How to Measure and Reduce Your Exposure to Toxins
What the science says about testing for molds, heavy metals, PFAS, organic acids, and pesticides
A study published in Nature Medicine in April 2026 analyzed brain aging data from 18,701 people across 34 countries and found that the combined burden of physical and social environmental exposures (collectively called the exposome, meaning the totality of everything a person is exposed to across their lifetime) was associated with a 3.3 to 9.1 times higher risk of accelerated brain aging. The authors concluded:
“The exposome accelerates brain aging in health and disease.”
That study measured country-level factors including air quality, temperature, greenspace, socioeconomic inequality. While, it was not granular enough to measure individual chemical exposures, reading it pushed me to think more carefully about whether I and my clients at NeuroAge should be doing environmental toxin testing, and if so, to what extent.
Standard medicine has a well-developed toolkit for finding common causes of unexplained illness. What it largely does not look for is environmental chemical burden. A patient with declining kidney function, persistent fatigue despite normal thyroid panels, or immune suppression without an identified cause may have contributing exposures that are rarely considered in standard workup. I certainly did not learn about this in medical school.
Many patients seek answers from integrative and functional medicine practitioners to explain their unexplained clinical findings.
Environmental toxin testing cannot confirm causation in the individual, but it can identify plausible contributors and support a reduce-and-retest approach. Find the source, reduce exposure, and retest months later to see whether levels fall. If they do and the clinical picture improves, that is not proof, but it is meaningful signal. The evidence behind the tests varies widely. Some chemicals have decades of human cohort data behind them, others rest on animal studies or contested epidemiology, and test quality ranges from methods validated by the CDC (Centers for Disease Control and Prevention) to poorly characterized reference populations. This article works through all of it, from a tiered test overview and evidence table to a cost breakdown and practical exposure reduction steps.
Available tests, how to evaluate them, and when to use each
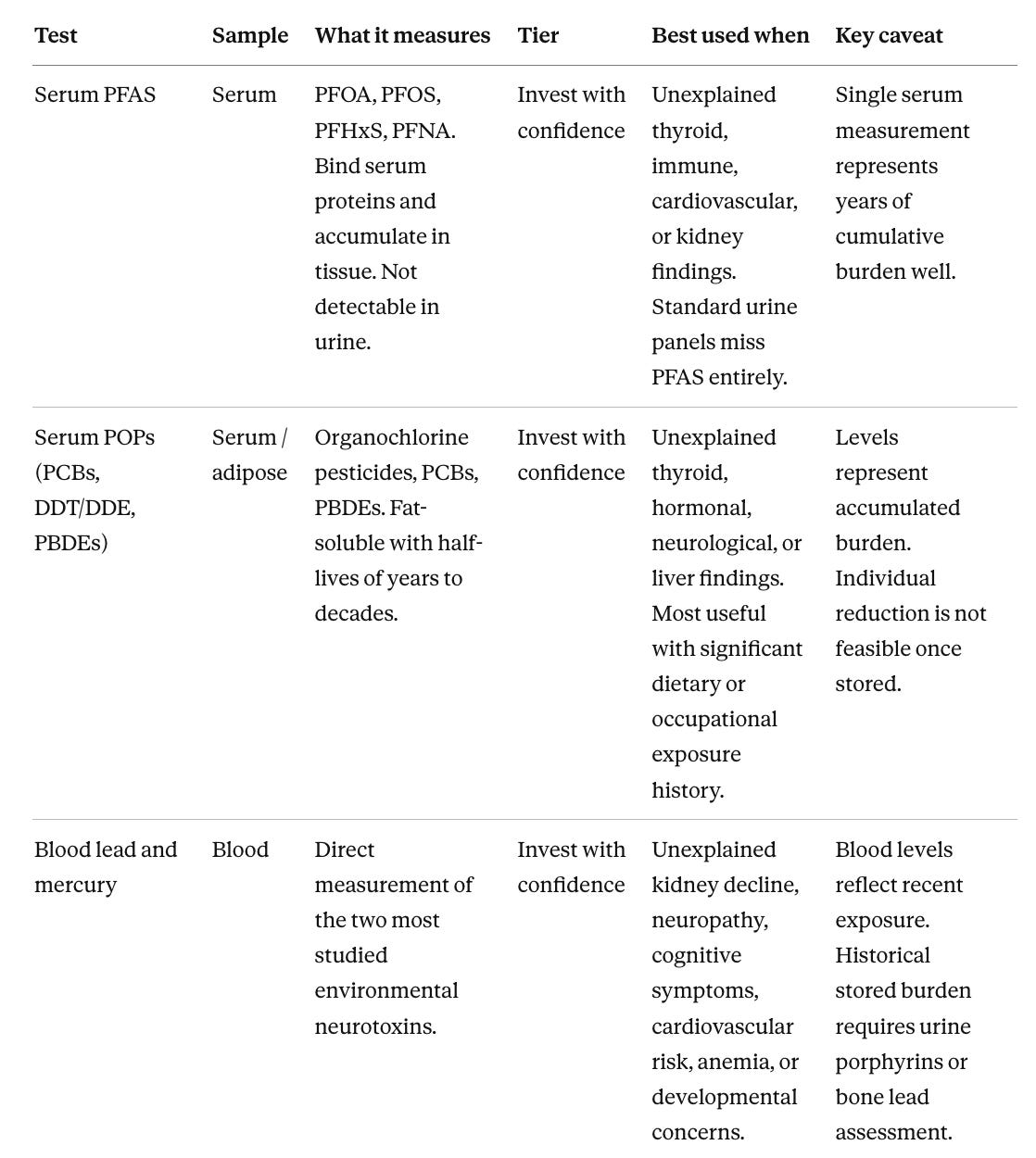
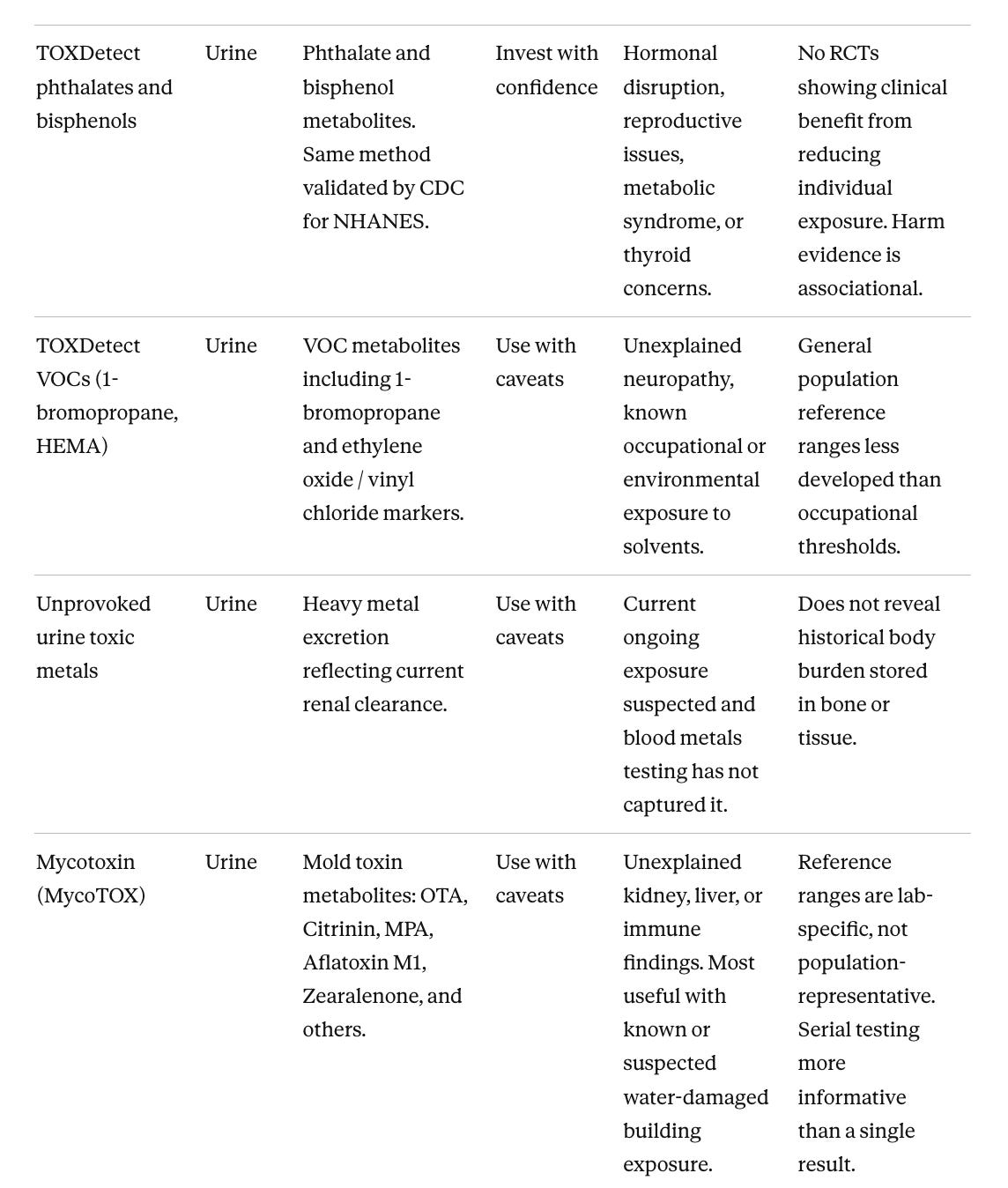
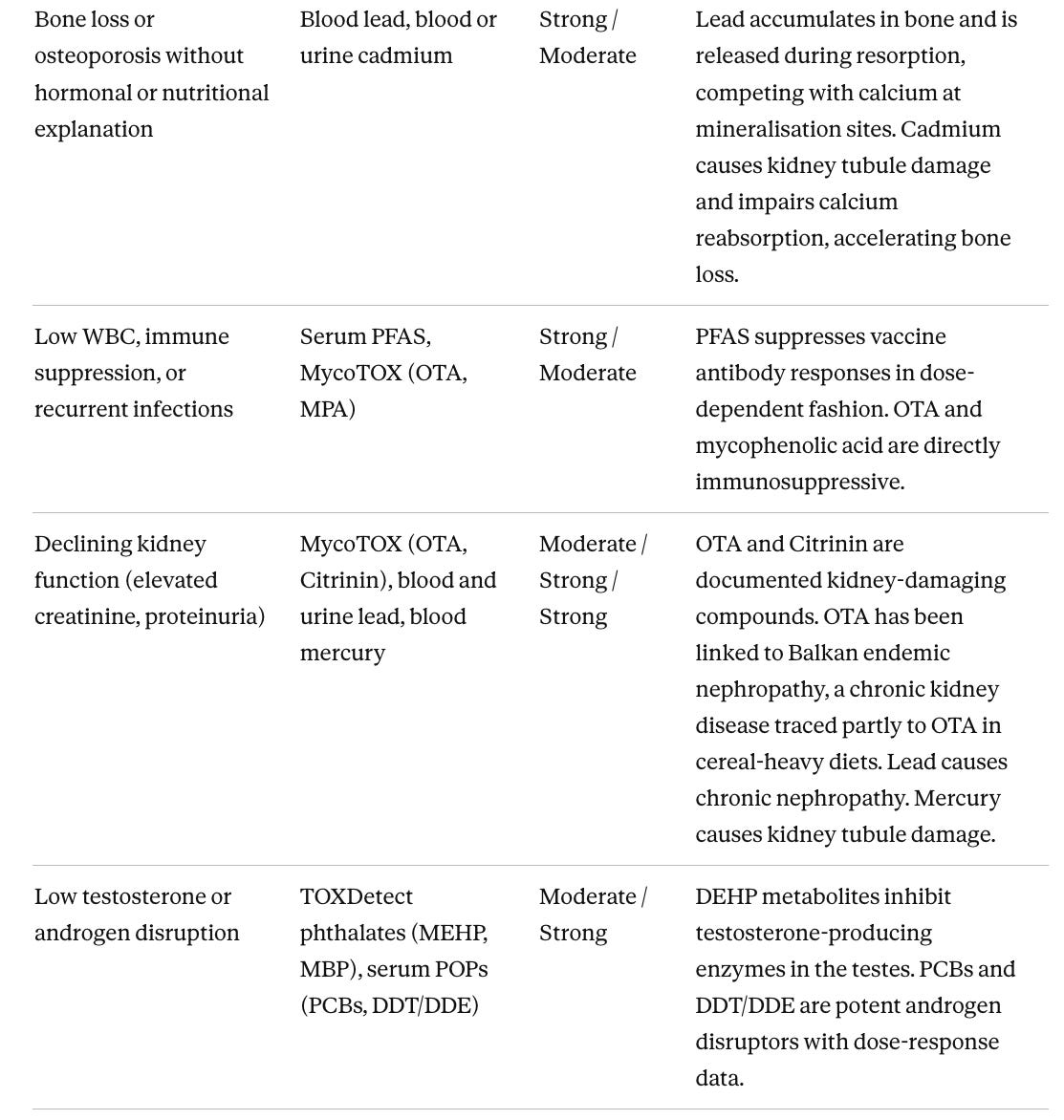
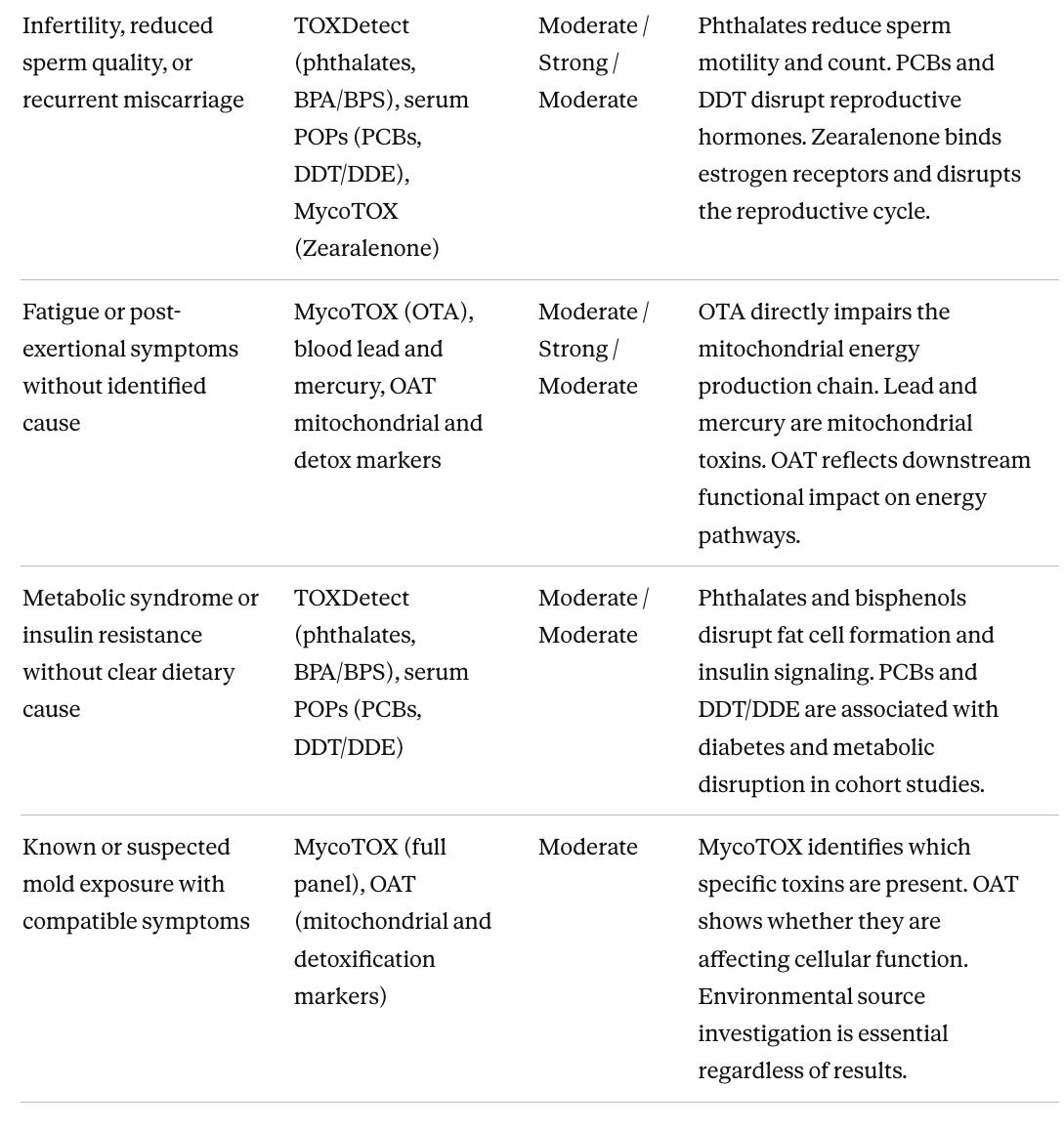
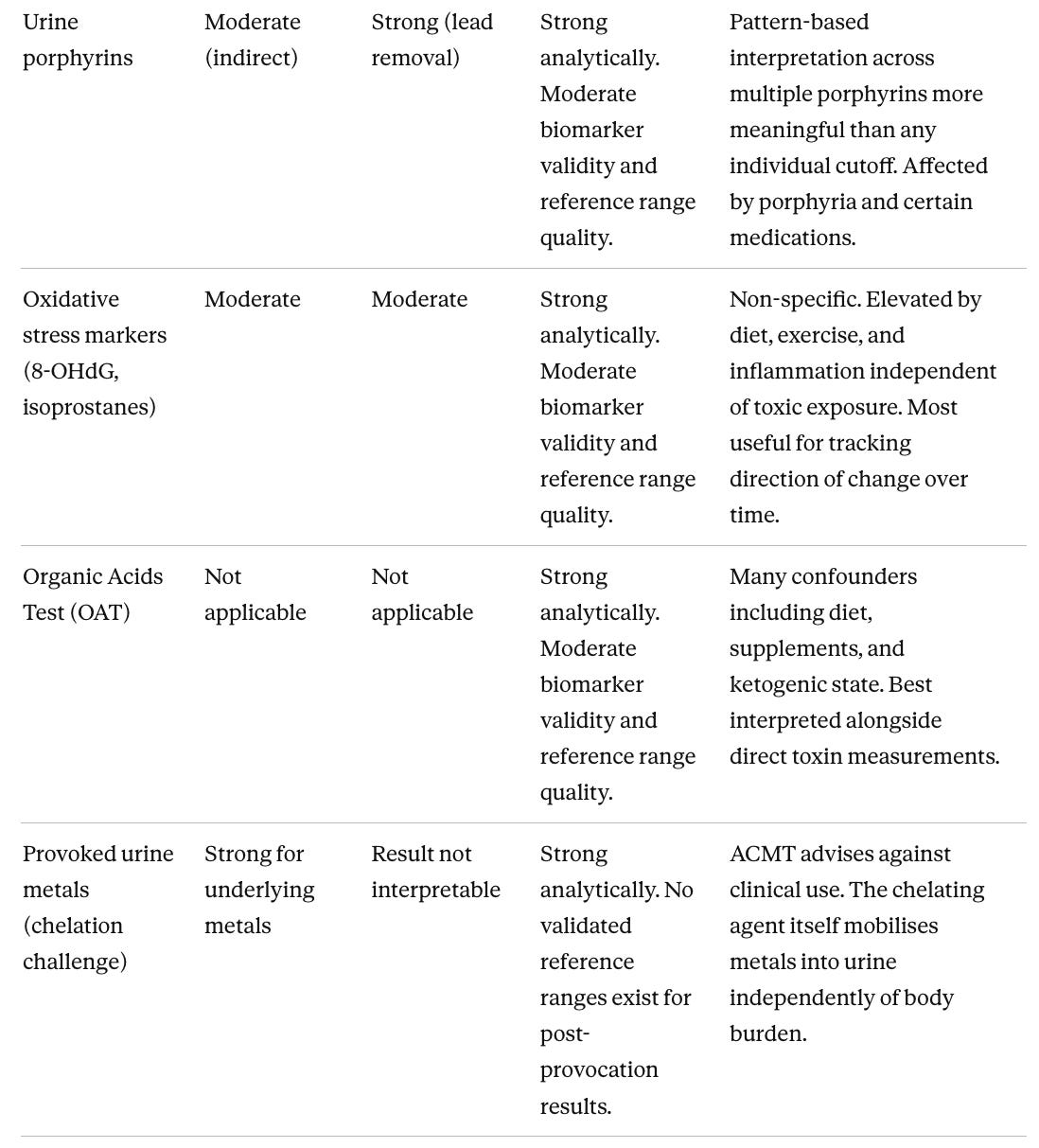
The first table lists available tests with its recommendation tier and practical guidance. The second maps specific unexplained clinical findings to the tests with the best-evidenced connection to that organ system. Together these two tables answer the questions most clinicians and patients have, which are which tests are worth ordering, and which tests make sense given this particular clinical problem.
The sample column matters in the first table. Fat-soluble compounds (those that dissolve in fat and store in fatty tissue) and protein-bound compounds (those that attach to proteins in the bloodstream rather than circulating freely) accumulate in tissue rather than being filtered into urine through the kidneys, so a standard urine panel will miss PFAS (per- and polyfluoroalkyl substances, a family of industrial chemicals) and persistent organic pollutants (long-lasting synthetic chemicals such as PCBs and pesticides that accumulate in fat and tissue) entirely. The Organic Acids Test (OAT) sits outside the tier structure because it does not measure toxic chemicals directly. It measures downstream byproducts of cellular metabolism, markers that reflect how the body is functioning rather than what it has been exposed to.
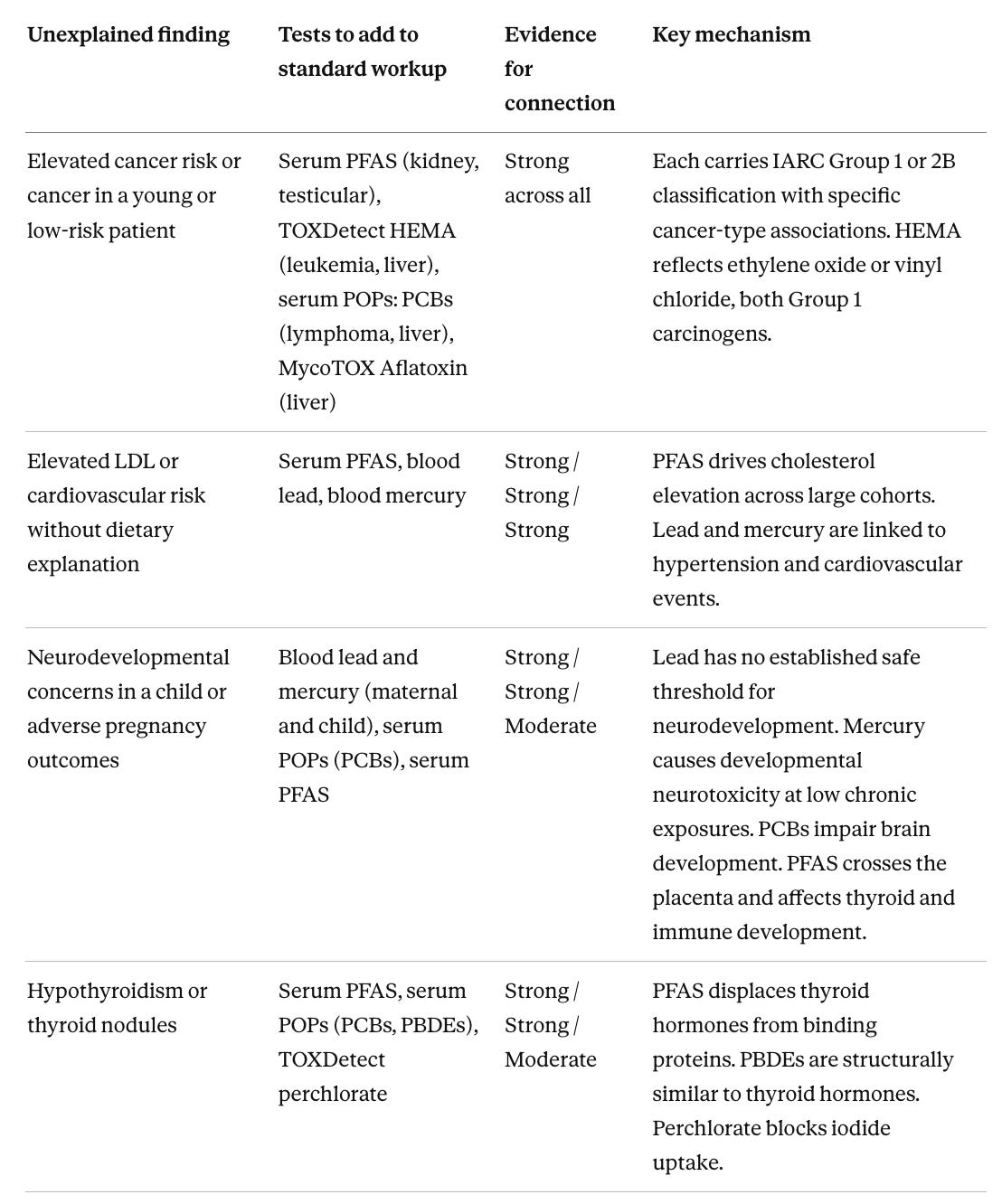
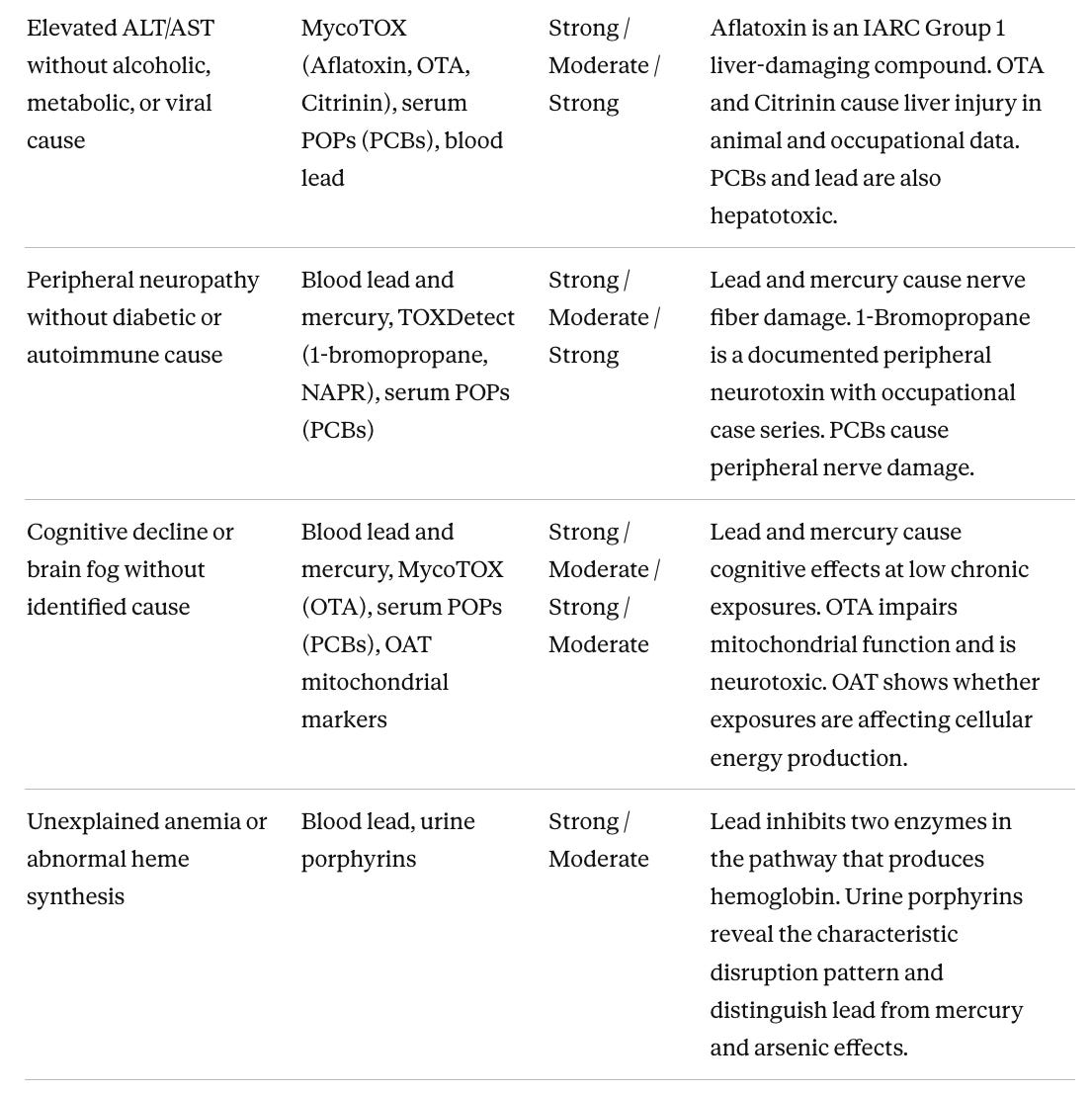
Matching tests to a specific clinical problem
The most efficient approach to evaluation is targeted testing driven by a clinical question. When a patient has a specific unexplained abnormality after standard workup, the relevant question is which toxins have documented effects on that organ system and which tests can detect them. The table below maps unexplained clinical presentations to the tests with the best-evidenced connection, with evidence levels rated for that specific organ connection rather than for the chemical overall. Many presentations overlap. Someone with thyroid dysfunction, fatigue, and immune suppression has three converging reasons to run serum PFAS. Glyphosate does not appear in this table because there is no well-established link between glyphosate and any specific unexplained clinical presentation in humans at typical population-level exposures.
How to read the evidence in this article
Every toxin test requires answering two independent questions:
Whether the exposure is harmful, and whether reducing it produces measurable benefit.
Whether the test itself accurately measures what it claims.
These two criteria don’t necessarily move together. Phthalate urine testing uses the same validated method as CDC national health surveys, so the measurement is reliable. There are no controlled trials showing that reducing individual phthalate exposure improves clinical outcomes. The CIRS panel is the reverse. The individual markers have real biological meaning in other contexts, but the composite diagnostic pattern has not been validated against independent patient cohorts.
Randomized controlled trial (RCT) evidence for exposure reduction is structurally sparse across this entire field. You cannot ethically randomize people to higher mold exposures, and most intervention data comes from case series and clinical observation rather than blinded trials with hard endpoints. Chelation therapy for lead (treatment using agents that bind to metals in the bloodstream and help remove them from the body) is one of the few areas where a controlled trial exists. The TACT trial showed modest cardiovascular benefit in diabetic patients with prior heart attacks. It is the exception, not the pattern. Treatment decisions based on these tests should be grounded in plausibility and risk-benefit, not proof of effect.
Reference ranges are also widely misunderstood. Most labs report the 95th percentile cutoff of their tested population. This is a statistical threshold, not a biological danger threshold. A result above it means a level is in the top 5% of those tested. A result below it does not mean the level is safe. It means it is typical for a population that may itself have broadly elevated exposures. The most interpretively meaningful results are those that are markedly elevated, fit a compatible clinical picture, and remain elevated on retesting despite attempted source removal.
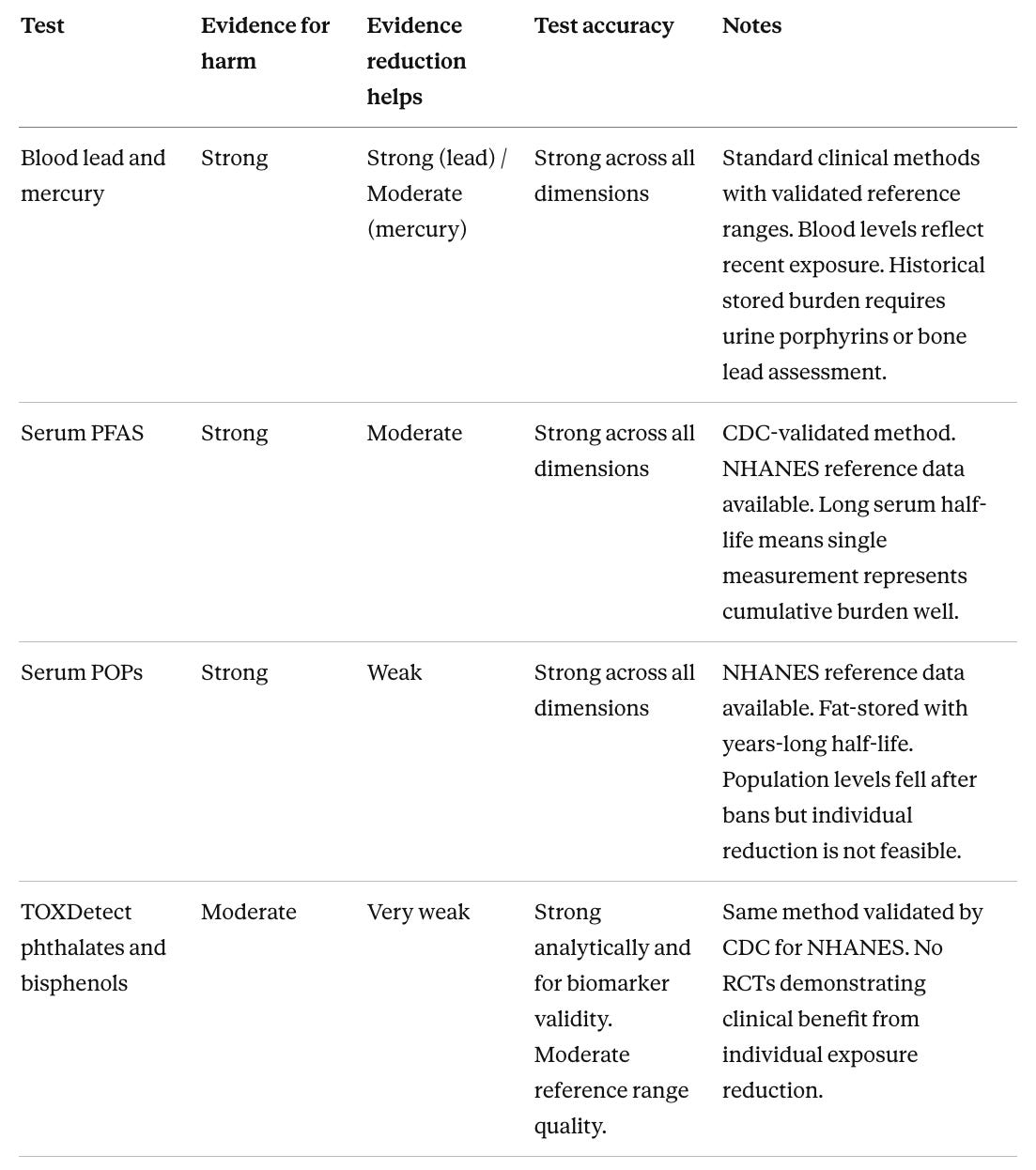
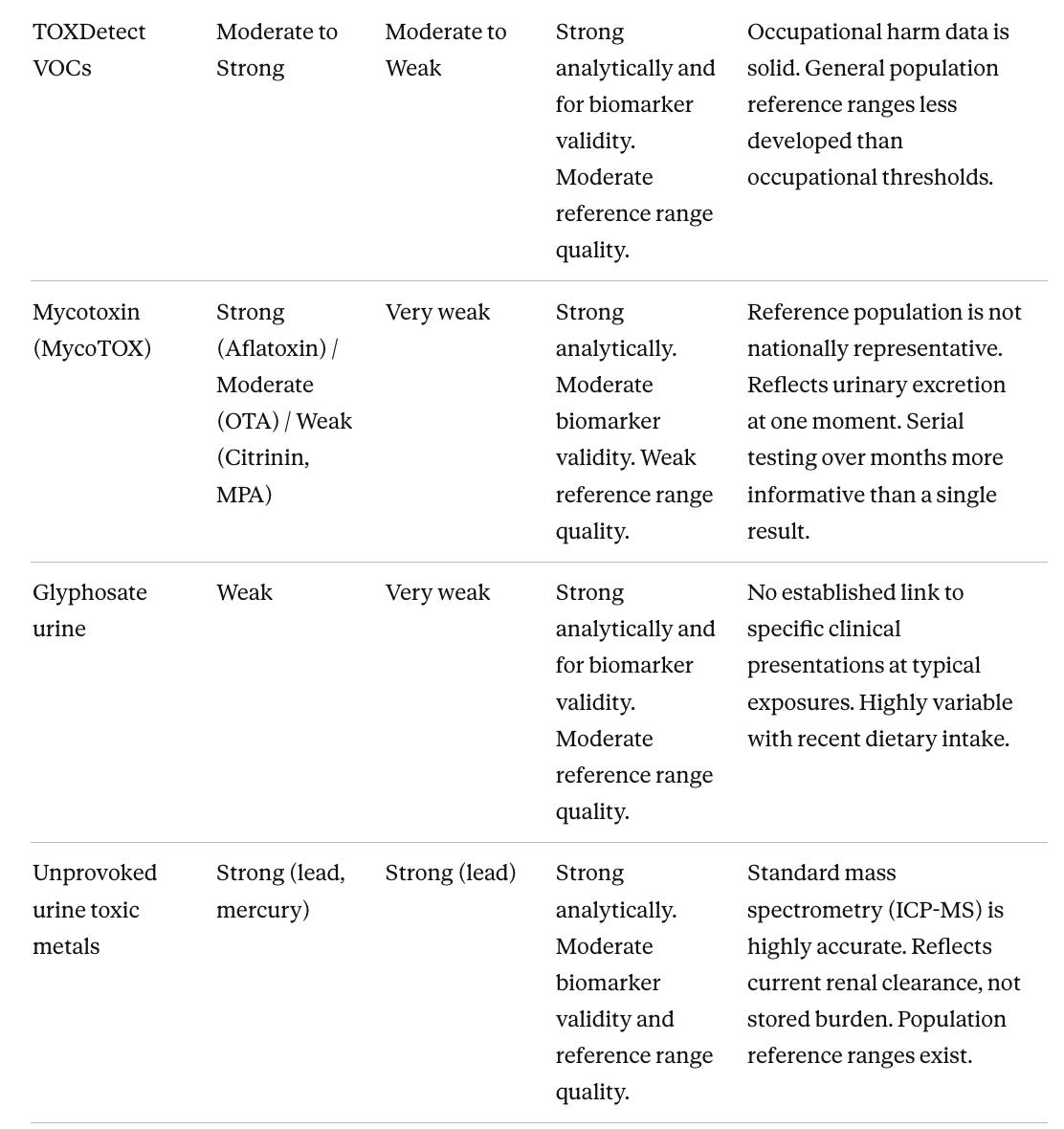
Evidence for harm, reduction benefit, and test accuracy
The first two columns below are rated on a four-level scale:
Strong reflects consistent human cohort data, dose-response relationships, or regulatory conclusions such as an IARC (International Agency for Research on Cancer) Group 1 classification, meaning confirmed evidence of causing cancer in humans.
Moderate reflects meaningful population associations or occupational data that fall short of that bar.
Weak reflects primarily animal data, inconsistent human findings, or contested epidemiology.
Very weak reflects a near absence of human clinical data.
The Test accuracy column summarizes three dimensions in plain language (analytical accuracy, biomarker validity, and reference range quality) since these frequently diverge. A Notes column gives the key practical caveat for each test. The most consistent pattern across the first two columns is the gap between harm evidence and reduction benefit. For nearly every chemical, the evidence that exposure causes harm is stronger than the evidence that reducing it improves outcomes. This reflects study design constraints, not a signal that reduction is ineffective. Lead is the clearest exception.
Interpreting results in context
The most useful approach is to look for convergent signals across multiple panels rather than treating any elevated marker in isolation. A low white blood cell count alongside markedly elevated OTA (ochratoxin A, a mold toxin) and HEMA (a urine marker of exposure to industrial solvents such as ethylene oxide) and low vitamin C on the OAT tells a coherent story of combined toxic burden with compromised immune function. No single result would be compelling on its own. Together they point in a clear direction.
A few practical points apply to all these tests. Serial testing carries more weight than a single measurement. A persistently elevated OTA on two tests a year apart is a much stronger signal of ongoing exposure than one elevated result. Collection conditions matter. Mycotoxin panels ideally use first-morning urine, and a dilute mid-day sample can underestimate true levels. Medications and supplements can skew results. Mycophenolate mofetil (CellCept) elevates urinary MPA (mycophenolic acid, a compound also produced by certain molds), NAC (N-acetyl cysteine) alters OAT glutathione markers, and high-dose vitamin B5 dramatically elevates the pantothenic acid marker on the OAT.
Source identification is more important than any treatment protocol. No binder or antioxidant regimen will produce lasting mycotoxin reduction in someone returning nightly to a water-damaged building. These panels are the beginning of an investigation, not the endpoint of one. They are most valuable when read alongside standard laboratory work (liver enzymes, complete blood count, and inflammatory markers).
On total test costs and whether it is worth it
Running the full suite described in this article would cost roughly $2,500 to $5,000 out of pocket. Almost none of it is covered by standard health insurance. The table below gives approximate costs by test category.
Serum POPs requires a specialty environmental laboratory and is not available through Quest or LabCorp in the standard catalog. The CIRS panel range is wide because the markers are typically ordered as separate assays rather than a bundled panel.
Whether testing is worth it depends entirely on clinical context. For someone with a specific unexplained finding that the clinical matching table maps to a particular exposure category (declining kidney function, unexplained thyroid disease, peripheral neuropathy), targeted testing in the $500–1,200 range is often a reasonable next step when standard workup has not provided an explanation. The reduce-and-retest cycle described in this article requires a baseline measurement to be meaningful.
The strongest case for testing, on both harm evidence and intervention benefit, is blood lead and mercury. Lead removal from gasoline and paint is one of the best-documented natural experiments in public health, and dietary reduction of high-mercury fish demonstrably lowers blood levels. Knowing where you stand gives you something actionable with evidence behind it.
Serum PFAS is worth adding, with strong harm evidence across thyroid, immune, cardiovascular, and cancer outcomes, but the intervention picture is more limited than it first appears. Existing body burden is not meaningfully reducible on a human timescale, given serum half-lives of 3 to 8 years. The value of testing is in identifying and stopping ongoing exposure sources such as drinking water and cookware, not in treating what is already there.
A mycotoxin panel is worth considering when the clinical picture is compatible, meaning unexplained kidney, liver, or immune findings or a known history of water-damaged building exposure. It’s worth keeping in mind that the intervention evidence is very limited. There are no clinical trials showing that reducing urinary OTA improves outcomes. That doesn’t mean that it won’t improve outcomes for an individual, but just that we don’t have the data from clinical trials to prove that this is helpful.
Those three together can be done for under $700 and cover the chemicals with the strongest harm data. That is a practical starting point for someone with compatible findings, approached with realistic expectations about what each result does and does not allow you to do.
Reducing exposure without testing
Testing identifies what is present. The actions below address the most prevalent and best-evidenced exposure sources regardless of what any panel might show, ranked roughly by the breadth of chemicals each action covers and the strength of the harm evidence behind those chemicals. I would argue that we all should be taking the below steps regardless of unexplained clinical findings or toxin testing. These actions are relatively easy and cheap to implement and the potential upside is large.
Filter drinking water. A reverse osmosis system or high-quality activated carbon filter reduces PFAS, lead, perchlorate, glyphosate, arsenic, and some VOCs (volatile organic compounds, airborne chemicals that can be inhaled or absorbed through skin) in a single intervention. This addresses more well-evidenced chemical exposures than any other single action. Both activated carbon filters and reverse osmosis are also among the most effective methods for removing pesticides from tap water, context I covered in Parkinson’s, Pesticides, and Proximity to Golf Courses.
Replace non-stick cookware. Pans coated with PTFE (polytetrafluoroethylene, the non-stick material in Teflon-style cookware) are a primary household PFAS source. Stainless steel, cast iron, or ceramic alternatives eliminate this route entirely.
Stop heating food in plastic and minimize plastic food contact. Microwaving in plastic, storing fatty foods in plastic containers, and running plastic through the dishwasher all increase phthalate and bisphenol migration into food. Glass and stainless steel storage removes this exposure category almost completely. I wrote about the evidence on microplastics leaching from plastic containers and their effects on brain health in Microplastics and the Brain.
Reduce canned food consumption or choose products from BPA-free lined cans. Epoxy can linings are a major BPA (bisphenol A, a synthetic compound used in many plastics and coatings) source, and this overlaps with broader plastic contact reduction. I covered what leaches from can linings and food packaging in depth in What’s Leaching Into Your Beans?
Wash all produce and prioritize organic for the highest-pesticide categories. Washing reduces surface residues but does not remove pesticides that have been absorbed into plant tissue, which includes glyphosate when it is applied as a pre-harvest treatment. Organochlorine pesticides (a long-lasting class of synthetic pesticides that includes DDT and accumulates in fatty tissue) bind to the waxy surface of produce in ways water does not fully remove, and peeling helps more than washing for those. For produce in the highest-residue categories, including strawberries, spinach, peppers, peaches, and grapes, studies still find detectable pesticide levels after washing. Switching those specific items to organic is the most practical approach for most households, and it does substantially reduce urinary glyphosate levels. Pesticide exposure is one of the more consistent environmental risk factors for Parkinson's disease, and I covered the evidence including the golf course proximity data in Parkinson's, Pesticides, and Proximity to Golf Courses.
Reduce fatty animal products from the highest-accumulation sources. PCBs (polychlorinated biphenyls, industrial chemicals now banned but still present in the environment), DDT/DDE (a legacy pesticide and its primary breakdown product), and PBDEs (polybrominated diphenyl ethers, used as flame retardants) concentrate in animal fat and accumulate further up the food chain. Farmed Atlantic salmon, full-fat conventional dairy, and fatty farm-raised fish carry the highest loads. Wild Pacific salmon and pasture-raised dairy are meaningfully lower. The evidence on dietary fats and brain health covers some of this source variation in more depth.
Limit high-mercury fish. Shark, swordfish, king mackerel, tilefish, and bigeye tuna carry the highest mercury concentrations because they are large, long-lived predators at the top of the marine food chain. Mercury accumulates in each fish from everything it eats and is not excreted, so it concentrates further with each step up the food chain. Small, short-lived species lower in the food chain such as wild salmon, sardines, anchovies, herring, and skipjack tuna carry far less mercury and are better choices fresh or frozen.
Switch to fragrance-free personal care and cleaning products. The word “fragrance” on a label is a common vehicle for undisclosed phthalates. Products from companies that publish full ingredient lists eliminate this source.
Inspect for and remediate any water-damaged areas in the home. Mycotoxin exposure from OTA, Citrinin, and MPA is driven by indoor mold. No dietary or supplement protocol will meaningfully reduce urinary mycotoxin levels while a mold source persists in the living environment.
Check for lead if the home was built before 1978 or has older plumbing. Lead paint and lead pipes remain the primary exposure route for many households. A water test and a visual paint inspection are low-cost starting points.
Reduce high-temperature cooking of starchy foods. Acrylamide forms when starchy foods such as potatoes, bread, cereals, and coffee are cooked at high heat. Lower cooking temperatures and less frequent consumption of heavily processed forms of these foods reduce dietary acrylamide. I recommend sautéing in olive oil as a primary cooking method. Olive oil is very good for brain health so there is an added benefit.
Vacuum with a HEPA (high-efficiency particulate air) filter and damp-mop hard floors regularly. Lead dust, pesticide residues, and flame retardants accumulate in household dust and are re-inhaled and ingested, particularly by children.
The first three actions (water filtration, cookware replacement, and eliminating plastic food contact) cover PFAS, lead, phthalates, and bisphenols simultaneously and are accessible for most households regardless of budget.
The science of environmental chemical testing is maturing rapidly. The gap between what we can measure and what we know how to do about it remains wide, but that gap is narrowing. For clinicians and patients navigating these results, the goal is calibrated, systematic interpretation that respects the limits of the evidence while taking meaningful exposures seriously.

Written by
Dr. Christin Glorioso, MD PhD
Dr. Glorioso is the founder and CEO of NeuroAge Therapeutics. With her background in neuroscience and medicine, she is dedicated to revolutionizing brain health and helping people maintain cognitive vitality.
Learn more about Dr. Glorioso