and repeating every one to two years, especially if you’re making significant changes to diet, exercise, or medication.
The Bottom Line
Midlife obesity consistently increases dementia risk across large meta-analyses, while the apparent “protective” effect of higher BMI in late life is almost certainly an artifact of Alzheimer’s pathology causing weight loss decades before diagnosis. Midlife underweight (BMI < 18.5) also carries roughly 39% increased risk, though this is heavily confounded by chronic illness, smoking, and malnutrition. The Hollywood-driven push toward ever-lower body weights, accelerated by GLP-1 agonists used without resistance training, may be trading one set of brain health risks for another. Mendelian randomization confirms that genetically elevated BMI causally increases dementia risk, particularly for vascular dementia.
But BMI is a crude metric that conflates muscle with fat. A muscular person at BMI 27 may have far lower dementia risk than a sedentary person at BMI 23 with high visceral fat. Central adiposity, best captured by waist-to-height ratio (keep it below 0.5), is a stronger predictor of cognitive decline than BMI.
What matters most is body composition. High muscle mass is independently protective, with sarcopenia nearly tripling Alzheimer’s risk and high grip strength reducing neurodegeneration risk by 26%. Bone density is linked to dementia through shared biology. The worst combination is sarcopenic obesity, which is low muscle and high visceral fat simultaneously.
The goal is not a number on the scale. It is a metabolic environment that supports brain health. Low visceral fat, high muscle mass, intact bone density, good insulin sensitivity, low systemic inflammation. A DEXA scan is the single most informative test for measuring where you stand, and everyone should get a baseline in their 40s.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso