APOE and Alzheimer's
Understanding the most common Alzheimer's risk gene and what to do about it
I was sitting in NeuroAge’s headquarters when I decided to “rip the bandaid off” and look at my genetic risk for Alzheimer’s Disease. I had been putting it off because I had for most of my life thought that there wasn’t much I could do about it.
One of my great grandmothers, one of my grandmothers, and one of my aunts all have had Alzheimer’s and it has been devastating. Imagining my own cognitive decline and the distress that it could put on others around me is a scary thought.
It nearly took my breath away to find out that I had one APOEε4 allele, the most common genetic factor for Alzheimer’s.
My co-founder, who was sitting next to me, asked “are you ok?”. “Yes” I said and I was. My initial shock over my genetic risk lasted just a few minutes and then I felt better. I actually felt aligned and calmer than I had been before. I knew that I could prevent my own Alzheimer’s, even with my genetic risk factor, through lifestyle choices.
On average, people have a 10-15% lifetime risk up to age 80–85 years for mild cognitive impairment or Alzheimer’s dementia according to population-based studies with a majority of individuals of European ancestry. That risk jumps to 20–25% for APOEε3/ε4 carriers and 30–55% for people with two copies of the APOEε4 allele. There is also a protective version of APOE, called APOEε2.
My own research out of MIT showed that people who were 5 years younger in their biological brain age had significantly lower risk than the average person, even if they had one or two APOEε4 alleles. This means that you can mitigate your genetic risk from APOEε4 entirely through lifestyle, which is remarkable.

Having a brain that is 5 years younger biologically is an achievable goal. It’s one standard deviation younger than the average person— or the equivalent of getting a “B” in brain aging.
If you would like to know how to slow down your biological brain aging, read my post on the NeuroAge Neuprint— the nine pillars of slowing down or reversing your brain aging. You can also take our lifsetyle quiz to see how you are doing.
My NeuroAge is currently 6.7 years younger than my chronological age, which means that I am on track to avoid Alzheimer’s Disease if I live to be 85 years old. I’d like to reduce my NeuroAge even further and am working on improving my lifestyle.
Having a younger biological brain age is important, not just for people genetically predisposed to Alzheimer’s, but for everyone. Our research showed a 2X increased risk of Alzheimer’s in people with biological brain ages 5 years older who do not have an APOEε4 allele. Like with many diseases, genetics and lifestyle choices work together to determine Alzheimer’s risk.
People who have less genetic risk just need to worry a bit less than people with APOEε4 alleles. Being average in their brain aging means they are unlikely to be diagnosed with Alzheimer’s in their lifetime if they live to be 80-85 years old.
However, for those people aiming to live to be 100 or more, biological brain aging mitigation is important to prevent cognitive decline regardless of genetic risk factors. Most of us will develop Alzheimer’s if we live long enough.
Having a biologically younger brain is also good at any age. It means faster reaction time and better memory. It even predicts less depression risk.
What is APOE and how do variations in it change risk for Alzheimer’s?
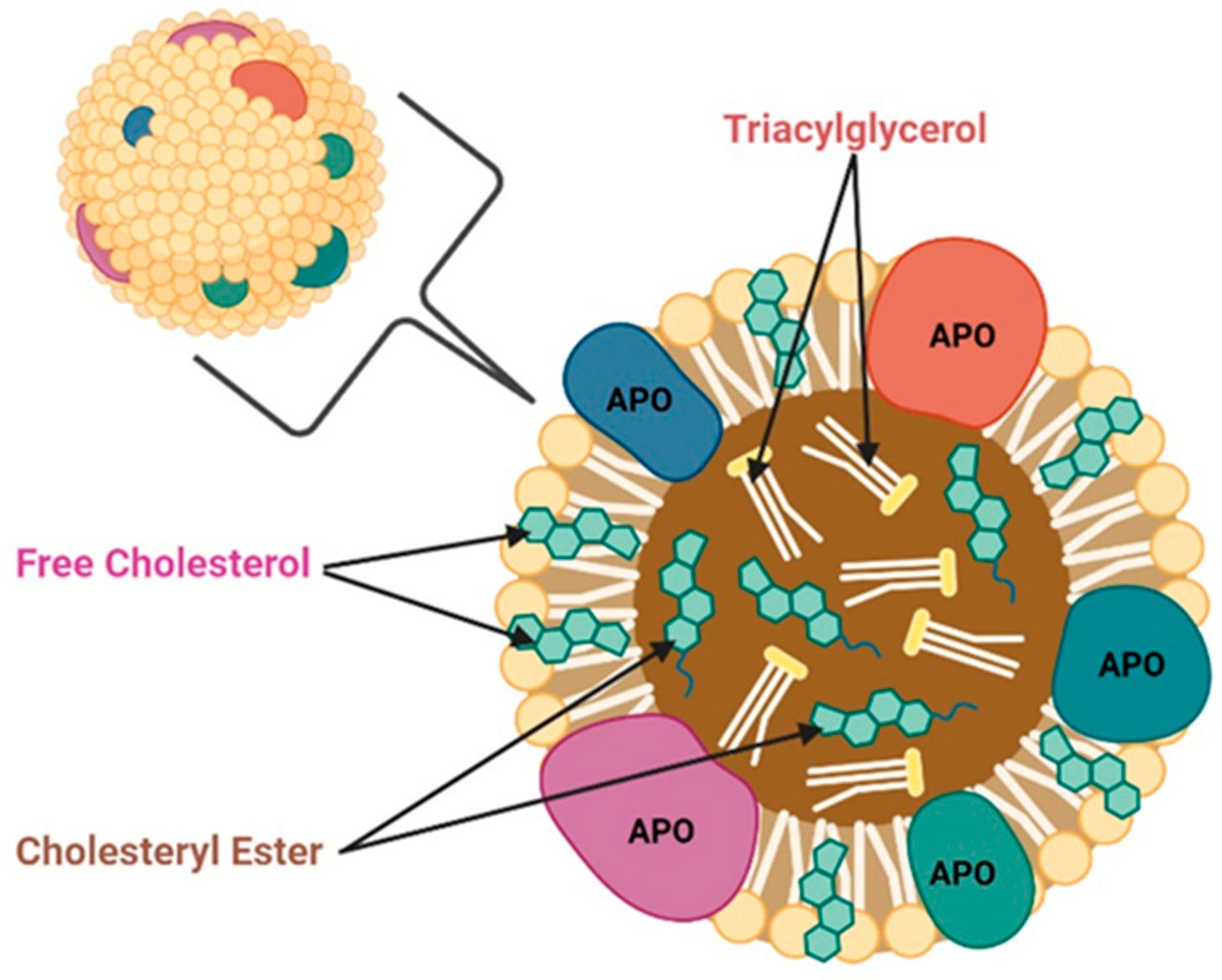
APOE is an apolipoprotein (APO), which help transport lipids (triglycerides and cholesterol) in the body. APOs move lipids in the blood stream and in tissues and deliver them from one cell type to another to feed and remove toxic lipids from cells.
You may be most familiar with APOEs “cousin”, APOB, which is commonly tested for in yearly exams at the doctor’s office. Having higher APOB levels is associated with worse cardiovascular health outcomes and more arterial plaque build up. One major difference between APOB and APOE, is that it is not produced and does not circulate in the brain. APOE is produced in the brain and regulates lipids there as well as in the rest of the body.
Some people have two one letter variations in the APOE gene that change the protein slightly by changing two of the building blocks of the protein, amino acids. People with the APOEε4 variant have two Arginines at position 112 and 158 instead of one Cysteine and one Arginine (APOEε3) or two Cysteines (APOEε2).
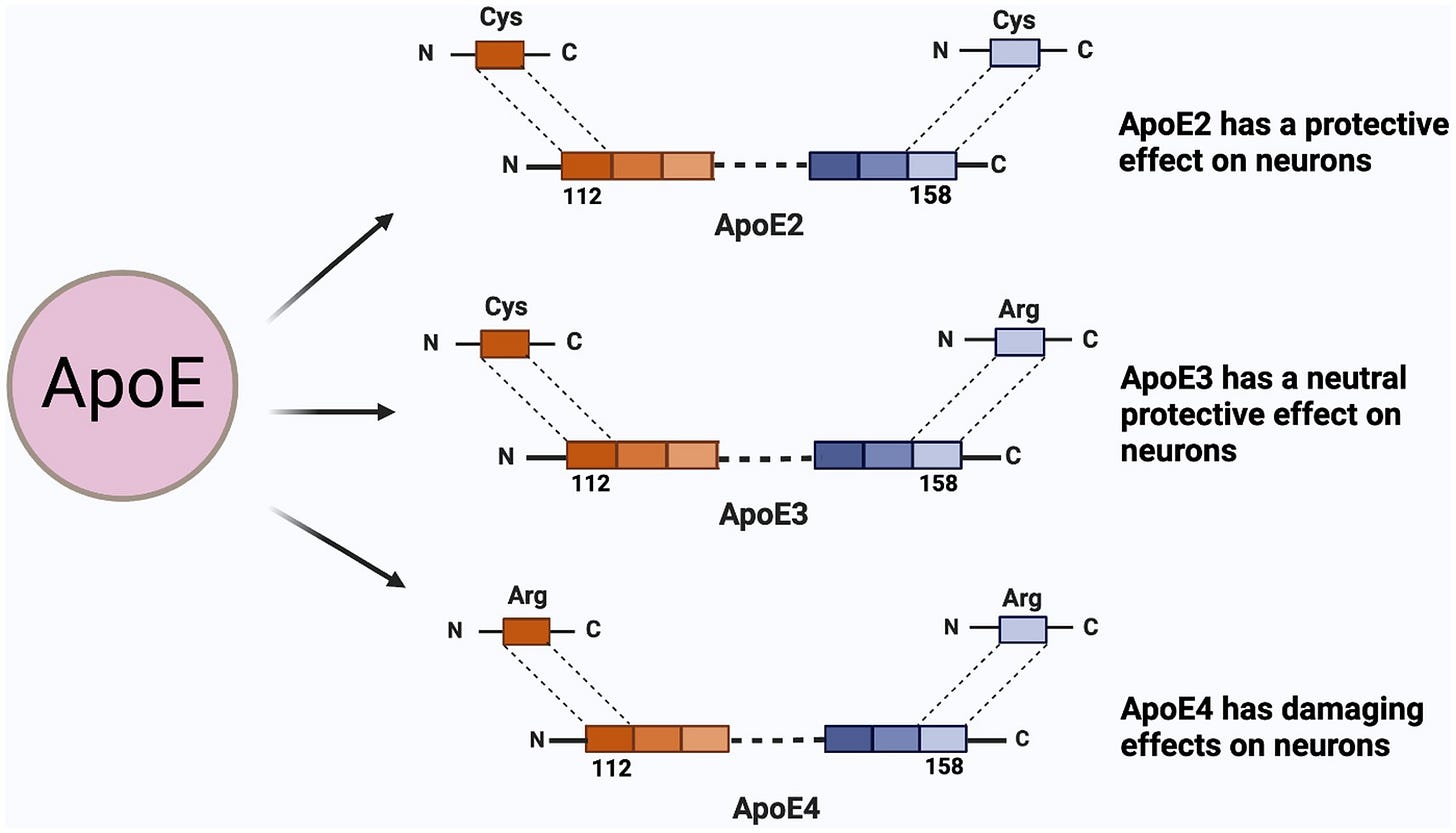
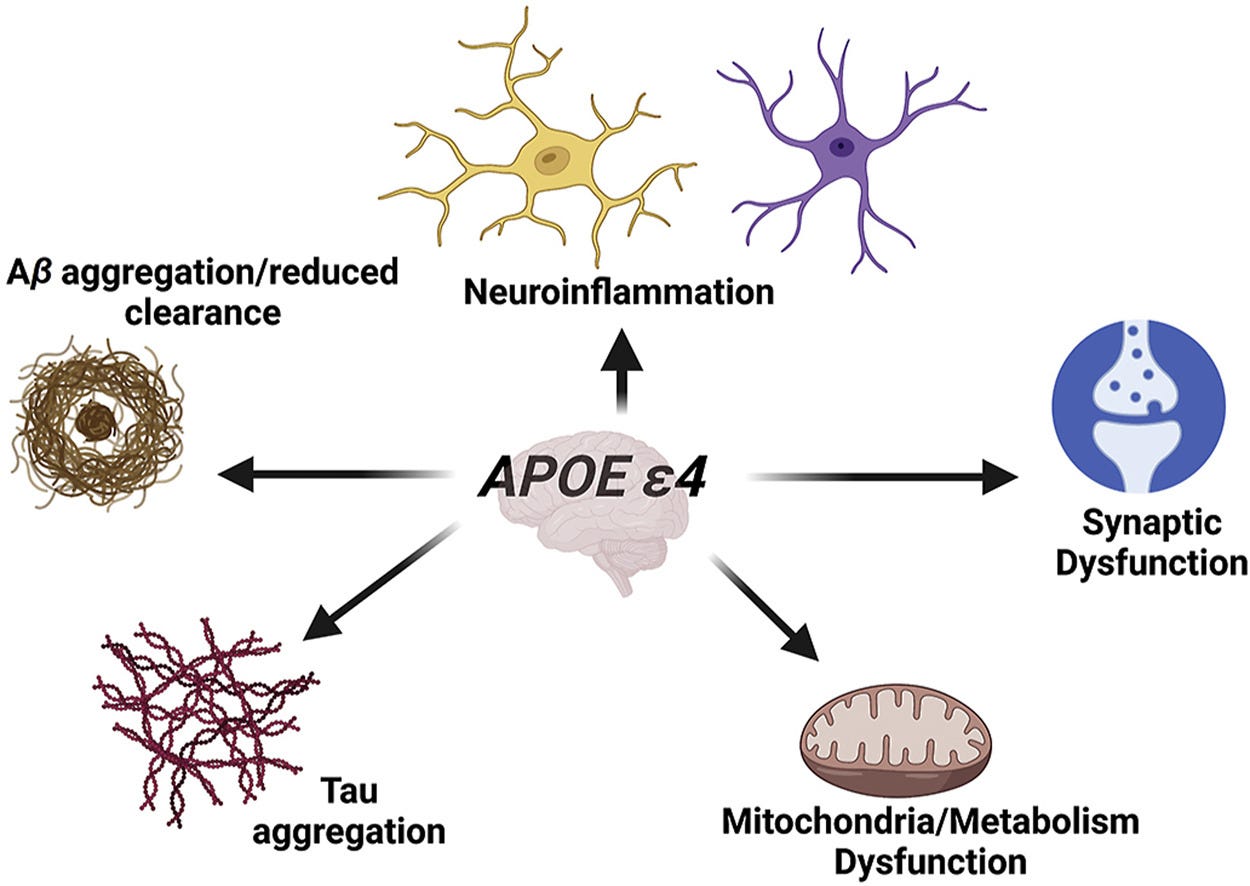
Changing the APOE protein slightly, makes it function a little differently. It has a slightly tighter shape and transports lipids less well, which has a variety of negative impacts on the brain.
APOEε4 is associated with neuroinflammation, synaptic dysfunction, toxic protein build up, and mitochondrial dysfunction. These problems lead to faster memory decline and more neuron death with age.
About 20% of people have one copy of the APOEε4 gene and about 2% of people have two copies. If you have had your genetic resilience tested with NeuroAge, you can look to see if you have the APOEε4 gene or the APOEε2 gene under your Alzheimer’s resilience score.
Note that APOE makes up only about 30% of your genetic risk for Alzheimer’s Disease. We test for >40 genes that contribute to your risk for a highly accurate score. Here I am just discussing APOE. There are past and will be upcoming posts on other genetic risk factors.
Other effects of APOE genotype
In addition to putting people at higher risk for Alzheimer’s Disease, the APOEε4 gene has other deleterious effects. Some scientists consider APOE to be a longevity gene as it also relates to lifespan, with the APOEε2 variant associated with longer lifespan and the APOEε4 with shorter lifespan. These associations seem to be independent of cholesterol, cardiovascular risk, and even Alzheimer’s diagnosis suggesting an as of yet unknown mechanism mediating APOE’s effects on lifespan.
In the UK BioBank and other large databases, the APOEε4 allele has been associated with higher LDL cholesterol and triglycerides levels, lower HDL cholesterol levels and an increased risk of coronary artery disease. Conversely, APOEε2 has been associated with lower LDL cholesterol levels, increased triglyceride levels, and increased risk of peripheral vascular disease.
Importantly, the rare people with two APOEε2 alleles are at increased risk of developing hyperlipoproteinaemia type III (also known as familial dysbetalipoproteinaemia), which causes very high triglyceride levels. This means that we should exercise caution in overcorrecting, with gene therapies for instance, by changing everyone to have two APOEε2 alleles with CRISPR.
If APOEε4 is so harmful, why did it stick around in the gene pool evolutionarily?
It has been suggested that earlier in life APOEε4 may have some benefits such as better immune system, improved fertility, reduced cancer risk, and even better cognition. These associations need to be more thoroughly studied.

This underscores the need for caution in creating therapies that target APOEε4 early in life as they might also create deleterious effects.
Since the positive benefits happen earlier in life, there might be a sweet spot for when to administer APOEε4 therapies, possibly in the earliest stages of cognitive decline when irreversible damage has not yet occurred but late enough in life that it won’t have negative consequences.
Therapies in development for APOE
Below are some of the approaches that researchers are investigating to repair APOEε4 and treat Alzheimer’s. These include blocking APOE’s interaction with toxic plaques in the brain, increasing APOEε2, decreasing APOEε4 with antibodies or ASOs, increasing lipidation on APOE, and changing the shape of APOEε4 to be more like APOEε3.
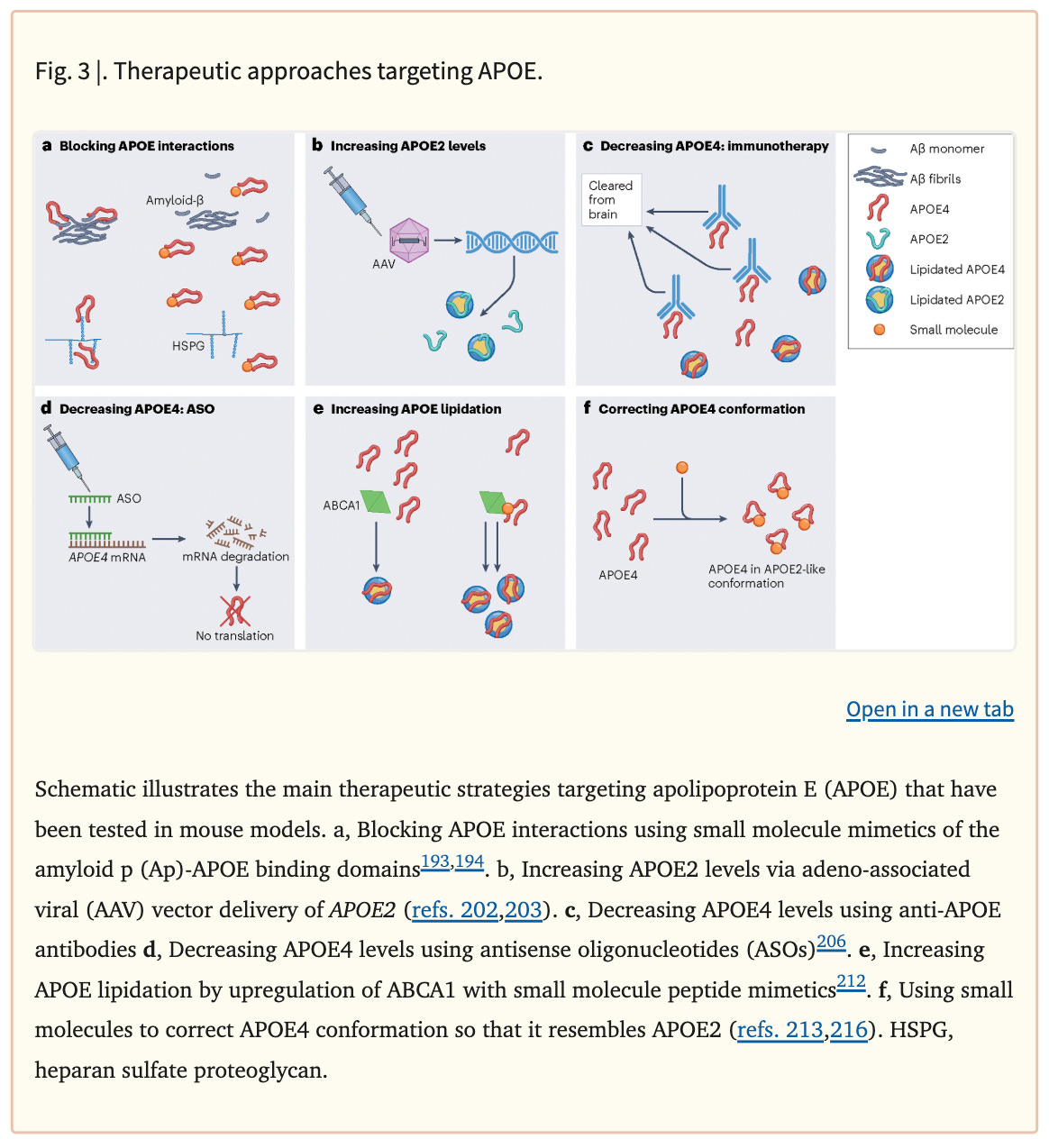
Much of this research has not yet reached the clinical trial phase and is still being studied in cells and in mice.
Existing clinical trials include repurposing FDA approved drugs, APOE “mimetics”, a small molecule to decrease toxic protein build up, and a gene therapy to add APOEε2 to people with two APOEε4 alleles.
Repurposing FDA approved drugs
Probucol
Probucol is a US FDA-approved, but no longer in use, non-statin lipid-lowering drug that has been used historically to lower cholesterol and treat cardiovascular disease and xanthomas (little fat deposits on the skin). It has been shown to counteract brain pathology and cognitive impairment in Alzheimer’s mouse models.
There are a few clinical trials being run for early Alzheimer’s including a phase 2 trial in Australia and a phase 1/2 in Canada. These appear to be run by non-profits and academic institutions, likely because Probucol is now off-patent and therefore not profitable for large pharmaceutical companies.
Bexarotene
Bexarotene is a US FDA-approved retinoid X receptor agonist for lymphoma that has been reported to cause a rapid reduction in toxic brain proteins (amyloid plaques) restoration of cognitive functioning in Alzheimer’s disease mouse models by enhancing APOE lipidation, and increasing APOE levels. This result was, at least partly, replicated by some investigators but not others, and led to examine bexarotene for Alzheimer’s disease in human clinical trials. A phase 1b trial in young volunteers revealed weak penetrance of bexarotene into the brain. Bexarotene was able to increase CSF APOE levels by 25%. In another clinical trial from the Cleveland Clinic in 20 patients with moderate Alzheimer’s disease, bexarotene was associated with a significant reduction in toxic brain proteins (Aβ PET burden) but contrary to the prediction, only in APOE ε4 non-carriers. It also increased triglyceride levels in participants, which could pose a safety risk.
These studies are very small and preliminary but suggest to me that bexarotene may be a good lead but improvements to BBB penetration and safety are required for it to be an effective drug for Alzheimer’s.
APOE mimetics
What if you could inject APOEε3 into the blood stream of people with APOEε4 alleles to correct the problem? This sounds like it could work but injected APOE is too big to cross the blood brain barrier (BBB). Instead, scientists have created a small 5 amino acid version of APOE that does cross the BBB, called CN-105. In a phase 1 clinical trial run out of Duke in 2016, it appears to be safe. However, I could not find any follow up clinical trials.
Small molecule disruption of amyloid aggregates
Alzheon Therapeutics recently completed a Phase 3 clinical trial for it’s small molecule amyloid oligomer disrupting therapy in people with two APOEε4 copies. It failed it’s primary endpoint but did seem to work better in mild cognitive impairment (early cognitive impairment that sometimes progresses to Alzheimer’s Disease).
Gene therapy
Lexeo Pharma has created an gene therapy (LX-1001) to add APOEε2 to people with two copies of APOEε4. A small 15 person phase 1 study showed that LX-1001 was safe and that 9/13 patient showed a decrease in various forms of the toxic protein, tau. They did not show decreases in amyloid.
These results are encouraging but require larger efficacy clinical trials, which demonstrate improvements not just in biomarkers but also in cognition.
Summary
Having one or two copies of the genetic risk factor APOEε4 does increase risk of Alzheimer’s, increase rates of cognitive decline, increase cholesterol and triglycerides, and increase mortality. However, it also has some potential benefits to cognition, immunity, and fertility earlier in life. The good news is that the harmful effects of APOEε4 can be mitigated by the lifestyle interventions outlined in NeuroAge’s Neuprint. APOEε4 is a lipid problem at its heart and taking care of your cardiovascular health is very important for dementia prevention, including use of statins.
Various strategies are being employed by researchers and pharma companies to target APOEε4, including repurposing drugs, APOE mimetics, a small molecule, and gene therapies. From my read, repurposing Probucol and LX-101, an APOEε2 gene therapy seem the most promising. I wonder why I haven’t heard much about CRISPR gene therapy to turn APOEε4 into APOEε2? This seems like an obvious solution and having only partial efficacy with delivery may be an advantage as too much APOEε2 also can have negative consequences.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso